化工学报 ›› 2022, Vol. 73 ›› Issue (12): 5615-5624.DOI: 10.11949/0438-1157.20220692
杨伟( ), 王昱杰, 方凯斌, 邹汉波, 陈胜洲(
), 王昱杰, 方凯斌, 邹汉波, 陈胜洲( ), 刘自力
), 刘自力
收稿日期:2022-05-06
修回日期:2022-10-21
出版日期:2022-12-05
发布日期:2023-01-17
通讯作者:
陈胜洲
作者简介:杨伟(1982—),男,博士,高级工程师,wyang@gzhu.edu.cn
基金资助:
Wei YANG( ), Yujie WANG, Kaibin FANG, Hanbo ZOU, Shengzhou CHEN(
), Yujie WANG, Kaibin FANG, Hanbo ZOU, Shengzhou CHEN( ), Zili LIU
), Zili LIU
Received:2022-05-06
Revised:2022-10-21
Online:2022-12-05
Published:2023-01-17
Contact:
Shengzhou CHEN
摘要:
采用共沉淀法,以草酸为沉淀剂,过渡金属锰作为钴的替代元素,制备了系列不同钴锰比例的镍钴锰铝锂(LiNi0.8Co0.10-y Mn0.05+y Al0.05O2,y=0.01,0.02,0.03,0.04)四元材料,研究锰对钴的逐步取代对镍钴锰铝锂四元材料的性能产生的影响。实验结果表明,当y=0.02时的LiNi0.8Co0.08Mn0.07Al0.05O2材料具有最好的形貌、结构发育程度以及电化学性能。钴锰共掺杂对合成的LiNi0.8Co0.10-y Mn0.05+y Al0.05O2材料产生了协同作用,且锰掺杂比例低于钴时可以提升材料电化学容量,当锰掺杂比例超过钴时容量开始下降,说明掺杂比例过多的锰对合成的高镍四元材料的电化学性能提升没有助益。
中图分类号:
杨伟, 王昱杰, 方凯斌, 邹汉波, 陈胜洲, 刘自力. Co-Mn比例调控对LiNi0.8Co0.10-y Mn0.05+y Al0.05O2材料性能影响探究[J]. 化工学报, 2022, 73(12): 5615-5624.
Wei YANG, Yujie WANG, Kaibin FANG, Hanbo ZOU, Shengzhou CHEN, Zili LIU. Influence of cobalt-manganese ratio adjustment on the properties of LiNi0.8Co0.10-y Mn0.05+y Al0.05O2 materials[J]. CIESC Journal, 2022, 73(12): 5615-5624.

图1 LiNi0.8Co0.10–y Mn0.05+y Al0.05O2(y=0.01,0.02,0.03,0.04)样品的X射线衍射全谱图(a), (003)峰区域扩展谱图(b),(101)、(006)/(012)峰区域扩展谱图(c)和(018)/(110)峰区域扩展谱图(d)
Fig.1 Full XRD patterns (a), expanded views of (003) peaks (b), expanded views of (101), (006)/(012) peaks (c) and expanded views of (018)/(110) peaks (d) of LiNi0.8Co0.10-y Mn0.05+y Al0.05O2 (y=0.01,0.02,0.03,0.04)
| 样品 | a/Å | c/Å | c/a | v/Å3 | I(003)/I(104) |
|---|---|---|---|---|---|
| LiNi0.8Co0.09Mn0.06Al0.05O2 | 2.8717 | 14.1813 | 4.9383 | 101.28 | 1.5408 |
