化工学报 ›› 2022, Vol. 73 ›› Issue (10): 4707-4721.DOI: 10.11949/0438-1157.20220715
郑喜1( ), 王涛1(
), 王涛1( ), 任永胜2, 赵珍珍1, 王雪琪1, 赵之平1
), 任永胜2, 赵珍珍1, 王雪琪1, 赵之平1
收稿日期:2022-04-16
修回日期:2022-05-26
出版日期:2022-10-05
发布日期:2022-11-02
通讯作者:
王涛
作者简介:郑喜(1999—),男,硕士研究生,zhengxihh@163.com
基金资助:
Xi ZHENG1( ), Tao WANG1(
), Tao WANG1( ), Yongsheng REN2, Zhenzhen ZHAO1, Xueqi WANG1, Zhiping ZHAO1
), Yongsheng REN2, Zhenzhen ZHAO1, Xueqi WANG1, Zhiping ZHAO1
Received:2022-04-16
Revised:2022-05-26
Online:2022-10-05
Published:2022-11-02
Contact:
Tao WANG
摘要:
以聚间苯二甲酰间苯二胺(PMIA)为制膜原料,氯化锂(LiCl)、聚乙二醇(PEG-400)和聚乙烯吡咯烷酮(PVP)为添加剂,通过非溶剂诱导相转化法制备了PMIA平板膜,系统考察了聚合物浓度、添加剂种类和含量对PMIA膜结构和性能的影响。结果表明,聚合物浓度和LiCl含量增加,铸膜液黏度增大,导致膜孔径减小,纯水通量降低。而PEG含量的增加,使得聚合物链呈现舒展状态,膜孔径增大,纯水通量升高,亲水性增强。随着PVP含量的增加,膜的纯水通量先升高后降低,膜的亲水性变差。当PMIA的质量分数为9%,LiCl的质量分数为2.8%,PVP的质量分数为1.2%时,膜的纯水通量高达1421.55 L·m-2·h-1·bar-1,对牛血清蛋白(BSA)的截留率为80%,展现出较高的渗透性,为制备高性能膜材料提供了新的思路。
中图分类号:
郑喜, 王涛, 任永胜, 赵珍珍, 王雪琪, 赵之平. 聚间苯二甲酰间苯二胺平板膜的制备及其性能研究[J]. 化工学报, 2022, 73(10): 4707-4721.
Xi ZHENG, Tao WANG, Yongsheng REN, Zhenzhen ZHAO, Xueqi WANG, Zhiping ZHAO. Preparation and properties research of poly(m-phenylene isophthalamide) flat-sheet membrane[J]. CIESC Journal, 2022, 73(10): 4707-4721.
| 膜 | PMIA/g | LiCl/g | PVP-K15/g | DMAc/g | 总计/g |
|---|---|---|---|---|---|
| A1 | 9 | 3.8 | 1.2 | 86 | 100 |
| A2 | 10 | 3.8 | 1.2 | 85 | 100 |
| A3 | 11 | 3.8 | 1.2 | 84 | 100 |
| A4 | 12 | 3.8 | 1.2 | 83 | 100 |
表1 不同聚合物浓度的铸膜液
Table 1 Casting solution of different polymer concentration
| 膜 | PMIA/g | LiCl/g | PVP-K15/g | DMAc/g | 总计/g |
|---|---|---|---|---|---|
| A1 | 9 | 3.8 | 1.2 | 86 | 100 |
| A2 | 10 | 3.8 | 1.2 | 85 | 100 |
| A3 | 11 | 3.8 | 1.2 | 84 | 100 |
| A4 | 12 | 3.8 | 1.2 | 83 | 100 |
| 膜 | 平均孔径/μm | 最可几孔径/μm | 孔隙率/% |
|---|---|---|---|
| A1 | 0.2375 | 0.2203 | 71.98±3.84 |
| A2 | 0.1145 | 0.1022 | 61.78±4.74 |
