化工学报 ›› 2021, Vol. 72 ›› Issue (5): 2810-2816.DOI: 10.11949/0438-1157.20201252
收稿日期:2020-09-01
修回日期:2020-10-26
出版日期:2021-05-05
发布日期:2021-05-05
通讯作者:
王家德
作者简介:叶志平(1987—),男,博士,副教授,基金资助:
YE Zhiping( ),ZHOU Danfei,LIU Zifeng,ZHOU Qingqing,WANG Jiade(
),ZHOU Danfei,LIU Zifeng,ZHOU Qingqing,WANG Jiade( )
)
Received:2020-09-01
Revised:2020-10-26
Online:2021-05-05
Published:2021-05-05
Contact:
WANG Jiade
摘要:
采用原位红外光谱法系统地研究了对甲基苯磺酸(p-TSA)在钛基二氧化铅(Ti/PbO2)电极上的电氧化反应信息。循环伏安曲线显示,Ti/PbO2对p-TSA有良好的电氧化活性,直接氧化电位区间为0.55~0.9 V。多步电位阶跃FTIRS(MSFTIRS)和时间分辨FTIRS(TRFTIRS)分析显示,电压小于1000 mV时,p-TSA主要发生了磺酸基的脱落和苯环侧链甲基的氧化;电压大于1000 mV时,p-TSA的苯环骨架被破坏,同时生成了酸、醇、酮。反应动力学研究结果表明,p-TSA电氧化表观速率常数K与电流密度j呈线性关系,化学需氧量(COD)去除量随比电荷增大而增加,平均电流效率则呈下降趋势。
中图分类号:
叶志平, 周丹飞, 刘梓锋, 周青青, 王家德. 对甲基苯磺酸在Ti/PbO2电极上的电氧化反应信息[J]. 化工学报, 2021, 72(5): 2810-2816.
YE Zhiping, ZHOU Danfei, LIU Zifeng, ZHOU Qingqing, WANG Jiade. Electro-oxidation information of p-toluene sulfonic acid on Ti/PbO2 electrode[J]. CIESC Journal, 2021, 72(5): 2810-2816.
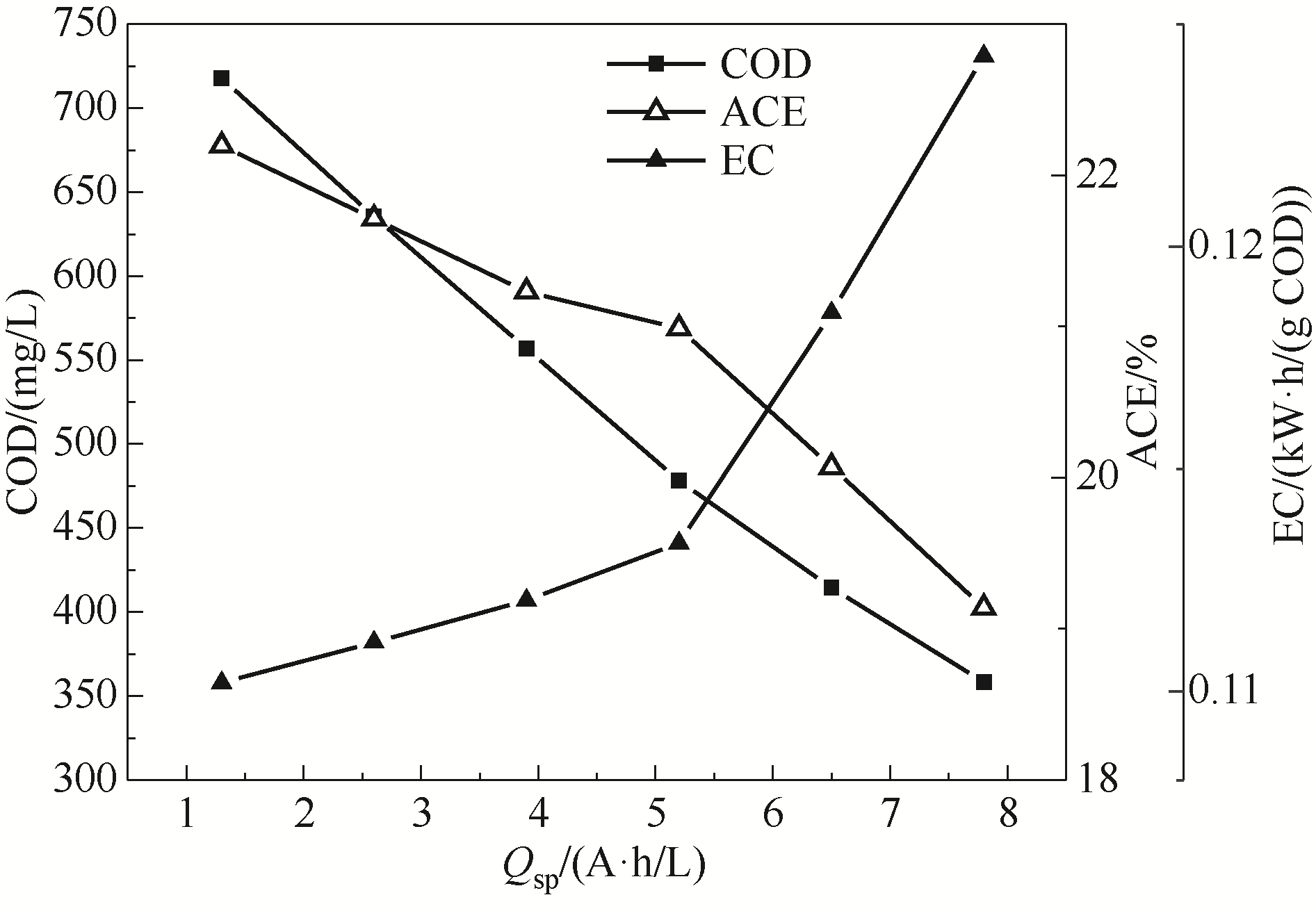
图7 比电荷(Qsp)对p-TSA去除、能耗(EC)和电流效率(ACE)的影响
Fig.7 The influence of specific electric charge(Qsp) on p-TSA romoval, energy consumption, and average current efficiency
| 1 | 杨倩. 对甲基苯磺酸高效降解菌的筛选、代谢机制及其在废水生物强化处理中的应用[D].南京: 南京农业大学, 2017. |
| Yang Q. Screening and metabolic mechanism of p-toluenesulfonic acid degrading straino and ITS application in bioaugmentation of wastewater[D]. Nanjing: Nanjing Agricultural University, 2017. | |
| 2 | 唐加翠. 对甲苯磺酸废水强化预处理研究[D]. 上海: 华东理工大学, 2011. |
| Tang J C. Research on pretreatment of p-toluene sulfonic acid wastewater[D]. Shanghai: East China University of Science and Technology, 2011. | |
| 3 | Sun Y, Li X, Zheng W, et al. Effect of functional group density of anion exchange resins on removal of p-toluene sulfonic acid from aqueous solution[J]. Applied Sciences, 2019, 10(1): 1-14. |
| 4 | Borthakur S, Srivastava R C. Kinetics of removal of p-toluene sulphonic acid from concentrated solution by granular activated carbon[J]. Journal of Chemical Technology & Biotechnology, 1991, 51(4): 497-506. |
| 5 | 张凌, 陶莹, 常志显, 等. 臭氧法降解水中对甲基苯磺酸的动力学研究[J].环境科学学报, 2011, 31(10): 2185-2191. |
| Zhang L, Tao Y, Chang Z X, et al. Kinetics of p-toluene sulfonic acid degradation by ozone in aqueous solutions[J]. Acta Scientiae Circumstantiae, 2011, 31(10): 2185-2191. | |
| 6 | Cao H, Feng Q. Kinetics of photocatalytic degradation of p-toluene sulfonic acid on TiO2 surface[J]. Asian Journal of Chemistry, 2013, 25(18): 10195-10198. |
