化工学报 ›› 2024, Vol. 75 ›› Issue (9): 3210-3220.DOI: 10.11949/0438-1157.20240323
唐宇昊1( ), 张迎迎1, 赵智伟1, 鲁梦悦1, 张飞飞1,2(
), 张迎迎1, 赵智伟1, 鲁梦悦1, 张飞飞1,2( ), 王小青1,2, 杨江峰1,2(
), 王小青1,2, 杨江峰1,2( )
)
收稿日期:2024-03-21
修回日期:2024-04-30
出版日期:2024-09-25
发布日期:2024-10-10
通讯作者:
张飞飞,杨江峰
作者简介:唐宇昊(2001—),男,学士,18727415796@163.com
基金资助:
Yuhao TANG1( ), Yingying ZHANG1, Zhiwei ZHAO1, Mengyue LU1, Feifei ZHANG1,2(
), Yingying ZHANG1, Zhiwei ZHAO1, Mengyue LU1, Feifei ZHANG1,2( ), Xiaoqing WANG1,2, Jiangfeng YANG1,2(
), Xiaoqing WANG1,2, Jiangfeng YANG1,2( )
)
Received:2024-03-21
Revised:2024-04-30
Online:2024-09-25
Published:2024-10-10
Contact:
Feifei ZHANG, Jiangfeng YANG
摘要:
低浓度煤层气的提质利用对缓解国内天然气不足的现状具有重要意义,然而煤层气中存在的氮气杂质限制了该类资源的进一步应用,进行低浓度煤层气中CH4/N2混合物的分离至关重要。制备了两种具有弱极性超微孔的金属有机框架材料Sc-CPM-66A和In-CPM-66A,研究材料从CH4/N2混合物中富集CH4的性能,利用PXRD、77 K N2吸附、TGA和FTIR光谱对材料的结构进行了表征。IAST 选择性计算表明,In-CPM-66A和Sc-CPM-66A的CH4/N2 选择性达到6.0。受益于材料表面存在的大量的甲基基团,两种材料对CH4的吸附热低于被报道的大部分材料,材料与甲烷分子之间弱的相互作用有利于吸附剂的脱附再生。穿透实验进一步表明,CPM-66A可以实现动态条件下CH4/N2混合物的分离,循环穿透实验显示该类材料具有良好的可重复性。
中图分类号:
唐宇昊, 张迎迎, 赵智伟, 鲁梦悦, 张飞飞, 王小青, 杨江峰. 弱极性超微孔Sc/In-CPM-66A用于CH4/N2吸附分离性能[J]. 化工学报, 2024, 75(9): 3210-3220.
Yuhao TANG, Yingying ZHANG, Zhiwei ZHAO, Mengyue LU, Feifei ZHANG, Xiaoqing WANG, Jiangfeng YANG. Ultra-microporous Sc/In-CPM-66A with low-polar pore surfaces for efficient separation of CH4/N2[J]. CIESC Journal, 2024, 75(9): 3210-3220.
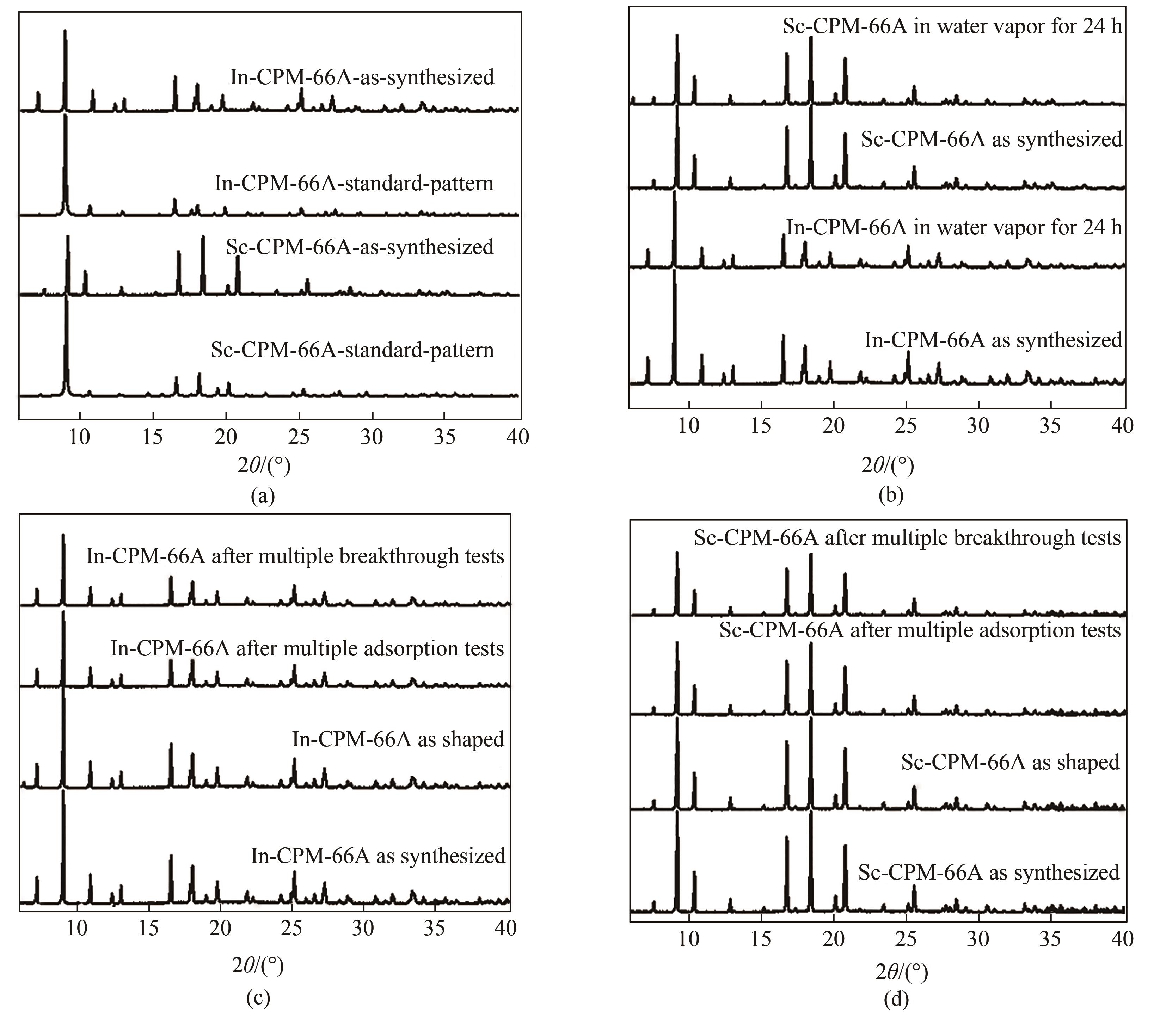
图2 (a)In-CPM-66A和 Sc-CPM-66A的PXRD谱图;(b)In-CPM-66A和 Sc-CPM-66A的水蒸气稳定性PXRD谱图;(c)In-CPM-66A在造粒/吸附/穿透后的PXRD谱图;(d)Sc-CPM-66A在造粒/吸附/穿透后的PXRD谱图
Fig.2 (a) PXRD patterns of In-CPM-66A and Sc-CPM-66A; (b) PXRD patterns of water vapor stability of In-CPM-66A and Sc-CPM-66A; (c) In-CPM-66A after as shaped/multiple adsorption tests/ multiple breakthrough tests; (d) Sc-CPM-66A after as shaped/multiple adsorption tests/multiple breakthrough tests