| A3 | 0.0947 | 0.0918 | 56.11±3.27 |
| A4 | 0.0379 | 0.0355 | 63.39±1.28 |
表2 不同聚合物浓度下膜的孔径和孔隙率数据
Table 2 Pore diameter and porosity data of membranes at different polymer concentrations
| 膜 | 平均孔径/μm | 最可几孔径/μm | 孔隙率/% |
|---|---|---|---|
| A1 | 0.2375 | 0.2203 | 71.98±3.84 |
| A2 | 0.1145 | 0.1022 | 61.78±4.74 |
| A3 | 0.0947 | 0.0918 | 56.11±3.27 |
| A4 | 0.0379 | 0.0355 | 63.39±1.28 |
| 膜 | LiCl/g | PMIA/g | PVP-K15/g | DMAc/g | 总计/g |
|---|---|---|---|---|---|
| B1 | 2.8 | 9 | 1.2 | 87 | 100 |
| B2 | 3.8 | 9 | 1.2 | 86 | 100 |
| B3 | 4.8 | 9 | 1.2 | 85 | 100 |
| B4 | 5.8 | 9 | 1.2 | 84 | 100 |
表3 不同无机添加剂浓度的铸膜液
Table 3 Casting solutions with different concentrations of inorganic additives
| 膜 | LiCl/g | PMIA/g | PVP-K15/g | DMAc/g | 总计/g |
|---|---|---|---|---|---|
| B1 | 2.8 | 9 | 1.2 | 87 | 100 |
| B2 | 3.8 | 9 | 1.2 | 86 | 100 |
| B3 | 4.8 | 9 | 1.2 | 85 | 100 |
| B4 | 5.8 | 9 | 1.2 | 84 | 100 |
| 膜 | 平均孔径/μm | 最可几孔径/μm | 孔隙率/% |
|---|---|---|---|
| B1 | 0.2247 | 0.2232 | 78.87±5.66 |
| B2 | 0.1768 | 0.1718 | 76.39±2.31 |
| B3 | 0.1113 | 0.1057 | 70.56±4.75 |
| B4 | 0.0913 | 0.0847 | 66.27±2.84 |
表4 不同无机添加剂浓度下膜的孔径和孔隙率数据
Table 4 Pore diameter and porosity data of membranes at different concentrations of inorganic additives
| 膜 | 平均孔径/μm | 最可几孔径/μm | 孔隙率/% |
|---|---|---|---|
| B1 | 0.2247 | 0.2232 | 78.87±5.66 |
| B2 | 0.1768 | 0.1718 | 76.39±2.31 |
| B3 | 0.1113 | 0.1057 | 70.56±4.75 |
| B4 | 0.0913 | 0.0847 | 66.27±2.84 |
| 膜 | PEG/g | PMIA/g | LiCl/g | PVP-K15/g | DMAc/g | 总计/g |
|---|---|---|---|---|---|---|
| C1 | 2 | 10 | 3.8 | 1.2 | 83 | 100 |
| C2 | 4 | 10 | 3.8 | 1.2 | 81 | 100 |
| C3 | 6 | 10 | 3.8 | 1.2 | 79 | 100 |
| C4 | 8 | 10 | 3.8 | 1.2 | 77 | 100 |
表5 不同小分子有机添加剂的铸膜液
Table 5 Casting solution of different organic additive with small molecule
| 膜 | PEG/g | PMIA/g | LiCl/g | PVP-K15/g | DMAc/g | 总计/g |
|---|---|---|---|---|---|---|
