化工学报 ›› 2024, Vol. 75 ›› Issue (9): 3287-3296.DOI: 10.11949/0438-1157.20240254
徐宏标1,2( ), 杨亮1,2(
), 杨亮1,2( ), 李子栋1,2, 刘道平1,2
), 李子栋1,2, 刘道平1,2
收稿日期:2024-03-04
修回日期:2024-05-20
出版日期:2024-09-25
发布日期:2024-10-10
通讯作者:
杨亮
作者简介:徐宏标(1999—),男,硕士研究生,Xhongbiao2023@163.com
基金资助:
Hongbiao XU1,2( ), Liang YANG1,2(
), Liang YANG1,2( ), Zidong LI1,2, Daoping LIU1,2
), Zidong LI1,2, Daoping LIU1,2
Received:2024-03-04
Revised:2024-05-20
Online:2024-09-25
Published:2024-10-10
Contact:
Liang YANG
摘要:
提高海水水合储气速率和储气密度对水合物技术规模化应用至关重要。将一定浓度的NaCl溶液与疏水性气相纳米二氧化硅高速搅拌分散成微米级盐水微滴。8.0 MPa、274.15 K条件下,利用不同硅含量的盐水微滴进行水合储甲烷实验,研究甲烷水合物生成动力学特性。结果表明,硅含量2.5%(质量分数)的盐水微滴分散性和储气性能最佳,储气量达141.01 cm3·cm-3,储气速率达7.37 cm3·cm-3·min-1。进一步将该硅含量盐水微滴填充至开孔泡沫铜中,构建盐水微滴/泡沫铜复合水合储气体系。研究发现,泡沫铜三维巢状金属骨架能显著加速水合反应热的转移,提高微滴水合储气性能。5.0~8.0 MPa下,相较于盐水微滴单一体系,复合体系水合储气量提高4.72%~21.70%,最大储气速率提高38.25%~110.58%。
中图分类号:
徐宏标, 杨亮, 李子栋, 刘道平. 盐水微滴/泡沫铜复合体系中甲烷水合物生成动力学研究[J]. 化工学报, 2024, 75(9): 3287-3296.
Hongbiao XU, Liang YANG, Zidong LI, Daoping LIU. Kinetics of methane hydrate formation in saline droplets/copper foam composite system[J]. CIESC Journal, 2024, 75(9): 3287-3296.
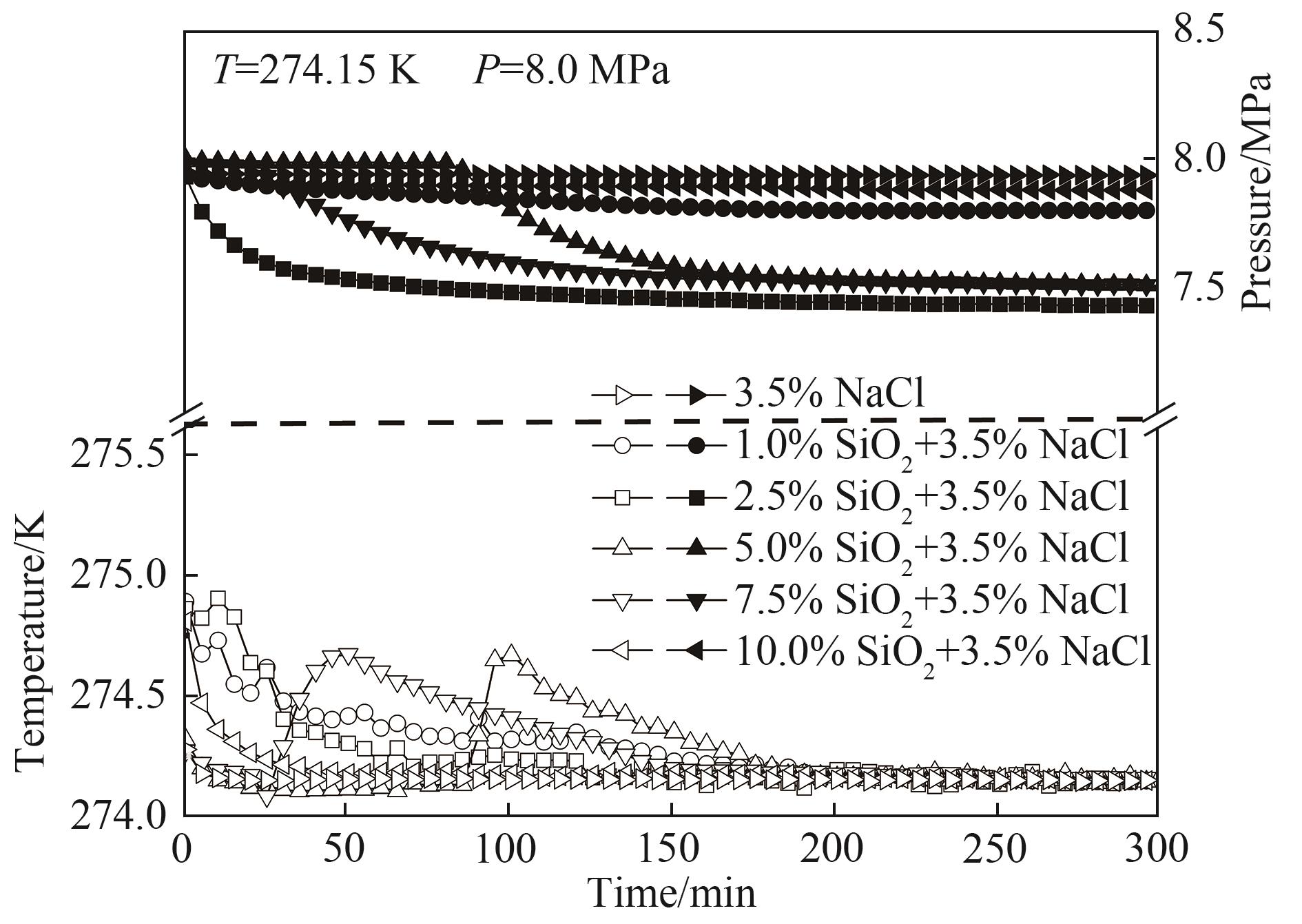
图4 甲烷水合物在不同硅含量盐水微滴中生成过程温压变化
Fig.4 Variation of temperature and pressure during the formation of methane hydrates in SD with different silicon contents
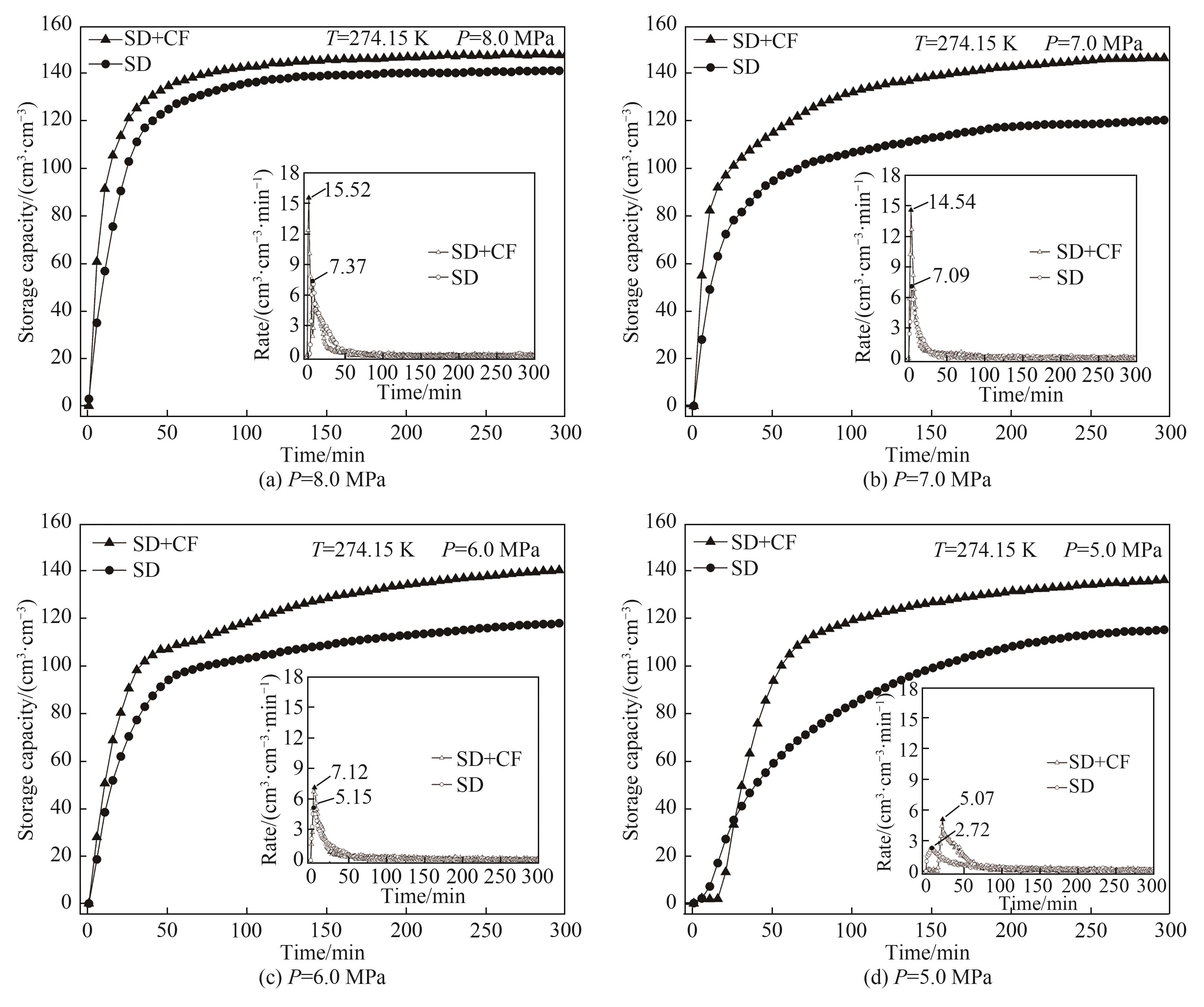
图7 不同压力下硅含量2.5%的盐水微滴及其泡沫铜填充体系水合储气量和储气速率
Fig.7 Gas storage capacity and storage rate of SD with 2.5% silicon content and its CF filling system under different pressures
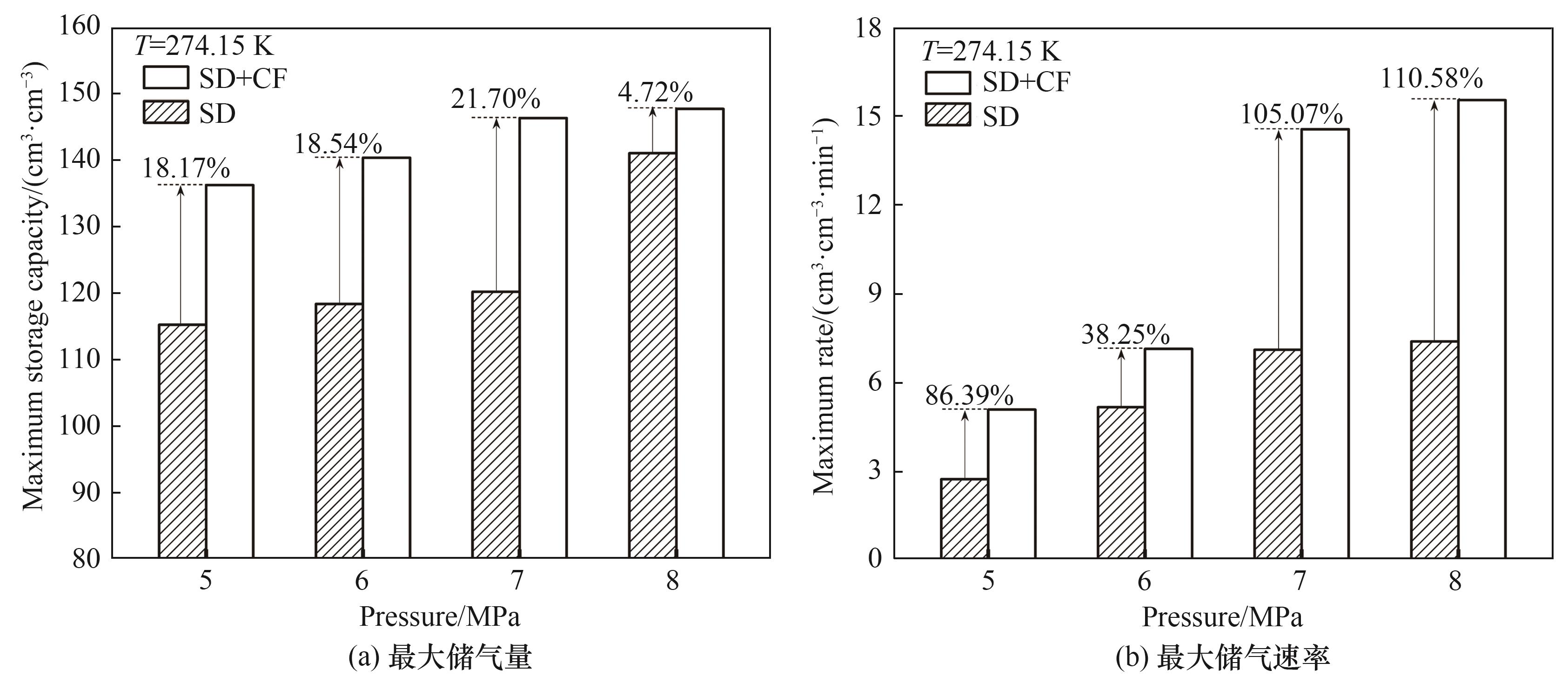