| 7 | Dai Q Z, Shen H, Xia Y J, et al. Typical rare earth doped lead dioxide electrode: preparation and application[J]. International Journal of Electrochemical Science, 2012, 7(10): 10054-10062. |
| 8 | 余丽娜, 宋玉栋, 周岳溪, 等. 三维电极法处理丙烯酸丁酯生产废水的研究[J].环境工程学报, 2011, 5(5): 1025-1028. |
| Yu L N, Song Y D, Zhou Y X, et al. Treatment of butyl acrylate production wastewater by three-dimensional electrode process[J]. Chinese Journal of Environmental Engineering, 2011, 5(5): 1025-1028. | |
| 9 | Taleb Z, Montilla F, Quijada C, et al. Electrochemical and in situ FTIR study of o-cresol on platinum electrode in acid medium[J]. Electrocatalysis, 2014, 5(2): 186-192. |
| 10 | Ye J Y, Jiang Y X, Sheng T, et al. In-situ FTIR spectroscopic studies of electrocatalytic reactions and processes[J]. Nano Energy, 2016, 29(SI): 414-427. |
| 11 | Gomes J F, Garcia A C, Gasparotto L H S, et al. Influence of silver on the glycerol electro-oxidation over AuAg/C catalysts in alkaline medium: a cyclic voltammetry and in situ FTIR spectroscopy study[J]. Electrochimica Acta, 2014, 144: 361-368. |
| 12 | Borrás C, Laredo T, Mostany J, et al. Study of the oxidation of solutions of p-chlorophenol and p-nitrophenol on Bi-doped PbO2 electrodes by UV-Vis and FTIR in situ spectroscopy[J]. Electrochimica Acta, 2004, 49(4): 641-648. |
| 13 | Labiadh L, Barbucci A, Carpanese M P, et al. Comparative depollution of methyl orange aqueous solutions by electrochemical incineration using TiRuSnO2, BDD and PbO2 as high oxidation power anodes[J]. Journal of Electroanalytical Chemistry, 2016, 766: 94-99. |
| 14 | Feng Y J, Li X Y. Electro-catalytic oxidation of phenol on several metal-oxide electrodes in aqueous solution[J]. Water Research, 2003, 37(10): 2399-2407. |
| 15 | He Y P, Huang W M, Chen R L, et al. Anodic oxidation of aspirin on PbO2, BDD and porous Ti/BDD electrodes: mechanism, kinetics and utilization rate[J]. Separation & Purification Technology, 2015, 156: 124-131. |
| 16 | 甘永平, 马淳安. 疏水性二氧化铅电极的镀制及强化寿命研究[J]. 化学世界, 2002, 43(11): 584-586. |
| Gan Y P, Ma C A. Eelctroplating and strengthening lifetime of PbO2 hydrophobic electrode[J]. Chemical World, 2002, 43(11): 584-586. | |
| 17 | 王家德, 袁通斌, 周丹飞, 等. 基于原位红外光谱的水相苯酚电氧化机理研究[J].化工学报, 2019, 70(12): 4821-4827. |