| C1 | 2 | 10 | 3.8 | 1.2 | 83 | 100 |
| C2 | 4 | 10 | 3.8 | 1.2 | 81 | 100 |
| C3 | 6 | 10 | 3.8 | 1.2 | 79 | 100 |
| C4 | 8 | 10 | 3.8 | 1.2 | 77 | 100 |
| 膜 | 平均孔径/μm | 最可几孔径/μm | 孔隙率/% |
|---|---|---|---|
| C1 | 0.0618 | 0.0483 | 69.58±0.68 |
| C2 | 0.0571 | 0.0537 | 80.09±1.72 |
| C3 | 0.0797 | 0.0783 | 79.67±0.80 |
| C4 | 0.1050 | 0.0774 | 76.11±3.35 |
表6 不同小分子有机添加剂浓度下膜的孔径和孔隙率数据
Table 6 Pore diameter and porosity data of membrane at different concentrations of organic additives with small molecule
| 膜 | 平均孔径/μm | 最可几孔径/μm | 孔隙率/% |
|---|---|---|---|
| C1 | 0.0618 | 0.0483 | 69.58±0.68 |
| C2 | 0.0571 | 0.0537 | 80.09±1.72 |
| C3 | 0.0797 | 0.0783 | 79.67±0.80 |
| C4 | 0.1050 | 0.0774 | 76.11±3.35 |
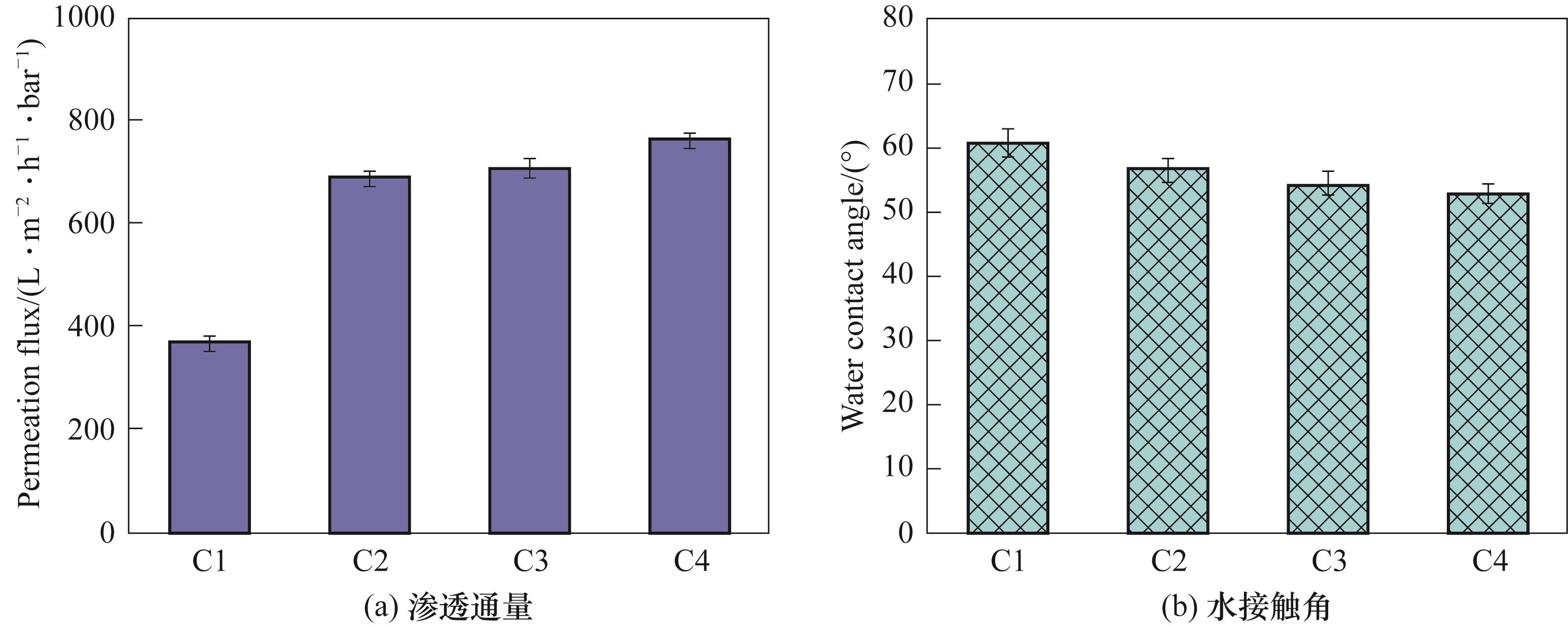
图13 不同小分子有机添加剂浓度下膜的渗透通量和水接触角
Fig.13 Permeation flux and water contact angle of membranes at different concentrations of organic additives with small molecule
| 膜 | PVP-K15/g | PMIA/g | LiCl/g | DMAc/g | 总计/g |
|---|---|---|---|---|---|
| D1 | 1.2 | 10 | 3.8 | 85 | 100 |
| D2 | 3.2 | 10 | 3.8 | 83 | 100 |
| D3 | 5.2 | 10 | 3.8 | 81 | 100 |
| D4 | 7.2 | 10 | 3.8 | 79 | 100 |
表7 不同大分子有机添加剂的铸膜液
Table 7 Casting solution of different organic additives with macromolecular
| 膜 | PVP-K15/g | PMIA/g | LiCl/g | DMAc/g | 总计/g |
|---|---|---|---|---|---|
| D1 | 1.2 | 10 | 3.8 | 85 | 100 |
| D2 | 3.2 | 10 | 3.8 | 83 | 100 |
| D3 | 5.2 | 10 | 3.8 | 81 | 100 |
| D4 | 7.2 | 10 | 3.8 | 79 | 100 |
| 膜 | 平均孔径/μm | 最可几孔径/μm | 孔隙率/% |
|---|---|---|---|
| D1 | 0.1250 | 0.1176 | 73.31±0.71 |
| D2 | 0.1522 | 0.1807 | 78.28±0.20 |
| D3 | 0.0613 | 0.0566 | 84.80±1.52 |
| D4 | 0.1354 | 0.0676 | 76.05±0.86 |
表8 不同大分子有机添加剂浓度下膜的孔径和孔隙率数据
Table 8 Pore diameter and porosity data of membranes at different concentrations of organic additives with macromolecule
| 膜 | 平均孔径/μm | 最可几孔径/μm | 孔隙率/% |
|---|---|---|---|