图8 不同压力下硅含量2.5%的盐水微滴及其泡沫铜填充体系最大储气量和储气速率对比
Fig.8 Comparison of maximum storage capacity and maximum storage rate of SD with 2.5% silicon content and its CF filling system
| 1 | Sloan Jr E D. Fundamental principles and applications of natural gas hydrates[J]. Nature, 2003, 426(6964): 353-363. |
| 2 | Hao W F, Wang J Q, Fan S S, et al. Evaluation and analysis method for natural gas hydrate storage and transportation processes[J]. Energy Conversion and Management, 2008, 49(10): 2546-2553. |
| 3 | Kumar R, Linga P, Moudrakovski I, et al. Structure and kinetics of gas hydrates from methane/ethane/propane mixtures relevant to the design of natural gas hydrate storage and transport facilities[J]. AIChE Journal, 2008, 54(8): 2132-2144. |
| 4 | 陈光进, 孙长宇, 马庆兰. 气体水合物科学与技术[M]. 北京: 化学工业出版社, 2008. |
| Chen G J, Sun C Y, Ma Q L. Gas Hydrate Science and Technology[M]. Beijing: Chemical Industry Press, 2008. | |
| 5 | Li C X, Yang L, Liu D P, et al. Accelerated methane storage in clathrate hydrates using the natural tobacco[J]. Energy, 2022, 241: 122549. |
| 6 | Veluswamy H P, Kumar A, Seo Y, et al. A review of solidified natural gas (SNG) technology for gas storage via clathrate hydrates[J]. Applied Energy, 2018, 216: 262-285. |
| 7 | Koh C A, Sloan E D. Natural gas hydrates: recent advances and challenges in energy and environmental applications[J]. AIChE Journal, 2007, 53(7): 1636-1643. |
| 8 | Boswell R, Collett T S. Current perspectives on gas hydrate resources[J]. Energy & Environmental Science, 2011, 4(4): 1206-1215. |