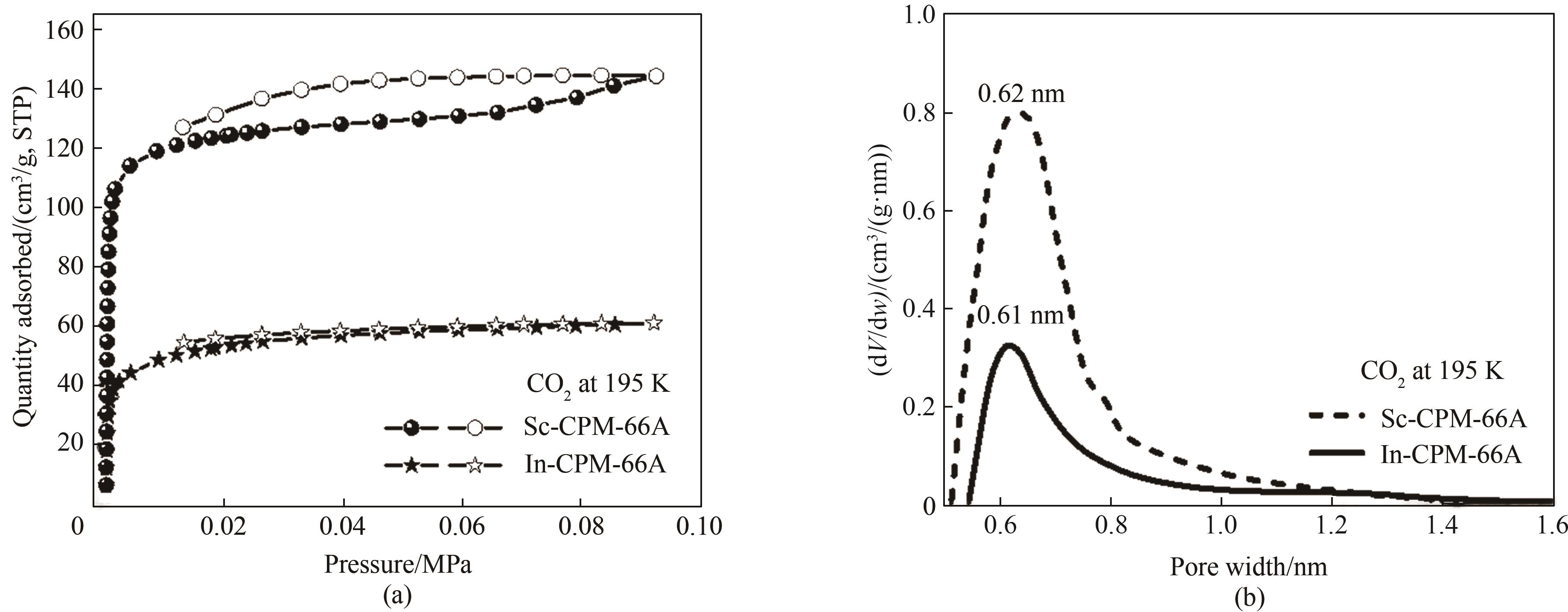
图4 (a)Sc-CPM-66A和In-CPM-66A在195 K下的CO2吸脱附等温线;(b)In-CPM-66A和Sc-CPM-66A的孔径分布
Fig.4 (a) CO2 absorption and desorption isotherms of In-CPM-66A and Sc-CPM-66A at 195 K; (b) Pore size distribution of In-CPM-66A and Sc-CPM-66A
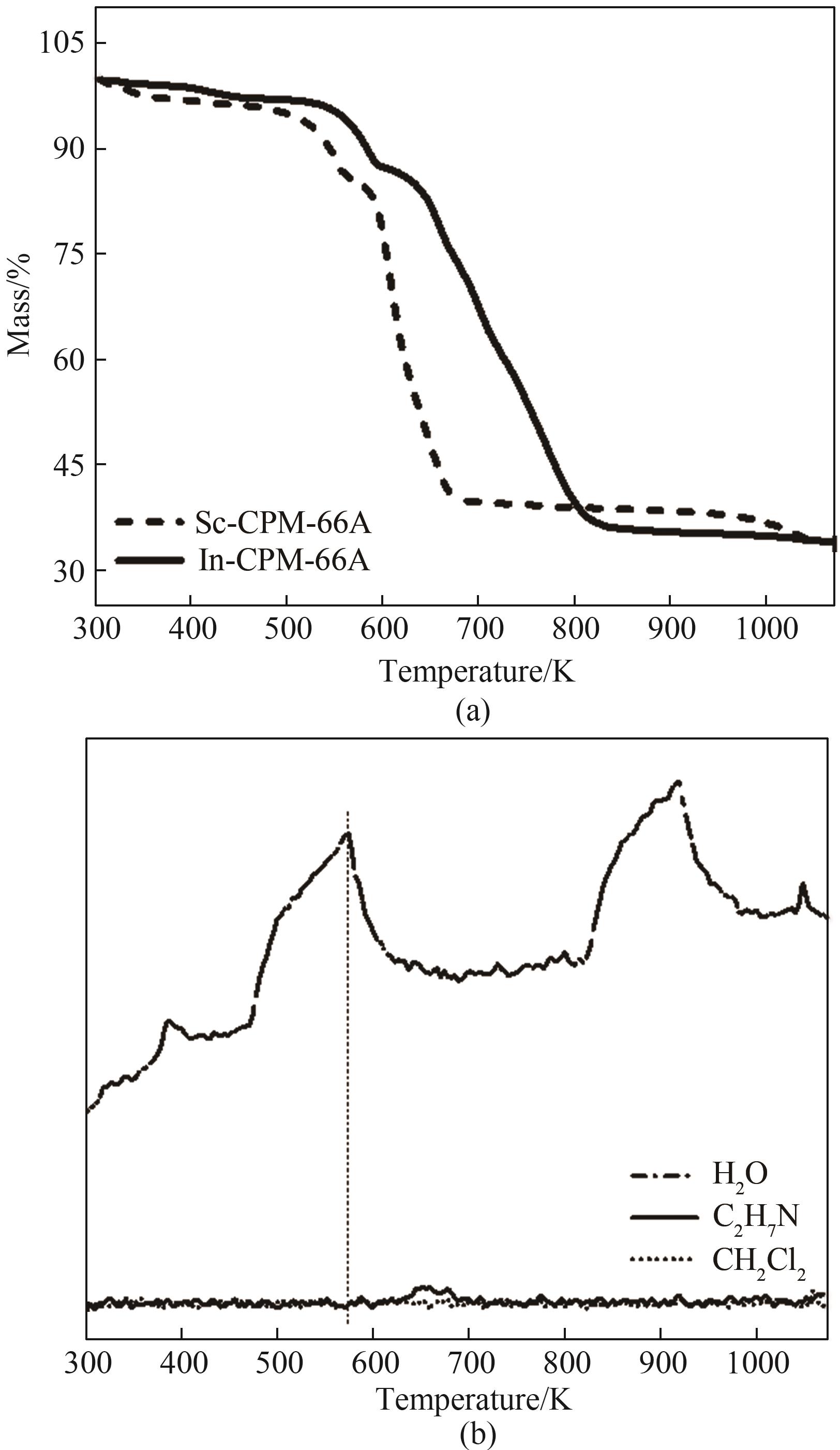
图5 (a) In-CPM-66A 和 Sc-CPM-66A的热重曲线;(b)Sc-CPM-66A的质谱曲线
Fig.5 (a) Thermogravimetric curves of In-CPM-66A and Sc-CPM-66A; (b) Mass spectrum curve of Sc-CPM-66A
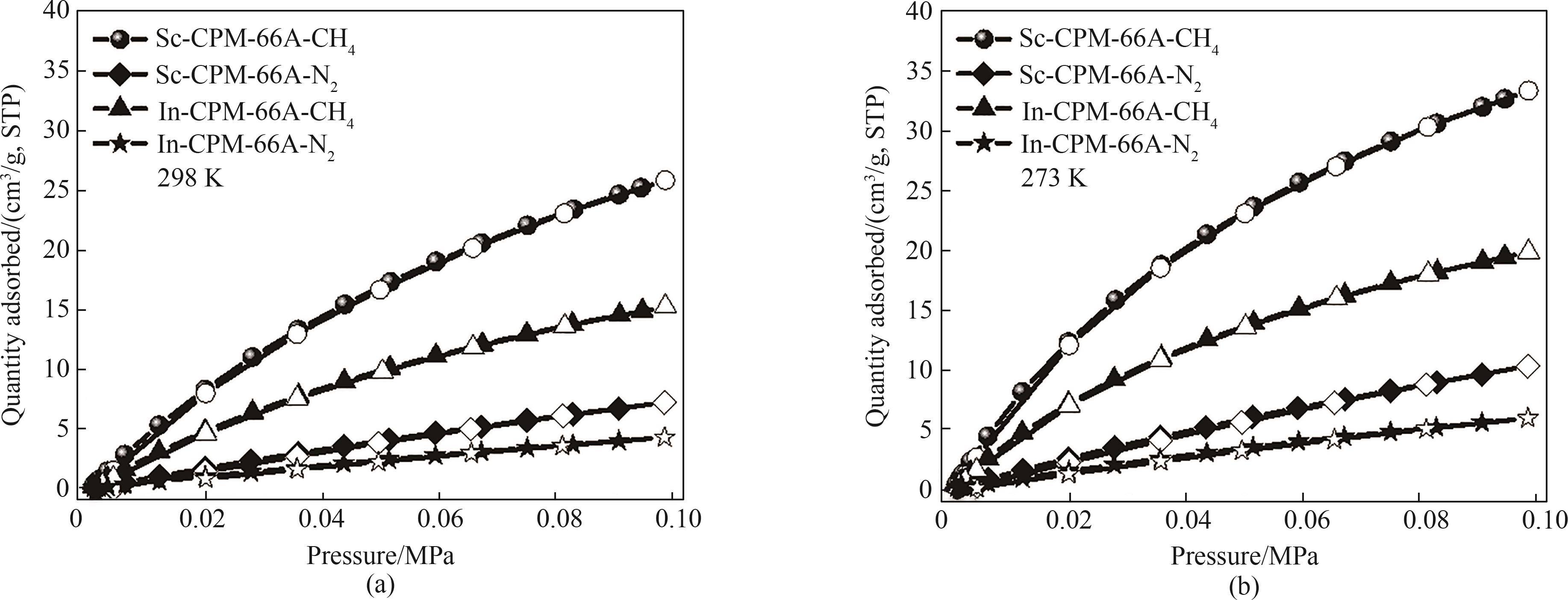
图7 (a)298 K下In-CPM-66A和Sc-CPM-66A的CH4/N2吸脱附等温线;(b) 273 K下In-CPM-66A和Sc-CPM-66A的CH4/N2吸脱附等温线
Fig.7 (a) CH4/N2 adsorption isotherms of In-CPM-66A and Sc-CPM-66A at 298 K;(b) CH4/N2 adsorption isotherms of In-CPM-66A and Sc-CPM-66A at 273 K
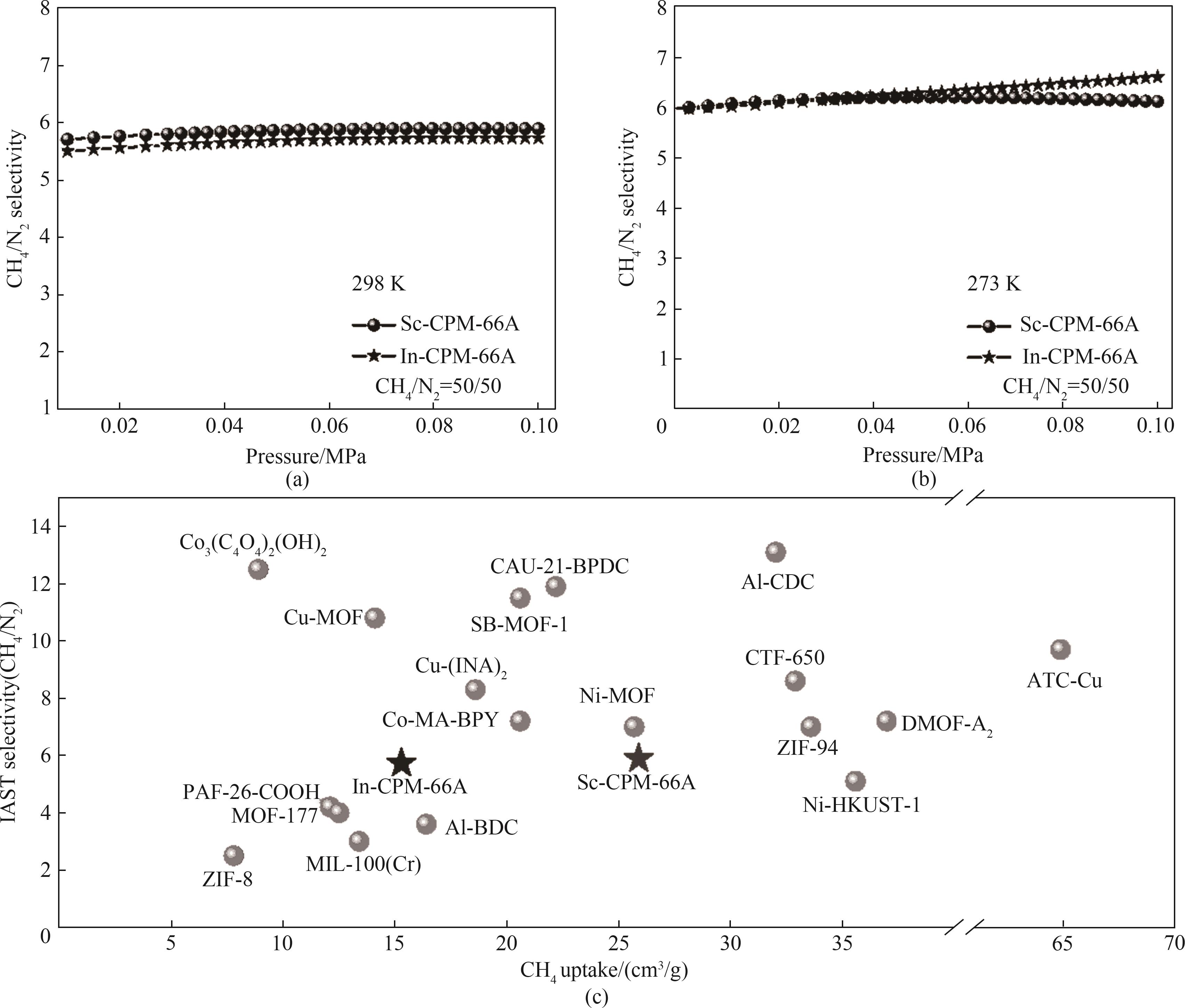
图8 (a)298 K下CH4/N2(体积比50/50)的IAST选择性;(b)273 K下CH4/N2(体积比50/50)的IAST选择性;(c)在298 K和0.1 MPa下一些典型MOF的CH4和N2的吸附容量和选择性对比
Fig.8 (a) IAST selectivity of CH4/N2 (50/50) at 298 K; (b) IAST selectivity of CH4/N2 (50/50) at 273 K; (c) Comparison of adsorption capacity and selectivity of CH4 and N2 in some typical MOF at 298 K and 0.1 MPa
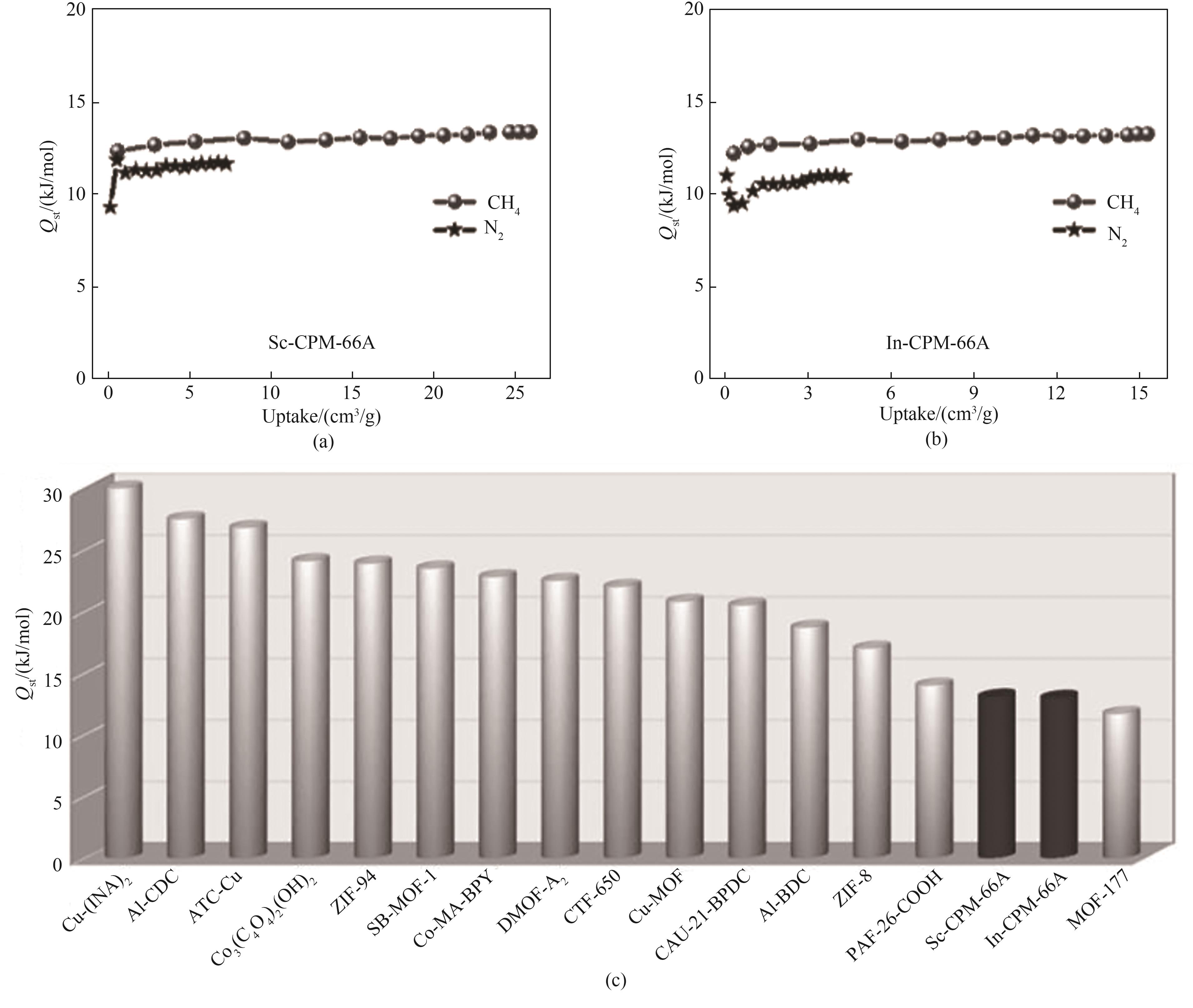
图9 (a)298 K下Sc-CPM-66A的CH4/N2吸附热;(b)298 K下In-CPM-66A的CH4/N2吸附热;(c)CH4选择性材料吸附热对比
Fig.9 (a)CH4/N2 adsorption heat of Sc-CPM-66A at 298 K; (b)CH4/N2 adsorption heat of In-CPM-66A at 298 K; (c)Comparison of CH4 adsorption heat of some previously reported CH4 selective materials
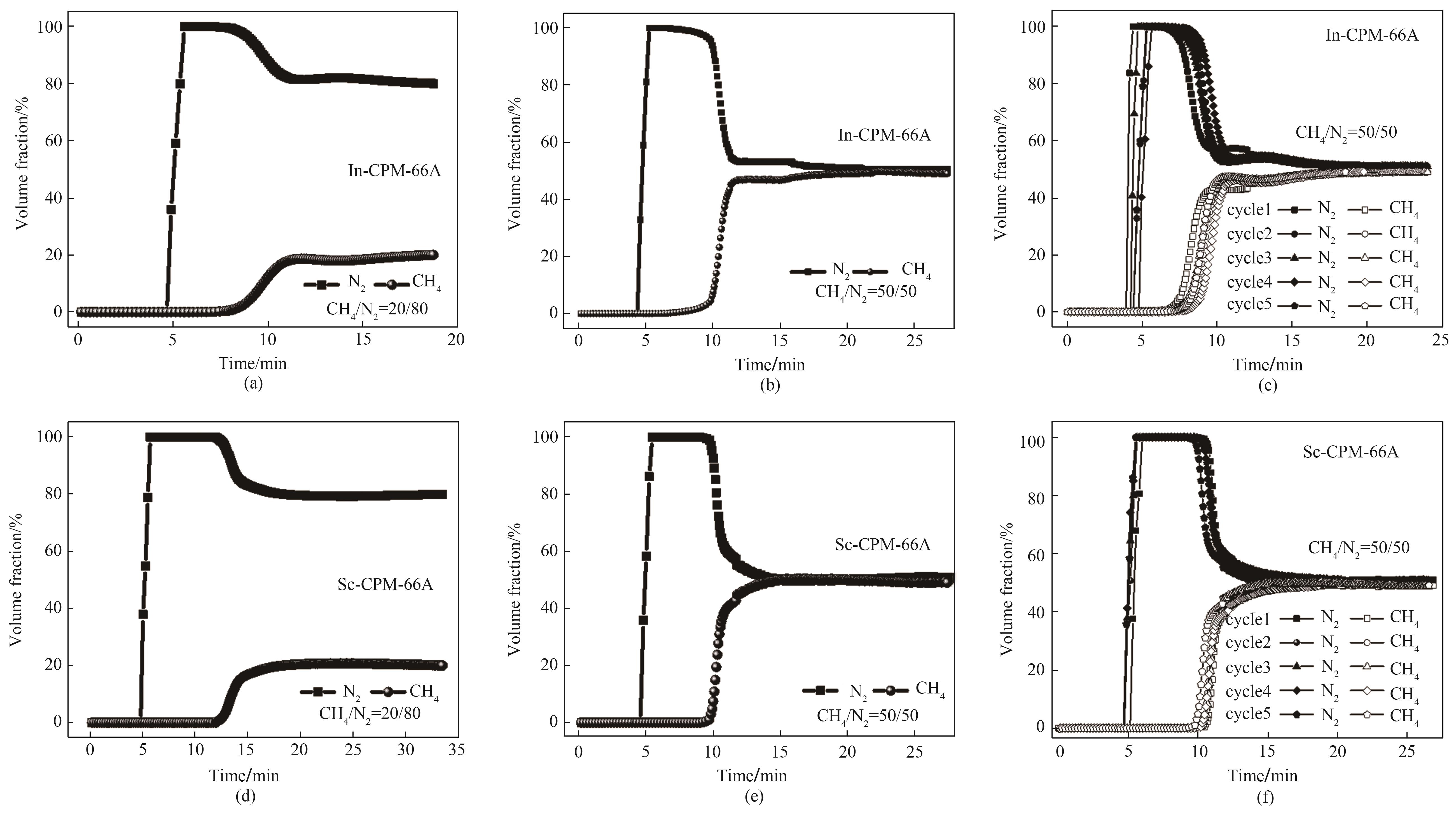
图10 (a)In-CPM-66A的CH4/N2(体积比20/80)穿透曲线;(b)In-CPM-66A的CH4/N2(体积比50/50)穿透曲线;(c)In-CPM-66A的CH4/N2(体积比50/50)穿透循环曲线;(d)Sc-CPM-66A的CH4/N2(体积比20/80)穿透曲线;(e)Sc-CPM-66A的CH4/N2(体积比50/50)穿透曲线;(f)Sc-CPM-66A的CH4/N2(体积比50/50)穿透循环曲线