| D1 | 0.1250 | 0.1176 | 73.31±0.71 |
| D2 | 0.1522 | 0.1807 | 78.28±0.20 |
| D3 | 0.0613 | 0.0566 | 84.80±1.52 |
| D4 | 0.1354 | 0.0676 | 76.05±0.86 |
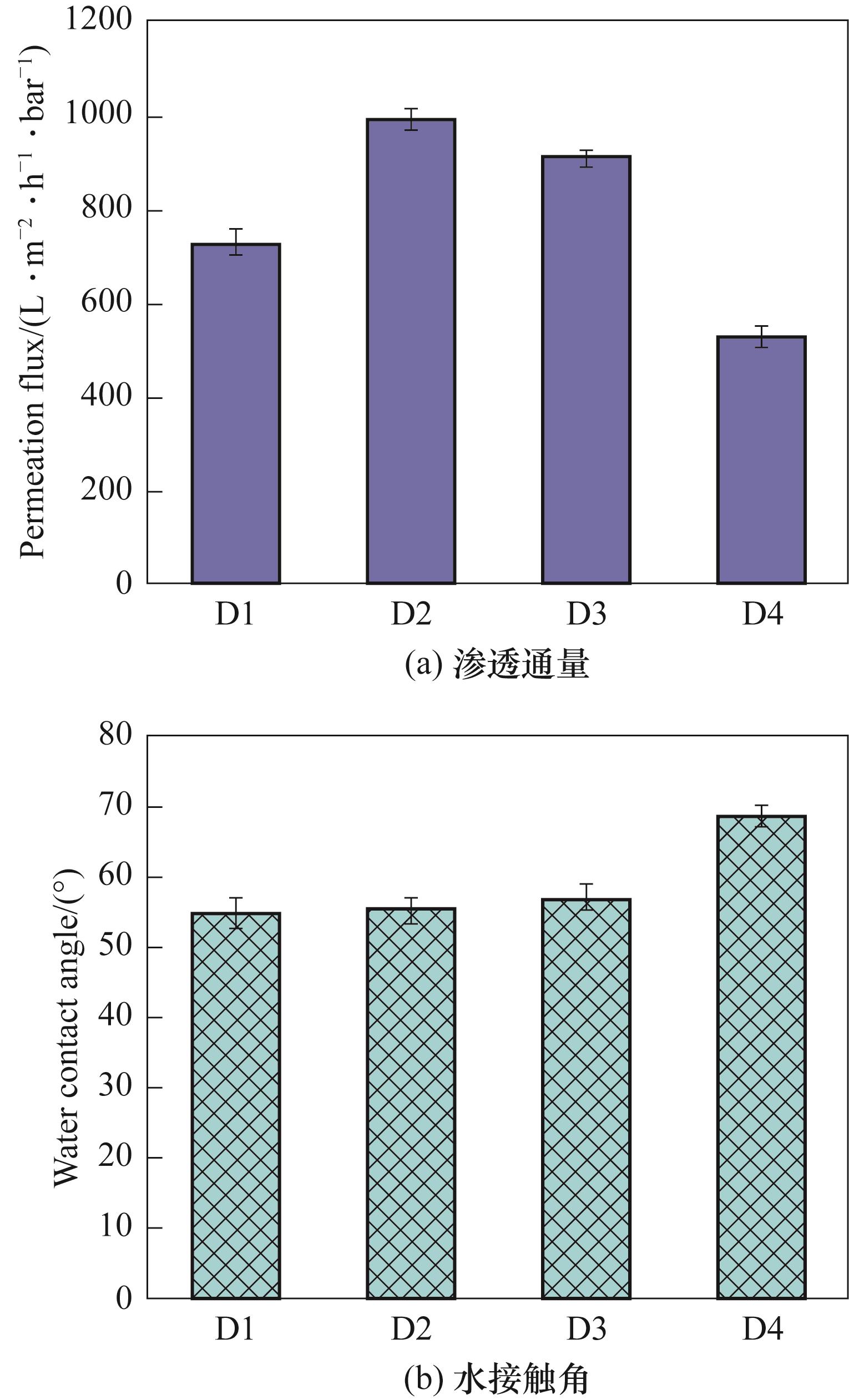
图16 不同大分子有机添加剂浓度下膜的渗透通量和水接触角
Fig.16 Permeation flux and water contact angle of membranes at different concentrations of organic additives with macromolecule
| 1 | He C S, Harden C P, Liu Y X. Comparison of water resources management between China and the United States[J]. Geography and Sustainability, 2020, 1(2): 98-108. |
| 2 | Cramer W, Guiot J, Fader M, et al. Climate change and interconnected risks to sustainable development in the Mediterranean[J]. Nature Climate Change, 2018, 8(11): 972-980. |
| 3 | Tan X, Rodrigue D. A review on porous polymeric membrane preparation(part Ⅰ): Production techniques with polysulfone and poly(vinylidene fluoride)[J]. Polymers, 2019, 11(7): 1160. |
| 4 | Peters T. Membrane technology for water treatment[J]. Chemical Engineering and Technology, 2010, 33(8): 1233-1240. |
| 5 | Ali A, Tufa R A, Macedonio F, et al. Membrane technology in renewable-energy-driven desalination[J]. Renewable and Sustainable Energy Reviews, 2018, 81: 1-21. |
| 6 | Wang W, Xu X, Zhang Z C, et al. Study on the improvement of PVDF flat ultrafiltration membrane with MWCNTs-OH as the additive and the influence of different MWCNTs-OH scales[J]. Colloid and Interface Science Communications, 2021, 43: 100433. |
| 7 | Lee A, Elam J W, Darling S B. Membrane materials for water purification: design, development, and application[J]. Environmental Science: Water Research and Technology, 2016, 2(1): 17-42. |
| 8 | Yang M, Zhao C, Zhang S, et al. Preparation of graphene oxide modified poly(m-phenylene isophthalamide) nanofiltration membrane with improved water flux and antifouling property[J]. Applied Surface Science, 2017, 394: 149-159. |