| 9 | Chong Z R, Yang S H B, Babu P, et al. Review of natural gas hydrates as an energy resource: prospects and challenges[J]. Applied Energy, 2016, 162: 1633-1652. |
| 10 | Yang L, Li C X, Pei J H, et al. Enhanced clathrate hydrate phase change with open-cell copper foam for efficient methane storage[J]. Chemical Engineering Journal, 2022, 440: 135912. |
| 11 | Linga P, Daraboina N, Ripmeester J A, et al. Enhanced rate of gas hydrate formation in a fixed bed column filled with sand compared to a stirred vessel[J]. Chemical Engineering Science, 2012, 68(1): 617-623. |
| 12 | Zhong D L, Lu Y Y, Sun D J, et al. Performance evaluation of methane separation from coal mine gas by gas hydrate formation in a stirred reactor and in a fixed bed of silica sand[J]. Fuel, 2015, 143: 586-594. |
| 13 | Xiao P, Yang X M, Sun C Y, et al. Enhancing methane hydrate formation in bulk water using vertical reciprocating impact[J]. Chemical Engineering Journal, 2018, 336: 649-658. |
| 14 | Fukumoto K, Tobe J I, Ohmura R, et al. Hydrate formation using water spraying in a hydrophobic gas: a preliminary study[J]. AIChE Journal, 2001, 47(8): 1899-1904. |
| 15 | Yamamura K, Fukuzaki J I, Mori Y H. Clathrate hydrate formation using liquid jets impinging on each other: an observational study using paired water jets or water and methylcyclohexane jets[J]. Chemical Engineering Science, 2011, 66(9): 1844-1858. |
| 16 | Partoon B, Sabil K M, Lau K K, et al. Production of gas hydrate in a semi-batch spray reactor process as a means for separation of carbon dioxide from methane[J]. Chemical Engineering Research and Design, 2018, 138: 168-175. |