| LiNi0.8Co0.08Mn0.07Al0.05O2 | 2.8748 | 14.1944 | 4.9376 | 101.59 | 1.5674 |
| LiNi0.8Co0.07Mn0.08Al0.05O2 | 2.8743 | 14.1844 | 4.9349 | 101.49 | 1.4205 |
| LiNi0.8Co0.06Mn0.09Al0.05O2 | 2.8735 | 14.1910 | 4.9385 | 101.48 | 1.3298 |
表1 LiNi0.8Co0.10–y Mn0.05+y Al0.05O2(y=0.01,0.02,0.03,0.04)样品的晶胞参数
Table 1 Cell parameter of LiNi0.8Co0.10–y Mn0.05+y Al0.05O2 (y=0.01,0.02,0.03,0.04) samples
| 样品 | a/Å | c/Å | c/a | v/Å3 | I(003)/I(104) |
|---|---|---|---|---|---|
| LiNi0.8Co0.09Mn0.06Al0.05O2 | 2.8717 | 14.1813 | 4.9383 | 101.28 | 1.5408 |
| LiNi0.8Co0.08Mn0.07Al0.05O2 | 2.8748 | 14.1944 | 4.9376 | 101.59 | 1.5674 |
| LiNi0.8Co0.07Mn0.08Al0.05O2 | 2.8743 | 14.1844 | 4.9349 | 101.49 | 1.4205 |
| LiNi0.8Co0.06Mn0.09Al0.05O2 | 2.8735 | 14.1910 | 4.9385 | 101.48 | 1.3298 |

图2 LiNi0.8Co0.10–y Mn0.05+y Al0.05O2(y=0.01,0.02,0.03,0.04)正极材料的SEM图
Fig.2 SEM images of LiNi0.8Co0.10–y Mn0.05+y Al0.05O2 (y=0.01,0.02,0.03,0.04) cathode materials
| 样品 | 含量/% (mol) | |||
|---|---|---|---|---|
| Ni(理论含量/实际含量) | Co(理论含量/实际含量) | Mn(理论含量/实际含量) | Al(理论含量/实际含量) | |
| LiNi0.8Co0.09Mn0.06Al0.05O2 | 0.80/0.8028 | 0.09/0.0851 | 0.06/0.0572 | 0.05/0.0549 |
| LiNi0.8Co0.08Mn0.07Al0.05O2 | 0.80/0.8074 | 0.08/0.0833 | 0.07/0.0726 | 0.05/0.0367 |
| LiNi0.8Co0.07Mn0.08Al0.05O2 | 0.80/0.7992 | 0.07/0.0728 | 0.08/0.0829 | 0.05/0.0451 |
| LiNi0.8Co0.06Mn0.09Al0.05O2 | 0.80/0.7927 | 0.06/0.0624 | 0.09/0.0909 | 0.05/0.054 |
表2 LiNi0.8Co0.10-y Mn0.05+y Al0.05O2(y=0.01,0.02,0.03,0.04)正极材料各金属的含量
Table 2 Content of the quaternary metal of as-prepared LiNi0.8Co0.10-y Mn0.05+y Al0.05O2 (y=0.01,0.02,0.03,0.04) samples
| 样品 | 含量/% (mol) | |||
|---|---|---|---|---|
| Ni(理论含量/实际含量) | Co(理论含量/实际含量) | Mn(理论含量/实际含量) | Al(理论含量/实际含量) | |