Fig.10 (a) Breakthrough curve of In-CPM-66A for CH4/N2 (20/80); (b) Breakthrough curve of In-CPM-66A for CH4/N2 (50/50); (c) Breakthrough cycle curves of In-CPM-66A for CH4/N2 (50/50); (d) Breakthroughcurve of Sc-CPM-66A for CH4/N2 (20/80); (e) Breakthrough curve of Sc-CPM-66A forCH4/N2 (50/50); (f) Breakthrough cycle curves of Sc-CPM-66A for CH4/N2 (50/50)
| 1 | Lenton T M, Rockström J, Gaffney O, et al. Climate tipping points-too risky to bet against[J]. Nature, 2019, 575: 592-595. |
| 2 | He Y B, Zhou W, Qian G D, et al. Methane storage in metal-organic frameworks[J]. Chemical Society Reviews, 2014, 43(16): 5657-5678. |
| 3 | Saha D, Grappe H A, Chakraborty A, et al. Postextraction separation, on-board storage, and catalytic conversion of methane in natural gas: a review[J]. Chemical Reviews, 2016, 116(19): 11436-11499. |
| 4 | Sun Q, Wang M, Li Z, et al. Nitrogen removal from natural gas using solid boron: a first-principles computational study[J]. Fuel, 2013, 109: 575-581. |
| 5 | Sun Q, Sun C X, Du A J, et al. Charged-controlled separation of nitrogen from natural gas using boron nitride fullerene[J]. The Journal of Physical Chemistry C, 2014, 118(51): 30006-30012. |
| 6 | Yang J F, Bai H H, Shang H, et al. Experimental and simulation study on efficient CH4/N2 separation by pressure swing adsorption on silicalite-1 pellets[J]. Chemical Engineering Journal, 2020, 388: 124222. |
| 7 | Wu Y Q, Yuan D H, Zeng S, et al. Significant enhancement in CH4/N2 separation with amine-modified zeolite Y[J]. Fuel, 2021, 301: 121077. |
| 8 | Kim J, Maiti A, Lin L C, et al. New materials for methane capture from dilute and medium-concentration sources[J]. Nature Communications, 2013, 4: 1694. |