| 9 | Kakida H, Chatani Y, Tadokoro H. Crystal structure of poly(m-phenylene isophthalamide)[J]. Journal of Polymer Science: Polymer Physics Edition, 1976, 14(3): 427-435. |
| 10 | Wang T, He X P, Li Y, et al. Novel poly(piperazine-amide) (PA) nanofiltration membrane based poly(m-phenylene isophthalamide) (PMIA) hollow fiber substrate for treatment of dye solutions[J]. Chemical Engineering Journal, 2018, 351: 1013-1026. |
| 11 | Lin C E, Wang J, Zhou M Y, et al. Poly(m-phenylene isophthalamide) (PMIA): a potential polymer for breaking through the selectivity-permeability trade-off for ultrafiltration membranes[J]. Journal of Membrane Science, 2016, 518: 72-78. |
| 12 | Song Q Q, Feng Y, Wu W W, et al. Blending modification of PMIA with poly(vinyl pyrrolidone): towards high-performance material with enhanced mechanical property[J]. The Journal of the Textile Institute, 2021, 112(12): 2004-2012. |
| 13 | Balakrishnan J, Pramila J. Elimination of toxic metal ion by means of poly m-phenylene (isophthalamide) ultra filtration membranes[J]. Materials Today: Proceedings, 2020, 33: 2858-2862. |
| 14 | Zhang L Y, Shi Y X, Wang T, et al. Fabrication of novel anti-fouling poly(m-phenylene isophthalamide) ultrafiltration membrane modified with Pluronic F127 via coupling phase inversion and surface segregation[J]. Separation and Purification Technology, 2022, 282: 120106. |
| 15 | Paul M, Jons S D. Chemistry and fabrication of polymeric nanofiltration membranes: a review[J]. Polymer, 2016, 103: 417-456. |
| 16 | 姜钦亮, 张凯松. PMIA和氯化锂含量对PMIA平板超滤膜结构与性能的影响[J]. 膜科学与技术, 2019, 39(5): 94-100. |
| Jiang Q L, Zhang K S. Effects of PMIA and LiCl content on structure and performance of PMIA flat sheet ultrafiltration membrane[J]. Membrane Science and Technology, 2019, 39(5): 94-100. | |