| LiNi0.8Co0.09Mn0.06Al0.05O2 | 0.80/0.8028 | 0.09/0.0851 | 0.06/0.0572 | 0.05/0.0549 |
| LiNi0.8Co0.08Mn0.07Al0.05O2 | 0.80/0.8074 | 0.08/0.0833 | 0.07/0.0726 | 0.05/0.0367 |
| LiNi0.8Co0.07Mn0.08Al0.05O2 | 0.80/0.7992 | 0.07/0.0728 | 0.08/0.0829 | 0.05/0.0451 |
| LiNi0.8Co0.06Mn0.09Al0.05O2 | 0.80/0.7927 | 0.06/0.0624 | 0.09/0.0909 | 0.05/0.054 |
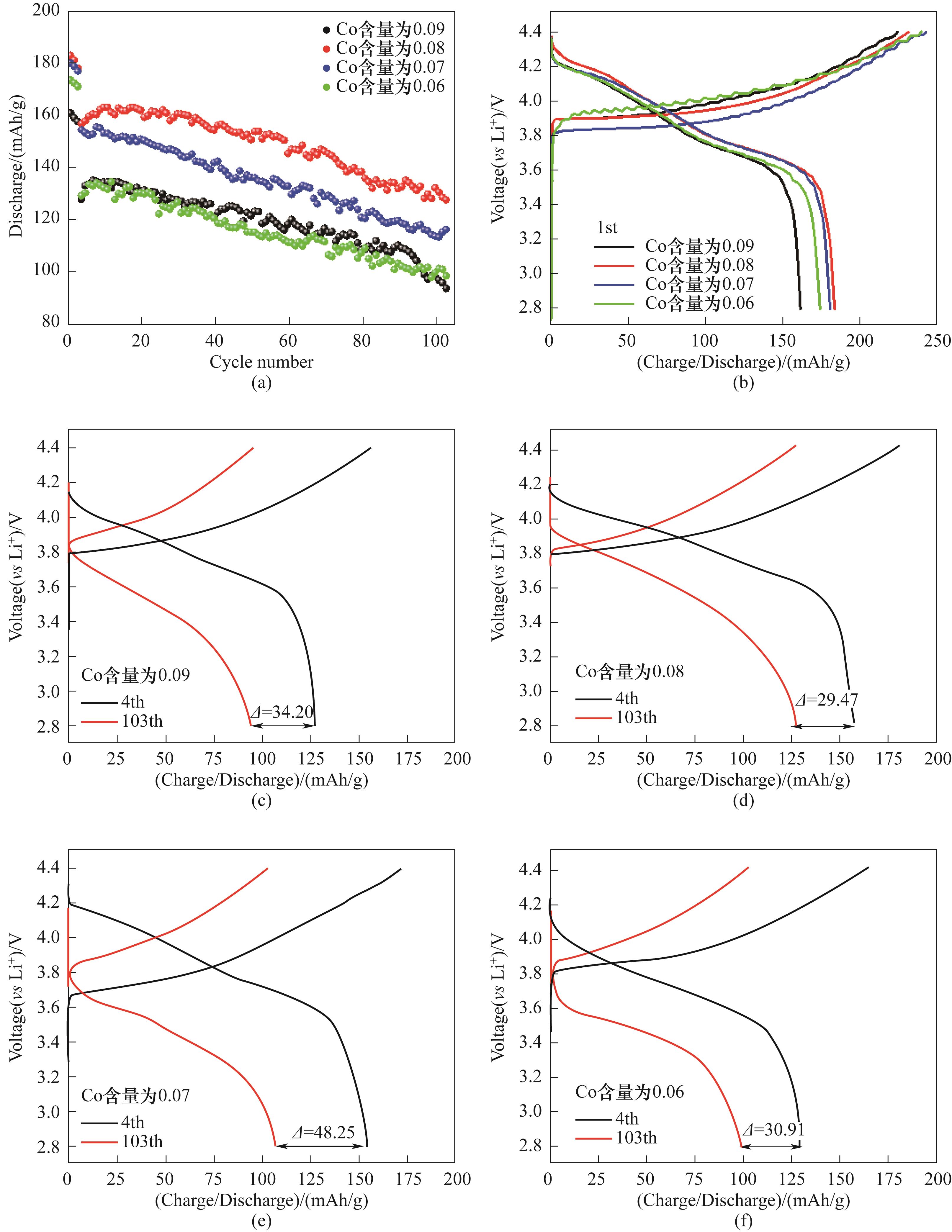
图3 LiNi0.8Co0.10-y Mn0.05+y Al0.05O2(y=0.01,0.02,0.03,0.04)在1C倍率下100圈循环性能(a)和首圈充放电平台(b); 1C倍率下第4圈和第103圈充放电平台:y=0.01(c),y=0.02(d),y=0.03(e),y=0.04(f)Fig.3 The cycle performance at 1C (a) and the initial charge-discharge curves (b) of LiNi0.8Co0.10-y Mn0.05+y Al0.05O2(y=0.01,0.02,0.03,0.04) cathode materials; the 4th and the 103th charge-discharge curves at 1C: y=0.01(c), y=0.02(d), y=0.03(e), y=0.04(f)
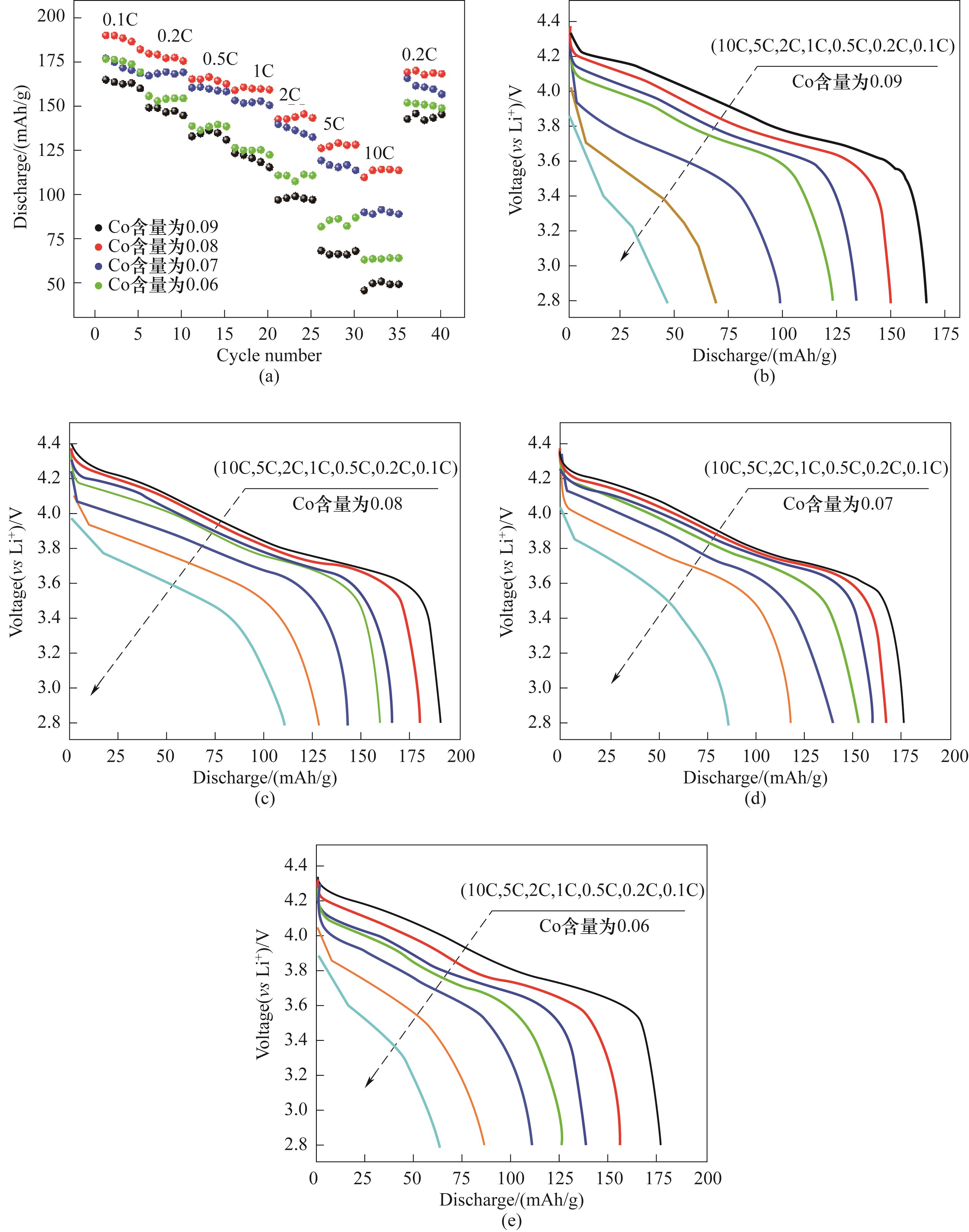
图4 LiNi0.8Co0.10-y Mn0.05+y Al0.05O2(y=0.01,0.02,0.03,0.04)正极材料的倍率性能(a)及对应的倍率放电电压平台曲线: y=0.01(b), y=0.02 (c), y=0.03 (d), y=0.04 (e)
Fig.4 The rate performances of LiNi0.8Co0.10-y Mn0.05+y Al0.05O2 (y=0.01,0.02,0.03,0.04) cathode materials (a) and the corresponding discharge curves: y=0.01 (b), y=0.02 (c), y=0.03 (d), y=0.04 (e)

图5 LiNi0.8Co0.10–y Mn0.05+y Al0.05O2(y=0.01,0.02,0.03,0.04)正极材料在1C倍率下第4圈与第103圈循环的dQ/dV图
Fig.5 dQ/dV plot for the 4th and 103th cycles of LiNi0.8Co0.10–y Mn0.05+y Al0.05O2 (y=0.01,0.02,0.03,0.04) at 1C rate
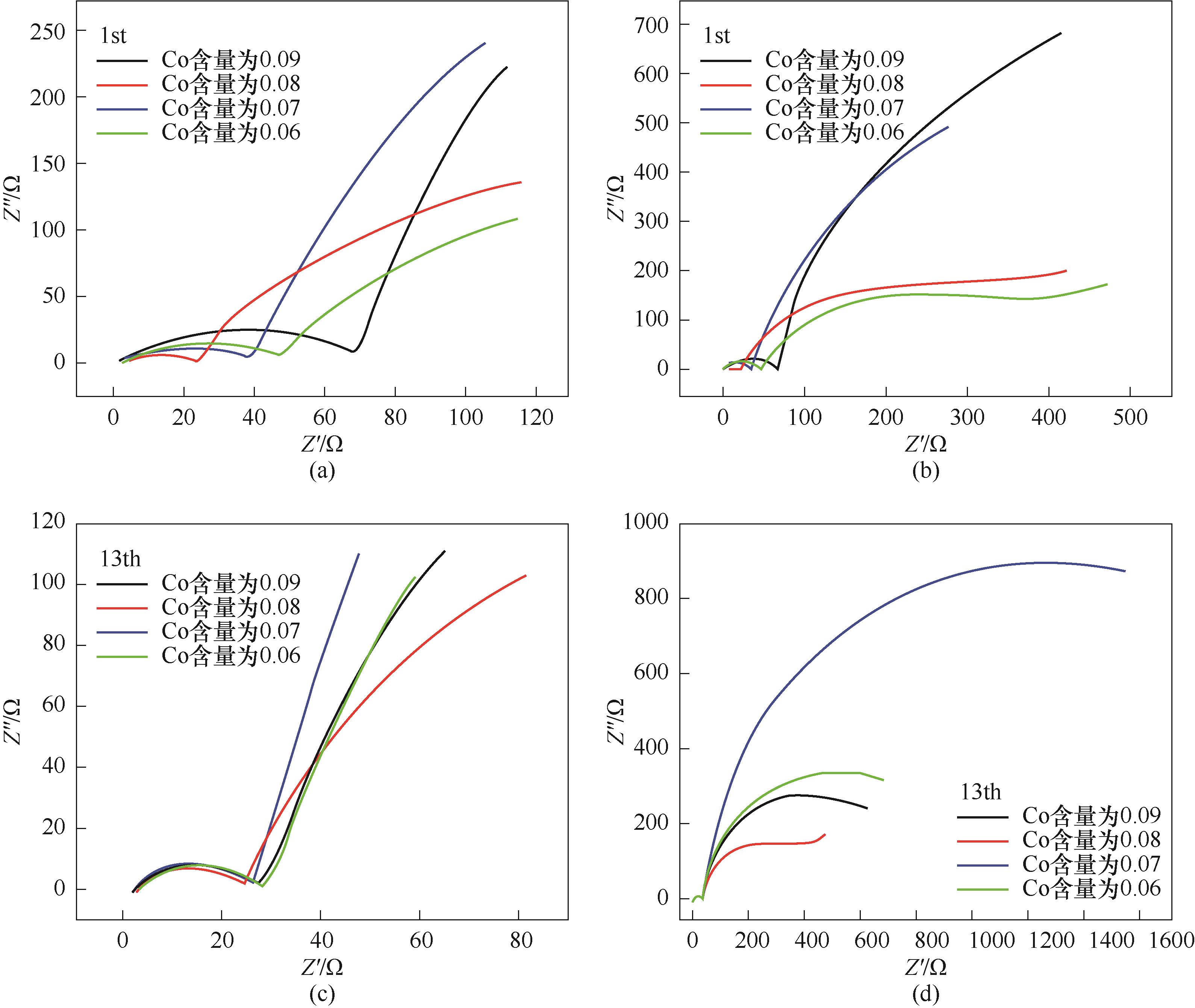
图6 LiNi0.8Co0.10–y Mn0.05+y Al0.05O2(y=0.01,0.02,0.03,0.04)正极材料在0.1C倍率下首圈循环的Nyquist曲线高频区域图(a),0.1C倍率下首圈循环的Nyquist曲线(b),1C倍率下第13圈循环的Nyquist曲线高频区域图(c),1C倍率下第13圈循环的Nyquist曲线(d)
Fig.6 The high frequency region of the Nyquist plots for the first cycle 0.1C rate (a), Nyquist plots for the first cycle at 0.1C rate (b), the high frequency region of the Nyquist plots for the 13th cycle at 1C rate (c) and Nyquist plots for the 13th cycle at 1C rate (d) of LiNi0.8Co0.10–y Mn0.05+y Al0.05O2 (y=0.01,0.02,0.03,0.04)