| 9 | Liu J Q, Shang H, Yang J F, et al. Novel zeolite/carbon monolith adsorbents for efficient CH4/N2 separation[J]. Chemical Engineering Journal, 2021, 426: 130163. |
| 10 | Kivi C E, Gelfand B S, Dureckova H, et al. 3D porous metal-organic framework for selective adsorption of methane over dinitrogen under ambient pressure[J]. Chemical Communications, 2018, 54(100): 14104-14107. |
| 11 | Bao Z B, Yu L, Ren Q L, et al. Adsorption of CO2 and CH4 on a magnesium-based metal organic framework[J]. Journal of Colloid and Interface Science, 2011, 353(2): 549-556. |
| 12 | Soroodan Miandoab E, Kentish S E, Scholes C A. Non-ideal modelling of polymeric hollow-fibre membrane systems: pre-combustion CO2 capture case study[J]. Journal of Membrane Science, 2020, 595: 117470. |
| 13 | Liang Z J, Marshall M, Chaffee A L. CO2 adsorption-based separation by metal organic framework (Cu-BTC) versus zeolite (13X)[J]. Energy & Fuels, 2009, 23(5): 2785-2789. |
| 14 | Chen R D, Li J Q, Zhou F R, et al. Zr-based metal-organic framework with wall-shared dual ultramicroporous channels for effective CH4/N2 separation[J]. Industrial & Engineering Chemistry Research, 2023, 62(33): 13144-13152. |
| 15 | Wu X F, Yuan B, Bao Z B, et al. Adsorption of carbon dioxide, methane and nitrogen on an ultramicroporous copper metal-organic framework[J]. Journal of Colloid and Interface Science, 2014, 430: 78-84. |
| 16 | Yu J M, Xie L H, Li J R, et al. CO2 capture and separations using MOF: computational and experimental studies[J]. Chemical Reviews, 2017, 117(14): 9674-9754. |