| 17 | Maini B B, Bishnoi P R. Experimental investigation of hydrate formation behaviour of a natural gas bubble in a simulated deep sea environment[J]. Chemical Engineering Science, 1981, 36(1): 183-189. |
| 18 | Luo Y T, Zhu J H, Fan S S, et al. Study on the kinetics of hydrate formation in a bubble column[J]. Chemical Engineering Science, 2007, 62(4): 1000-1009. |
| 19 | 方书起, 张欣悦, 李思齐, 等. 撞击流式反应器内水合物法分离沼气中CO2研究[J]. 化工学报, 2020, 71(5): 2099-2108. |
| Fang S Q, Zhang X Y, Li S Q, et al. Investigation on separation of CO2 from biogas by hydrate method in impinging stream reactor[J]. CIESC Journal, 2020, 71(5): 2099-2108. | |
| 20 | 刘有智, 邢银全, 崔磊军. 超重力旋转填料床中天然气水合物含气量研究[J]. 化工进展, 2007, 26(6): 853-856. |
| Liu Y Z, Xing Y Q, Cui L J. Experimental study on capacity of gas hydrate in a rotating packed bed[J]. Chemical Industry and Engineering Progress, 2007, 26(6): 853-856. | |
| 21 | 胡慧慧, 杨亮, 刘道平, 等. 低剂量超吸水树脂溶液微滴中甲烷水合物生成动力学[J]. 化工学报, 2022, 73(10): 4659-4667. |
| Hu H H, Yang L, Liu D P, et al. Kinetics of methane hydrate formation in droplets of low-dose superabsorbent resin solution[J]. CIESC Journal, 2022, 73(10): 4659-4667. | |
| 22 | Yoslim J, Linga P, Englezos P. Enhanced growth of methane-propane clathrate hydrate crystals with sodium dodecyl sulfate, sodium tetradecyl sulfate, and sodium hexadecyl sulfate surfactants[J]. Journal of Crystal Growth, 2010, 313(1): 68-80. |