| 17 | Prince J A, Bhuvana S, Boodhoo K V K, et al. Synthesis and characterization of PEG-Ag immobilized PES hollow fiber ultrafiltration membranes with long lasting antifouling properties[J]. Journal of Membrane Science, 2014, 454: 538-548. |
| 18 | Wang T, Zheng X, Wang Y J, et al. Fabrication and performance of novel poly(piperazine-amide) composite nanofiltration membranes based on various poly(m-phenylene isophthalamide) substrates[J]. Industrial and Engineering Chemistry Research, 2021, 60(49): 18106-18120. |
| 19 | Jiang B, Zhang N, Zhang L H, et al. Enhanced separation performance of PES ultrafiltration membranes by imidazole-based deep eutectic solvents as novel functional additives[J]. Journal of Membrane Science, 2018, 564: 247-258. |
| 20 | Li H B, Shi W Y, Zhang Y F, et al. Effects of additives on the morphology and performance of PPTA/PVDF in situ blend UF membrane[J]. Polymers, 2014, 6(6): 1846-1861. |
| 21 | Murakata T, Sato S, Ohgawara T, et al. Control of pore size distribution of silica gel through sol-gel process using inorganic salts and surfactants as additives[J]. Journal of Materials Science, 1992, 27(6): 1567-1574. |
| 22 | Ma Y X, Shi F M, Wang Z J, et al. Preparation and characterization of PSf/clay nanocomposite membranes with PEG 400 as a pore forming additive[J]. Desalination, 2012, 286: 131-137. |
| 23 | Idris A, Ahmed I, Limin M A. Influence of lithium chloride, lithium bromide and lithium fluoride additives on performance of polyethersulfone membranes and its application in the treatment of palm oil mill effluent[J]. Desalination, 2010, 250(2): 805-809. |
| 24 | Zheng L B, Wu Z J, Wei Y S, et al. Preparation of PVDF-CTFE hydrophobic membranes for MD application: effect of LiCl-based mixed additives[J]. Journal of Membrane Science, 2016, 506: 71-85. |
| 25 | 王旭东, 满丽, 王磊, 等. 添加剂对PVDF平板超滤膜性能和结构的影响[J]. 水处理技术, 2010, 36(3): 33-37. |