| 17 | Wu Y Q, Yuan D H, He D W, et al. Decorated traditional zeolites with subunits of metal-organic frameworks for CH4/N2 separation[J]. Angewandte Chemie International Edition, 2019, 58(30): 10241-10244. |
| 18 | Lin R B, Wu H, Li L B, et al. Boosting ethane/ethylene separation within isoreticular ultramicroporous metal-organic frameworks[J]. Journal of the American Chemical Society, 2018, 140(40): 12940-12946. |
| 19 | Yao K X, Chen Y L, Lu Y, et al. Ultramicroporous carbon with extremely narrow pore distribution and very high nitrogen doping for efficient methane mixture gases upgrading[J]. Carbon, 2017, 122: 258-265. |
| 20 | Nguyen P T K, Nguyen H T D, Pham H Q, et al. Synthesis and selective CO2 capture properties of a series of hexatopic linker-based metal-organic frameworks[J]. Inorganic Chemistry, 2015, 54(20): 10065-10072. |
| 21 | Li L Y, Yang L F, Wang J W, et al. Highly efficient separation of methane from nitrogen on a squarate-based metal-organic framework[J]. Chemical Engineers Journal, 2018, 64(10): 3681-3689. |
| 22 | Li J R, Sculley J, Zhou H C. Metal-organic frameworks for separations[J]. Chemical Reviews, 2012, 112(2): 869-932. |
| 23 | Cui X L, Chen K J, Xing H B, et al. Pore chemistry and size control in hybrid porous materials for acetylene capture from ethylene[J]. Science, 2016, 353(6295): 141-144. |
| 24 | Chen B L, Liang C D, Yang J, et al. A microporous metal-organic framework for gas-chromatographic separation of alkanes[J]. Angewandte Chemie International Edition, 2006, 45(9): 1390-1393. |
| 25 | Zhang F F, Li K J, Chen J, et al. Efficient N2/CH4 separation in a stable metal-organic framework with high density of open Cr sites[J]. Separation and Purification Technology, 2022, 281: 119951. |