| 1 | Yang C K, Shao R W, Mi Y Y, et al. Stable interstitial layer to alleviate fatigue fracture of high nickel cathode for lithium-ion batteries[J]. Journal of Power Sources, 2018, 376: 200-206. |
| 2 | Kong F T, Liang C P, Wang L H, et al. Kinetic stability of bulk LiNiO2 and surface degradation by oxygen evolution in LiNiO2-based cathode materials[J]. Advanced Energy Materials, 2019, 9(2): 1802586. |
| 3 | Bai X T, Ban L Q, Zhuang W D. Research progress on coating and doping modification of nickel rich ternary cathode materials[J]. Journal of Inorganic Materials, 2020, 35(9): 972. |
| 4 | Zhu L, Liu Y, Wu W Y, et al. Surface fluorinated LiNi0.8Co0.15Al0.05O2 as a positive electrode material for lithium ion batteries[J]. Journal of Materials Chemistry A, 2015, 3(29): 15156-15162. |
| 5 | Yang W, Xie Q, Fang K B, et al. Inhibition of adverse phase transition at 4.2 V via increasing cobalt content on Ni-rich layered cathode materials[J]. ACS Applied Energy Materials, 2021, 4(7): 7365-7375. |
| 6 | Aishova A, Park G T, Yoon C S, et al. Cobalt-free high-capacity Ni-rich layered Li[Ni0.9Mn0.1]O2 cathode[J]. Advanced Energy Materials, 2020, 10(4): 1903179. |
| 7 | Muralidharan N, Essehli R, Hermann R P, et al. LiNi x Fe y Al z O2, a new cobalt-free layered cathode material for advanced Li-ion batteries[J]. Journal of Power Sources, 2020, 471: 228389. |
| 8 | Muto S, Tatsumi K, Kojima Y, et al. Effect of Mg-doping on the degradation of LiNiO2-based cathode materials by combined spectroscopic methods[J]. Journal of Power Sources, 2012, 205: 449-455. |