| 23 | Kang S P, Lee J W. Kinetic behaviors of CO2 hydrates in porous media and effect of kinetic promoter on the formation kinetics[J]. Chemical Engineering Science, 2010, 65(5): 1840-1845. |
| 24 | Zhou L, Sun Y, Zhou Y P. Enhancement of the methane storage on activated carbon by preadsorbed water[J]. AIChE Journal, 2002, 48(10): 2412-2416. |
| 25 | Song Y M, Wang F, Luo S J, et al. Methane hydrate formation improved by water-soluble carbon nanotubes via π-π conjugated molecules functionalization[J]. Fuel, 2019, 243: 185-191. |
| 26 | Andres-Garcia E, Dikhtiarenko A, Fauth F, et al. Methane hydrates: nucleation in microporous materials[J]. Chemical Engineering Journal, 2019, 360: 569-576. |
| 27 | Yang M J, Song Y C, Jiang L L, et al. Effects of operating mode and pressure on hydrate-based desalination and CO2 capture in porous media[J]. Applied Energy, 2014, 135: 504-511. |
| 28 | Stern L A, Kirby S H, Durham W B. Peculiarities of methane clathrate hydrate formation and solid-state deformation, including possible superheating of water ice[J]. Science, 1996, 273(5283): 1843-1848. |
| 29 | Xiao P, Li J J, Chen W, et al. Enhanced formation of methane hydrate from active ice with high gas uptake[J]. Nature Communications, 2023, 14: 8068. |
| 30 | Wang W X, Bray C L, Adams D J, et al. Methane storage in dry water gas hydrates[J]. Journal of the American Chemical Society, 2008, 130(35): 11608-11609. |
| 31 | Carter B O, Wang W X, Adams D J, et al. Gas storage in “dry water” and “dry gel” clathrates[J]. Langmuir, 2010, 26(5): 3186-3193. |