| 26 | Pillai R S, Yoon J W, Lee S J, et al. N2 capture performances of the hybrid porous MIL-101(Cr): from prediction toward experimental testing[J]. The Journal of Physical Chemistry C, 2017, 121(40): 22130-22138. |
| 27 | Jaramillo D E, Reed D A, Jiang H Z H, et al. Selective nitrogen adsorption via backbonding in a metal-organic framework with exposed vanadium sites[J]. Nature Materials, 2020, 19(5): 517-521. |
| 28 | Zhang F F, Shang H, Wang L, et al. Construction of a porous metal-organic framework with a high density of open Cr sites for record N2/O2 separation[J]. Advanced Materials, 2021, 33(37): e2100866. |
| 29 | Chen Y, Wang Y, Wang Y, et al. Improving CH4 uptake and CH4/N2 separation in pillar-layered metal-organic frameworks using a regulating strategy of interlayer channels[J]. Chemical Engineers Journal, 2022, 68(11): e17819. |
| 30 | Wang S M, Shivanna M, Yang Q Y. Nickel-based metal-organic frameworks for coal-bed methane purification with record CH4/N2 selectivity[J]. Angewandte Chemie International Edition, 2022, 61(15): e202201017. |
| 31 | Oschatz M, Antonietti M. A search for selectivity to enable CO2 capture with porous adsorbents[J]. Energy & Environmental Science, 2018, 11(1): 57-70. |
| 32 | Chang M, Wang F, Wei Y, et al. Separation of CH4/N2 by an ultra-stable metal-organic framework with the highest breakthrough selectivity[J]. Chemical Engineers Journal, 2022, 68(9): e17794. |
| 33 | Chang M, Ren J H, Yang Q Y, et al. A robust calcium-based microporous metal-organic framework for efficient CH4/N2 separation[J]. Chemical Engineering Journal, 2021, 408: 127294. |
| 34 | Ren X Y, Sun T J, Hu J L, et al. Highly enhanced selectivity for the separation of CH4 over N2 on two ultra-microporous frameworks with multiple coordination modes[J]. Microporous and Mesoporous Materials, 2014, 186: 137-145. |