| 9 | Li W D, Lee S, Manthiram A. High-nickel NMA: a cobalt-free alternative to NMC and NCA cathodes for lithium-ion batteries[J]. Advanced Materials, 2020, 32(33): 2002718. |
| 10 | Fang K, Xie Q, Wang C, et al. Understanding the feasibility of manganese substitution for cobalt in the synthesis of nickel-rich and cobalt-free cathode materials[J]. ACS Applied Energy Materials, 2021, 4(7): 7190-7200. |
| 11 | Park S H, Park K S, Sun Y K, et al. Structural and electrochemical characterization of lithium excess and Al-doped nickel oxides synthesized by the sol-gel method[J]. Electrochimica Acta, 2001, 46(8): 1215-1222. |
| 12 | Guilmard M, Croguennec L, Denux D, et al. Thermal stability of lithium nickel oxide derivatives(1): Li x Ni1.02O2 and Li x Ni0.89Al0.16O2 (x = 0.50 and 0.30)[J]. Chemistry of Materials, 2003, 15(23): 4476-4483. |
| 13 | Bai G L, Wang C H, Luo J J, et al. High-capacity spherical LiNi0.82Co0.15Al0.03O2 cathode for lithium-ion batteries[J]. ChemistrySelect, 2019, 4(31): 9050-9054. |
| 14 | Vadivel S, Phattharasupakun N, Wutthiprom J, et al. High-performance Li-ion batteries using nickel-rich lithium nickel cobalt aluminium oxide-nanocarbon core-shell cathode: in operando X-ray diffraction[J]. ACS Applied Materials & Interfaces, 2019, 11(34): 30719-30727. |
| 15 | Lin S P, Fung K Z, Hon Y M, et al. Effect of Al addition on formation of layer-structured LiNiO2 [J]. Journal of Solid State Chemistry, 2002, 167(1): 97-106. |
| 16 | Jo M, Noh M, Oh P, et al. A new high power LiNi0.81Co0.1Al0.09O2 cathode material for lithium-ion batteries[J]. Advanced Energy Materials, 2014, 4(13): 1301583. |
| 17 | Xia Y, Zheng J M, Wang C M, et al. Designing principle for Ni-rich cathode materials with high energy density for practical applications[J]. Nano Energy, 2018, 49: 434-452. |