| 32 | Yang L, Lan X, Liu D P, et al. Multi-cycle methane hydrate formation in micro droplets of gelatinous dry solution[J]. Chemical Engineering Journal, 2019, 374: 802-810. |
| 33 | Yang L, Liu Z Z, Liu D P, et al. Enhanced natural gas hydrates formation in the suspension with metal particles and fibers[J]. Journal of Molecular Liquids, 2020, 301: 112410. |
| 34 | Hu P, Wu G Z, Zi M C, et al. Effects of modified metal surface on the formation of methane hydrate[J]. Fuel, 2019, 255: 115720. |
| 35 | Li R L, Liu D P, Yang L, et al. Rapid methane hydrate formation in aluminum honeycomb[J]. Fuel, 2019, 252: 574-580. |
| 36 | Bhattacharya A, Calmidi V V, Mahajan R L. Thermophysical properties of high porosity metal foams[J]. International Journal of Heat and Mass Transfer, 2002, 45(5): 1017-1031. |
| 37 | Veluswamy H P, Kumar A, Kumar R, et al. Investigation of the kinetics of mixed methane hydrate formation kinetics in saline and seawater[J]. Applied Energy, 2019, 253: 113515. |
| 38 | Redlich O, Kwong J N S. On the thermodynamics of solutions (Ⅴ): An equation of state. Fugacities of gaseous solutions[J]. Chemical Reviews, 1949, 44(1): 233-244. |
| 39 | Nesterov A N, Reshetnikov A M. Combined effect of NaCl and sodium dodecyl sulfate on the mechanism and kinetics of methane hydrate formation in an unstirred system[J]. Journal of Natural Gas Science and Engineering, 2022, 99: 104424. |
| 40 | Sloan Jr E D, Koh C A, Koh C A. Clathrate Hydrates of Natural Gases[M]. Boca Raton: CRC Press, 2007. |