| 35 | Nandi S, Mansouri A, Dovgaliuk I, et al. A robust ultra-microporous cationic aluminum-based metal-organic framework with a flexible tetra-carboxylate linker[J]. Communications Chemistry, 2023, 6(1): 144. |
| 36 | Guo P T, Chen Y L, Chang M, et al. A stable Cu(Ⅰ)-based ultramicroporous NKMOF-8-Br with high CH4 uptake for efficient separation of CH4/N2 mixtures[J]. Journal of Chemical & Engineering Data, 2022, 67(7): 1654-1662. |
| 37 | Du S J, Wu Y, Wang X J, et al. Facile synthesis of ultramicroporous carbon adsorbents with ultra-high CH4 uptake by in situ ionic activation[J]. Chemical Engineers Journal, 2020, 66(7): e16231. |
| 38 | Li J R, Kuppler R J, Zhou H C. Selective gas adsorption and separation in metal-organic frameworks[J]. Chemical Society Reviews, 2009, 38(5): 1477-1504. |
| 39 | Lv D F, Wu Y, Chen J Y, et al. Improving CH4/N2 selectivity within isomeric Al-based MOF for the highly selective capture of coal-mine methane[J]. Chemical Engineers Journal, 2020, 66(9): e16287. |
| 40 | Niu Z, Cui X L, Pham T, et al. A metal-organic framework based methane nano-trap for the capture of coal-mine methane[J]. Angewandte Chemie International Edition, 2019, 58(30): 10138-10141. |
| 41 | Hu J L, Sun T J, Liu X W, et al. Rationally tuning the separation performances of [M3(HCOO)6] frameworks for CH4/N2 mixtures via metal substitution[J]. Microporous and Mesoporous Materials, 2016, 225: 456-464. |
| 42 | Chang M, Yan T A, Wei Y, et al. Enhancing CH4 capture from coalbed methane through tuning van der Waals affinity within isoreticular Al-based metal-organic frameworks[J]. ACS Applied Materials & Interfaces, 2022, 14(22): 25374-25384. |