| Wang X D, Man L, Wang L, et al. Effect of additives on the properties and structures of polyvinylidene fluoride (PVDF) flat ultrafiltration membranes[J]. Technology of Water Treatment, 2010, 36(3): 33-37. | |
| 26 | Lee H J, Won J, Lee H, et al. Solution properties of poly(amic acid)-NMP containing LiCl and their effects on membrane morphologies[J]. Journal of Membrane Science, 2002, 196(2): 267-277. |
| 27 | Aryanti P T P, Joscarita S R, Wardani A K, et al. The influence of PEG400 and acetone on polysulfone membrane morphology and fouling behaviour[J]. Journal of Engineering and Technological Sciences, 2016, 48(2): 135-149. |
| 28 | Liang B, Li Q, Cao B, et al. Water permeance, permeability and desalination properties of the sulfonic acid functionalized composite pervaporation membranes[J]. Desalination, 2018, 433: 132-140. |
| 29 | Chakrabarty B, Ghoshal A K, Purkait M K. Effect of molecular weight of PEG on membrane morphology and transport properties[J]. Journal of Membrane Science, 2008, 309(1/2): 209-221. |
| 30 | Kim J H, Lee K H. Effect of PEG additive on membrane formation by phase inversion[J]. Journal of Membrane Science, 1998, 138(2): 153-163. |
| 31 | 赵姗姗, 王鹏, 郑彤, 等. 添加剂对聚醚砜酮超滤膜结构和性能的影响[J]. 水处理技术, 2013, 39(4): 28-32. |
| Zhao S S, Wang P, Zheng T, et al. Effect of additives on the structures and properties of poly(phthalazine ether sulfone ketone)(PPESK) ultrafiltration membrane[J]. Technology of Water Treatment, 2013, 39(4): 28-32. | |
| 32 | Zhu T, Chang S C, Song Y F, et al. PVP-encapsulated CoFe2O4/rGO composites with controllable electromagnetic wave absorption performance[J]. Chemical Engineering Journal, 2019, 373: 755-766. |
| 33 | 田立, 张云, 汪朝晖, 等. 铸膜液中大分子量PVP含量对PVDF膜的影响[J]. 化工新型材料, 2014, 42(12): 185-188. |
| Tian L, Zhang Y, Wang Z H, et al. Influence of macromolecular PVP composition in casting solution on the formation of PVDF membranes[J]. New Chemical Materials, 2014, 42(12): 185-188. | |