| 41 | Ohmura R, Ogawa M, Yasuoka K, et al. Statistical study of clathrate-hydrate nucleation in a water/hydrochlorofluorocarbon system: search for the nature of the “memory effect”[J]. The Journal of Physical Chemistry B, 2003, 107(22): 5289-5293. |
| [1] | 关朝阳, 黄国庆, 张一喃, 陈宏霞, 杜小泽. 泡沫铜导离气泡强化流动沸腾换热实验研究[J]. 化工学报, 2024, 75(5): 1765-1776. |
| [2] | 刘礼豪, 黄婷, 雍宇, 罗昕浩, 赵泽明, 宋尚飞, 史博会, 陈光进, 宫敬. 含粉砂盐水体系甲烷水合物生成与固相沉积规律[J]. 化工学报, 2024, 75(5): 1987-2000. |
| [3] | 马旭, 滕亚栋, 刘杰, 王宇璐, 张鹏, 张莲海, 姚万龙, 展静, 吴青柏. 喷雾法水合物法捕集分离烟道气中CO2[J]. 化工学报, 2024, 75(5): 2001-2016. |
| [4] | 李彬, 徐正虎, 姜爽, 张天永. 双氧水催化氧化法清洁高效合成促进剂CBS[J]. 化工学报, 2023, 74(7): 2919-2925. |
| [5] | 邢美波, 张中天, 景栋梁, 张洪发. 磁调控水基碳纳米管协同多孔材料强化相变储/释能特性[J]. 化工学报, 2023, 74(7): 3093-3102. |
| [6] | 胡晗, 杨亮, 李春晓, 刘道平. 天然烟浸滤液水合物法储甲烷动力学研究[J]. 化工学报, 2023, 74(3): 1313-1321. |
| [7] | 宋彭辉, 张国栋, 王飞. 气体水合物高效生成、分离与储存一体化技术研究[J]. 化工学报, 2023, 74(11): 4670-4678. |
| [8] | 张炜, 李昊阳, 徐纯刚, 李小森. 气体水合物生成微观机理及分析方法研究进展[J]. 化工学报, 2022, 73(9): 3815-3827. |
| [9] | 胡慧慧, 杨亮, 刘道平, 张柯. 低剂量超吸水树脂溶液微滴中甲烷水合物生成动力学[J]. 化工学报, 2022, 73(10): 4659-4667. |
| [10] | 张天永, 杨坤龙, 崔现宝, 李彬, 宋禹昕, 姜爽. 橡胶促进剂NS的绿色合成工艺与应用研究进展[J]. 化工学报, 2021, 72(2): 876-885. |
| [11] | 裴俊华, 杨亮, 汪鑫, 胡晗, 刘道平. 泡沫铜强化甲烷水合物生成动力学实验研究[J]. 化工学报, 2021, 72(11): 5751-5760. |
| [12] | 刘明, 徐哲. 甲烷水合物声子导热及量子修正[J]. 化工学报, 2020, 71(4): 1424-1431. |
| [13] | 辛亚男, 张建文, 张淑珍, 姜爱国. 螺旋内槽管内天然气-水-表面活性剂体系的水合物生成动力学计算[J]. 化工学报, 2018, 69(6): 2463-2473. |
| [14] | 施尚, 余建祖, 陈梦东, 高红霞, 谢永奇. 基于泡沫铜/石蜡的锂电池热管理系统性能[J]. 化工学报, 2017, 68(7): 2678-2683. |
| [15] | 李文昭, 潘振, 马贵阳, 李存磊, 仇阳, 刘畅达. 表面活性剂吸附对促进甲烷水合物生成效果的影响[J]. 化工学报, 2017, 68(4): 1542-1549. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备 11010102001995号
京公网安备 11010102001995号