| 43 | Yang H J, Peng F, Schier D E, et al. Selective crystallization of rare-earth ions into cationic metal-organic frameworks for rare-earth separation[J]. Angewandte Chemie International Edition, 2021, 60(20): 11148-11152. |
| [1] | 杨明军, 巩广军, 郑嘉男, 宋永臣. 泥质低渗水合物降压开采特性与模型研究[J]. 化工学报, 2024, 75(8): 2909-2916. |
| [2] | 吴哲明, 张碧云, 郑仁朝. 腈水解酶立体选择性改造及其合成布瓦西坦[J]. 化工学报, 2024, 75(7): 2633-2643. |
| [3] | 周文轩, 刘珍, 张福建, 张忠强. 高通量-高截留率时间维度膜法水处理机理研究[J]. 化工学报, 2024, 75(7): 2583-2593. |
| [4] | 王涛虹, 王超, 李政, 刘莹, 田歌, 常刚刚, 阳晓宇, 鲍宗必. 固载Cu(Ⅰ)的π络合MOF吸附剂用于乙烷/乙烯的选择性分离[J]. 化工学报, 2024, 75(7): 2565-2573. |
| [5] | 王岩, 周佳文, 孙培亮, 陈勇, 齐元红, 彭冲. 磁性聚氨基噻唑吸附剂脱除水体Hg2+性能[J]. 化工学报, 2024, 75(6): 2283-2298. |
| [6] | 冀钟, 赵彦玲, 陈雨濛, 高林霞, 王翼鹏, 刘欢. ZSM-5分子筛对典型涂装VOCs的吸附性能及机理研究[J]. 化工学报, 2024, 75(6): 2332-2343. |
| [7] | 张子佳, 仇昕月, 孙翔, 罗志斌, 罗海中, 贺高红, 阮雪华. 聚酰亚胺膜材料分子结构设计强化CO2渗透性研究进展[J]. 化工学报, 2024, 75(4): 1137-1152. |
| [8] | 李添翼, 武玉泰, 王永胜, 顾佳锐, 宋沂恒, 杨丰铖, 郝广平. 轻同位素分离纯化与催化标记研究进展[J]. 化工学报, 2024, 75(4): 1284-1301. |
| [9] | 张凯博, 沈佳新, 李玉霞, 谈朋, 刘晓勤, 孙林兵. Y沸石中Cu(Ⅰ)的可控构筑及其乙烯/乙烷吸附分离性能研究[J]. 化工学报, 2024, 75(4): 1607-1615. |
| [10] | 孟园, 倪善, 刘亚锋, 王文杰, 赵越, 朱育丹, 杨良嵘. 功能化多孔氮化碳材料对铀的吸附性能研究[J]. 化工学报, 2024, 75(4): 1616-1629. |
| [11] | 刘莹, 郑芳, 杨启炜, 张治国, 任其龙, 鲍宗必. 二甲苯异构体吸附分离研究进展[J]. 化工学报, 2024, 75(4): 1081-1095. |
| [12] | 张天永, 张晶怡, 姜爽, 李彬, 吕东军, 陈都民, 陈雪. 弱酸性蓝AS染料排放的废盐制碳基吸附剂及利用[J]. 化工学报, 2024, 75(3): 890-899. |
| [13] | 王宝凤, 王术高, 程芳琴. 固废基硫掺杂多孔炭材料制备及其对CO2吸附性能研究进展[J]. 化工学报, 2024, 75(2): 395-411. |
| [14] | 吴凡, 彭旭东, 江锦波, 孟祥铠, 梁杨杨. 分子动力学模拟预测天然气密度和黏度的可行性研究[J]. 化工学报, 2024, 75(2): 450-462. |
| [15] | 刘琦, 陈子康, 朴宇, 肖鹏, 葛亚粉, 巩雁军. 烃类催化裂解高选择性制低碳烯烃的分子筛催化剂[J]. 化工学报, 2024, 75(1): 120-137. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备 11010102001995号
京公网安备 11010102001995号