| 34 | Al Malek S A, Abu Seman M N, Johnson D, et al. Formation and characterization of polyethersulfone membranes using different concentrations of polyvinylpyrrolidone[J]. Desalination, 2012, 288: 31-39. |
| [1] | 杨百玉, 寇悦, 姜峻韬, 詹亚力, 王庆宏, 陈春茂. 炼化碱渣湿式氧化预处理过程DOM的化学转化特征[J]. 化工学报, 2023, 74(9): 3912-3920. |
| [2] | 赵亚欣, 张雪芹, 王荣柱, 孙国, 姚善泾, 林东强. 流穿模式离子交换层析去除单抗聚集体[J]. 化工学报, 2023, 74(9): 3879-3887. |
| [3] | 张佳怡, 何佳莉, 谢江鹏, 王健, 赵鹬, 张栋强. 渗透汽化技术用于锂电池生产中N-甲基吡咯烷酮回收的研究进展[J]. 化工学报, 2023, 74(8): 3203-3215. |
| [4] | 张瑞航, 曹潘, 杨锋, 李昆, 肖朋, 邓春, 刘蓓, 孙长宇, 陈光进. ZIF-8纳米流体天然气乙烷回收工艺的产品纯度关键影响因素分析[J]. 化工学报, 2023, 74(8): 3386-3393. |
| [5] | 邢雷, 苗春雨, 蒋明虎, 赵立新, 李新亚. 井下微型气液旋流分离器优化设计与性能分析[J]. 化工学报, 2023, 74(8): 3394-3406. |
| [6] | 刘爽, 张霖宙, 许志明, 赵锁奇. 渣油及其组分黏度的分子层次组成关联研究[J]. 化工学报, 2023, 74(8): 3226-3241. |
| [7] | 杨欣, 彭啸, 薛凯茹, 苏梦威, 吴燕. 分子印迹-TiO2光电催化降解增溶PHE废水性能研究[J]. 化工学报, 2023, 74(8): 3564-3571. |
| [8] | 张缘良, 栾昕奇, 苏伟格, 李畅浩, 赵钟兴, 周利琴, 陈健民, 黄艳, 赵祯霞. 离子液体复合萃取剂选择性萃取尼古丁的研究及DFT计算[J]. 化工学报, 2023, 74(7): 2947-2956. |
| [9] | 高金明, 郭玉娇, 鄂承林, 卢春喜. 一种封闭罩内顺流多旋臂气液分离器的分离特性研究[J]. 化工学报, 2023, 74(7): 2957-2966. |
| [10] | 文兆伦, 李沛睿, 张忠林, 杜晓, 侯起旺, 刘叶刚, 郝晓刚, 官国清. 基于自热再生的隔壁塔深冷空分工艺设计及优化[J]. 化工学报, 2023, 74(7): 2988-2998. |
| [11] | 王杰, 丘晓琳, 赵烨, 刘鑫洋, 韩忠强, 许雍, 蒋文瀚. 聚电解质静电沉积改性PHBV抗氧化膜的制备与性能研究[J]. 化工学报, 2023, 74(7): 3068-3078. |
| [12] | 刘杰, 吴立盛, 李锦锦, 罗正鸿, 周寅宁. 含乙烯基胺酯键聚醚类可逆交联聚合物的制备及性能研究[J]. 化工学报, 2023, 74(7): 3051-3057. |
| [13] | 韩奎奎, 谭湘龙, 李金芝, 杨婷, 张春, 张永汾, 刘洪全, 于中伟, 顾学红. 四通道中空纤维MFI分子筛膜用于二甲苯异构体分离[J]. 化工学报, 2023, 74(6): 2468-2476. |
| [14] | 朱兴驰, 郭志远, 纪志永, 汪婧, 张盼盼, 刘杰, 赵颖颖, 袁俊生. 选择性电渗析镁锂分离过程模拟优化[J]. 化工学报, 2023, 74(6): 2477-2485. |
| [15] | 龙臻, 王谨航, 任俊杰, 何勇, 周雪冰, 梁德青. 离子液体协同PVCap抑制天然气水合物生成实验研究[J]. 化工学报, 2023, 74(6): 2639-2646. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备 11010102001995号
京公网安备 11010102001995号