| 18 | Yang K. Influence of Al and Mn on the electrochemical performance of LiNi x Co y Mn1- x- y O2 as a cathode for lithium-ion batteries[J]. International Journal of Electrochemical Science, 2019, 14(9): 9007-9016. |
| 19 | Yudha C S, Muzayanha S U, Widiyandari H, et al. Synthesis of LiNi0.85Co0.14Al0.01O2 cathode material and its performance in an NCA/graphite full-battery[J]. Energies, 2019, 12(10): 1886. |
| 20 | Yan P F, Zheng J M, Gu M, et al. Intragranular cracking as a critical barrier for high-voltage usage of layer-structured cathode for lithium-ion batteries[J]. Nature Communications, 2017, 8: 14101. |
| 21 | Xu C L, Xiang W, Wu Z G, et al. Constructing a protective pillaring layer by incorporating gradient Mn4+ to stabilize the surface/interfacial structure of LiNi0.815Co0.15Al0.035O2 cathode[J]. ACS Applied Materials & Interfaces, 2018, 10(33): 27821-27830. |
| 22 | Yang W, Qiu X L, Wang C Y, et al. Controllable morphology tailoring with solvothermal method toward LiMnPO4/C cathode materials for improved performance and favorable thermostability[J]. Acta Metallurgica Sinica, 2022, 35(5): 790-800. |
| 23 | Cao C H, Zhang J, Xie X H, et al. Composition, structure, and performance of Ni-based cathodes in lithium ion batteries[J]. Ionics, 2017, 23(6): 1337-1356. |
| 24 | Liu Q, Du K, Guo H W, et al. Structural and electrochemical properties of Co-Mn-Mg multi-doped nickel based cathode materials LiNi0.9Co0.1- x [Mn1/2Mg1/2] x O2 for secondary lithium ion batteries[J]. Electrochimica Acta, 2013, 90: 350-357. |
| 25 | 叶乃清, 刘长久, 沈上越. 锂离子电池正极材料LiNiO2存在的问题与解决办法[J]. 无机材料学报, 2004, 19(6): 1217-1224. |