| Wang J D, Yuan T B, Zhou D F, et al. Mechanism of phenol electro-oxidation in aqueous solution based on in situ infrared spectroscopy[J]. CIESC Journal, 2019, 70(12): 4821-4827. | |
| 18 | 蔡少卿, 何月峰, 戴启洲, 等. 臭氧氧化对甲基苯磺酸的动力学模型[J].化工学报, 2011, 62(5): 1414-1420. |
| Cai S Q, He Y F, Dai Q Z, et al. Kinetic model on ozonation of p-toluene sulfonic acid[J]. CIESC Journal, 2011, 62(5): 1414-1420. | |
| 19 | Wang L Z, Yang S X, Wu B, et al. The influence of anode materials on the kinetics toward electrochemical oxidation of phenol[J]. Electrochimica Acta, 2016, 206: 270-277. |
| 20 | Song S, Fan J, He Z, et al. Electrochemical degradation of azo dye CI Reactive Red 195 by anodic oxidation on Ti, SnO2-Sb, PbO2 electrodes[J]. Electrochimica Acta, 2010, 55(11): 3606-3613. |
| 21 | 翁诗甫.傅里叶变换红外光谱分析[M]. 3版. 北京: 化学工业出版社, 2017. |
| Weng S F. Fourier Transform Infrared Spectroscopy[M]. 3rd ed. Beijing: Chemical Industry Press, 2017. | |
| 22 | 张凌, 楚红英, 常志显, 等. 超声波光催化协同降解对甲基苯磺酸水溶液的机理研究[J].环境工程学报, 2011, 5(4): 819-824. |
| Zhang L, Chu H Y, Chang Z X, et al. Study on mechanism of degradation 4-toluene sulfonic acid by sonophotocatalysis [J]. Chinese Journal of Environmental Engineering, 2011, 5(4): 819-824. | |
| 23 | Li M C, Bao D D, Ma C A. Studies on electrochemical hydrodebromination mechanism of 2, 5-dibromobenzoic acid on Ag electrode by in situ FTIR spectroscopy[J]. Electrochimica Acta, 2011, 56(11): 4100-4104. |
| 24 | 叶咏薇, 王希, 郑万芳, 等. 间溴苯甲酸在Pt电极上的电氧化反应[J].物理化学学报, 2013, 29(3): 553-558. |
| Ye Y W, Wang X, Zheng W F, et al. Electrooxidation reaction of 3-bromobenzoic acid on Pt electrode[J]. Acta Phys. -Chim. Sin., 2013, 29 (3): 553-558. | |
| 25 | Chumillas S, Palomaki T, Zhang M, et al. Analysis of catechol, 4-methylcatechol and dopamine electrochemical reactions on different substrate materials and pH conditions[J]. Electrochimica Acta, 2018, 292: 309-321. |
| 26 | Vigier F, Coutanceau C, Hahn F, et al. On the mechanism of ethanol electro-oxidation on Pt and PtSn catalysts: electrochemical and in situ IR reflectance spectroscopy studies[J]. Journal of Electroanalytical Chemistry, 2004, 563(1): 81-89. |
| 27 | You L X, Fang Y M, Guo J W, et al. Mechanism of electro-catalytic oxidation of shikimic acid on Cu electrode based on in situ FTIRS and theoretical calculations[J]. Electrochimica Acta, 2011, 58: 165-171. |
| 28 | Chen L C, Uchida T, Chang H C, et al. Adsorption and oxidation of glycine on Au electrode: an in situ surface-enhanced infrared study[J]. Electrochemistry Communications, 2013, 34: 56-59. |