| Ye N Q, Liu C J, Shen S Y. Drawbacks and improve ways of LiNiO2 as a cathode material for lithium ion batteries[J]. Journal of Inorganic Materials, 2004, 19(6): 1217-1224. | |
| 26 | Myung S T, Maglia F, Park K J, et al. Nickel-rich layered cathode materials for automotive lithium-ion batteries: achievements and perspectives[J]. ACS Energy Letters, 2017, 2(1): 196-223. |
| 27 | Liu L L, Wang S L, Zhang Z Y, et al. Fluoroethylene carbonate as an electrolyte additive for improving interfacial stability of high-voltage LiNi0.6Co0.2Mn0.2O2 cathode[J]. Ionics, 2019, 25(3): 1035-1043. |
| 28 | Janssen P, Kasnatscheew J, Streipert B, et al. Fluorinated electrolyte compound as a bi-functional interphase additive for both anodes and cathodes in lithium-ion batteries[J]. Journal of the Electrochemical Society, 2018, 165(14): A3525-A3530. |
| 29 | Liu Y D, Xie J. Failure study of commercial LiFePO4 cells in overcharge conditions using electrochemical impedance spectroscopy[J]. Journal of the Electrochemical Society, 2015, 162(10): A2208-A2217. |
| 30 | Huang B, Li X H, Wang Z X, et al. Synthesis of Mg-doped LiNi0.8Co0.15Al0.05O2 oxide and its electrochemical behavior in high-voltage lithium-ion batteries[J]. Ceramics International, 2014, 40(8): 13223-13230. |
| 31 | Zhao D N, Wang P, Cui X L, et al. Robust and sulfur-containing ingredient surface film to improve the electrochemical performance of LiDFOB-based high-voltage electrolyte[J]. Electrochimica Acta, 2018, 260: 536-548. |
| 32 | Yang J, Xia Y Y. Suppressing the phase transition of the layered Ni-rich oxide cathode during high-voltage cycling by introducing low-content Li2MnO3 [J]. ACS Applied Materials & Interfaces, 2016, 8(2): 1297-1308. |
| 33 | Weber R, Louli A J, Plucknett K P, et al. Resistance growth in lithium-ion pouch cells with LiNi0.80Co0.15Al0.05O2 positive electrodes and proposed mechanism for voltage dependent charge-transfer resistance[J]. Journal of the Electrochemical Society, 2019, 166(10): A1779-A1784. |
| [1] | 王琪, 张斌, 张晓昕, 武虎建, 战海涛, 王涛. 氯铝酸-三乙胺离子液体/P2O5催化合成伊索克酸和2-乙基蒽醌[J]. 化工学报, 2023, 74(S1): 245-249. |
| [2] | 刘远超, 关斌, 钟建斌, 徐一帆, 蒋旭浩, 李耑. 单层XSe2(X=Zr/Hf)的热电输运特性研究[J]. 化工学报, 2023, 74(9): 3968-3978. |
| [3] | 程业品, 胡达清, 徐奕莎, 刘华彦, 卢晗锋, 崔国凯. 离子液体基低共熔溶剂在转化CO2中的应用[J]. 化工学报, 2023, 74(9): 3640-3653. |
| [4] | 康飞, 吕伟光, 巨锋, 孙峙. 废锂离子电池放电路径与评价研究[J]. 化工学报, 2023, 74(9): 3903-3911. |
| [5] | 胡亚丽, 胡军勇, 马素霞, 孙禹坤, 谭学诣, 黄佳欣, 杨奉源. 逆电渗析热机新型工质开发及电化学特性研究[J]. 化工学报, 2023, 74(8): 3513-3521. |
| [6] | 陈佳起, 赵万玉, 姚睿充, 侯道林, 董社英. 开心果壳基碳点的合成及其对Q235碳钢的缓蚀行为研究[J]. 化工学报, 2023, 74(8): 3446-3456. |
| [7] | 杨菲菲, 赵世熙, 周维, 倪中海. Sn掺杂的In2O3催化CO2选择性加氢制甲醇[J]. 化工学报, 2023, 74(8): 3366-3374. |
| [8] | 葛加丽, 管图祥, 邱新民, 吴健, 沈丽明, 暴宁钟. 垂直多孔碳包覆的FeF3正极的构筑及储锂性能研究[J]. 化工学报, 2023, 74(7): 3058-3067. |
| [9] | 屈园浩, 邓文义, 谢晓丹, 苏亚欣. 活性炭/石墨辅助污泥电渗脱水研究[J]. 化工学报, 2023, 74(7): 3038-3050. |
| [10] | 涂玉明, 邵高燕, 陈健杰, 刘凤, 田世超, 周智勇, 任钟旗. 钙基催化剂的设计合成及应用研究进展[J]. 化工学报, 2023, 74(7): 2717-2734. |
| [11] | 张琦钰, 高利军, 苏宇航, 马晓博, 王翊丞, 张亚婷, 胡超. 碳基催化材料在电化学还原二氧化碳中的研究进展[J]. 化工学报, 2023, 74(7): 2753-2772. |
| [12] | 张蒙蒙, 颜冬, 沈永峰, 李文翠. 电解液类型对双离子电池阴阳离子储存行为的影响[J]. 化工学报, 2023, 74(7): 3116-3126. |
| [13] | 邢美波, 张中天, 景栋梁, 张洪发. 磁调控水基碳纳米管协同多孔材料强化相变储/释能特性[J]. 化工学报, 2023, 74(7): 3093-3102. |
| [14] | 余娅洁, 李静茹, 周树锋, 李清彪, 詹国武. 基于天然生物模板构建纳米材料及集成催化剂研究进展[J]. 化工学报, 2023, 74(7): 2735-2752. |
| [15] | 王志龙, 杨烨, 赵真真, 田涛, 赵桐, 崔亚辉. 搅拌时间和混合顺序对锂离子电池正极浆料分散特性的影响[J]. 化工学报, 2023, 74(7): 3127-3138. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备 11010102001995号
京公网安备 11010102001995号