| 29 | Salavagione H J, Joaquín A P, Pedro G T, et al. Spectroelectrochemical study of the oxidation of aminophenols on platinum electrode in acid medium[J]. Journal of Electroanalytical Chemistry, 2004, 565(2): 375-383. |
| 30 | Planes G A, Moran E, Rodriguez J L, et al. Electrochemical behavior of benzaldehyde on polycrystalline platinum: an in situ FTIR and DEMS study[J]. Langmuir, 2003, 19(21): 8899-8906. |
| 31 | Clémence L, Frédéric M, Martin V, et al. Degradation of carbon-supported platinum-group-metal electrocatalysts in alkaline media studied by in situ Fourier transform infrared spectroscopy and identical-location transmission electron microscopy[J]. ACS Catalysis, 2019, 9(6): 5613-5622. |
| 32 | Zheng J, Yan K, Wu Z, et al. Effective removal of sulfanilic acid from water using a low-pressure electrochemical RuO2-TiO2@Ti/PVDF composite membrane[J]. Frontiers in Chemistry, 2018, 6: 395. |
| [1] | 程成, 段钟弟, 孙浩然, 胡海涛, 薛鸿祥. 表面微结构对析晶沉积特性影响的格子Boltzmann模拟[J]. 化工学报, 2023, 74(S1): 74-86. |
| [2] | 汪林正, 陆俞冰, 张睿智, 罗永浩. 基于分子动力学模拟的VOCs热氧化特性分析[J]. 化工学报, 2023, 74(8): 3242-3255. |
| [3] | 张蒙蒙, 颜冬, 沈永峰, 李文翠. 电解液类型对双离子电池阴阳离子储存行为的影响[J]. 化工学报, 2023, 74(7): 3116-3126. |
| [4] | 禹进, 余彬彬, 蒋新生. 一种基于虚拟组分的燃烧调控化学作用量化及分析方法研究[J]. 化工学报, 2023, 74(3): 1303-1312. |
| [5] | 陈晨, 杨倩, 陈云, 张睿, 刘冬. 不同氧浓度下煤挥发分燃烧的化学动力学研究[J]. 化工学报, 2022, 73(9): 4133-4146. |
| [6] | 陈玉弓, 陈昊, 黄耀松. 基于分子反应动力学模拟的六甲基二硅氧烷热解机理研究[J]. 化工学报, 2022, 73(7): 2844-2857. |
| [7] | 何聪, 钟文琪, 周冠文, 陈曦. 高海拔地区水泥生料悬浮炉分解特性研究[J]. 化工学报, 2022, 73(5): 2120-2129. |
| [8] | 王敏, 程金兰, 李鑫, 陆晶晶, 尹崇鑫, 戴红旗. 酸性助水溶剂脱除木质素机理分析[J]. 化工学报, 2022, 73(5): 2206-2221. |
| [9] | 杨霄, 丁锐, 李墨含, 宋正昶. 氧浓度对微通道内甲烷均相/非均相耦合反应特性的影响[J]. 化工学报, 2022, 73(12): 5427-5437. |
| [10] | 龚翔, 李林森, 姜召. PdCo/SiO2双金属催化剂用于杂环储氢载体的高效脱氢[J]. 化工学报, 2022, 73(10): 4448-4460. |
| [11] | 张利合, 张凡, 李昌伦, 许德平, 徐振刚, 王永刚. BGL煤气化动力学模型构建与验证[J]. 化工学报, 2022, 73(10): 4668-4678. |
| [12] | 赵旭, 卜昌盛, 王昕晔, 张鑫, 程晓磊, 王乃继, 朴桂林. 铁基载氧体辅助无烟煤焦富氧燃烧动力学分析[J]. 化工学报, 2022, 73(1): 384-392. |
| [13] | 王婷婷, 曾玺, 韩振南, 王芳, 武鹏, 许光文. 微型流化床中生物质半焦水蒸气气化反应特性及动力学研究[J]. 化工学报, 2022, 73(1): 294-307. |
| [14] | 陈旭杰, 吕喜蕾, 史欢欢, 郑丽萍, 魏茜文, 田鹏辉, 蒋雨希, 吕秀阳. HBr-MgBr2催化己糖二酸脱水环合制备2,5-呋喃二甲酸的研究[J]. 化工学报, 2021, 72(9): 4658-4664. |
| [15] | 平甜甜, 尹鑫, 董玉, 申淑锋. 有机胺非水溶液吸收CO2的动力学研究进展[J]. 化工学报, 2021, 72(8): 3968-3983. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备 11010102001995号
京公网安备 11010102001995号