化工学报 ›› 2019, Vol. 70 ›› Issue (S2): 50-53.DOI: 10.11949/0438-1157.20190579
收稿日期:2019-05-28
修回日期:2019-06-12
出版日期:2019-09-06
发布日期:2019-09-06
通讯作者:
宋渤
作者简介:宋渤(1986—),男,博士,副教授,基金资助:
Bo SONG1( ),Min YANG1,Ran AN2,Xiaopo WANG1
),Min YANG1,Ran AN2,Xiaopo WANG1
Received:2019-05-28
Revised:2019-06-12
Online:2019-09-06
Published:2019-09-06
Contact:
Bo SONG
摘要:
采用当前文献中已知最佳的从头算势能和极化率计算了多种稀有气体的第二介电维里系数。研究对象包括氦-4、氦-3、氖和氩。根据经典统计力学公式和一二阶量子修正项对第二介电维里系数进行了计算,在此基础上,通过[1/1]帕德近似将研究的温度下限拓展至25 K。第二介电维里系数理论值的不确定度可以由势能和极化率的不确定度进行评估,再结合与文献实验数据的比较可以发现,理论计算结果为计量、仪器标定等领域提供了所需的高精度基础数据。
中图分类号:
宋渤,杨敏,安然,王晓坡. 单原子气体第二介电维里系数准确计算[J]. 化工学报, 2019, 70(S2): 50-53.
Bo SONG,Min YANG,Ran AN,Xiaopo WANG. Accurate calculations of second dielectric virial coefficient of noble gases[J]. CIESC Journal, 2019, 70(S2): 50-53.
| 文献 | 年份 | 对象 | 不确定度 |
|---|---|---|---|
| [ | 2017 | He-He | 0.0023% |
| [ | 2008 | Ne-Ne | 未给出 |
| [ | 2010 | Ar-Ar | 0.32% |
表1 从头算分子间势能
Table 1 Ab initio interatomic potential
| 文献 | 年份 | 对象 | 不确定度 |
|---|---|---|---|
| [ | 2017 | He-He | 0.0023% |
| [ | 2008 | Ne-Ne | 未给出 |
| [ | 2010 | Ar-Ar | 0.32% |
| 文献 | 年份 | 对象 | 不确定度 |
|---|---|---|---|
| [ | 2011 | He-He | 0.00023% |
| [ | 2003 | Ne-Ne | 未给出 |
| [ | 2010 | Ar-Ar | 未给出 |
表2 从头算分子间极化率
Table 2 Ab initio interatomic polarizability
| 文献 | 年份 | 对象 | 不确定度 |
|---|---|---|---|
| [ | 2011 | He-He | 0.00023% |
| [ | 2003 | Ne-Ne | 未给出 |
| [ | 2010 | Ar-Ar | 未给出 |
| T/K | B ε/ (cm6·mol-2) | [1/1] Padé/(cm6·mol-2) | σ/ (cm6·mol-2) | σ from V/% | σ from Δα ave/% |
|---|---|---|---|---|---|
| 25 | -0.01162 | -0.008735 | 0.000177 | 0.13 | 99.86 |
| 30 | -0.01179 | -0.01017 | 0.00018 | 0.13 | 99.87 |
| 35 | -0.01255 | -0.01153 | 0.00017 | 0.13 | 99.87 |
| 40 | -0.01353 | -0.01285 | 0.00017 | 0.13 | 99.87 |
| 45 | -0.01459 | -0.01411 | 0.00017 | 0.12 | 99.88 |
| 50 | -0.01568 | -0.01533 | 0.00017 | 0.12 | 99.88 |
| 60 | -0.01785 | -0.01764 | 0.00017 | 0.12 | 99.88 |
| 70 | -0.01995 | -0.01982 | 0.00017 | 0.12 | 99.88 |
| 80 | -0.02197 | -0.02188 | 0.00017 | 0.12 | 99.88 |
| 90 | -0.02391 | -0.02385 | 0.00017 | 0.12 | 99.88 |
| 100 | -0.02578 | -0.02573 | 0.00017 | 0.12 | 99.88 |
| 150 | -0.03417 | -0.03415 | 0.00017 | 0.11 | 99.89 |
| 200 | -0.04142 | -0.04141 | 0.00017 | 0.11 | 99.89 |
| 250 | -0.04787 | -0.04787 | 0.00018 | 0.11 | 99.89 |
| 273.15 | -0.05065 | -0.05065 | 0.00018 | 0.11 | 99.89 |
| 273.16 | -0.05065 | -0.05065 | 0.00018 | 0.11 | 99.89 |
| 293.15 | -0.05296 | -0.05296 | 0.00018 | 0.11 | 99.89 |
| 298.15 | -0.05353 | -0.05352 | 0.00018 | 0.11 | 99.89 |
| 300 | -0.05373 | -0.05373 | 0.00018 | 0.11 | 99.89 |
| 350 | -0.05913 | -0.05912 | 0.00018 | 0.11 | 99.89 |
| 400 | -0.06414 | -0.06414 | 0.00018 | 0.11 | 99.89 |
| 450 | -0.06884 | -0.06884 | 0.00018 | 0.11 | 99.89 |
| 500 | -0.07327 | -0.07326 | 0.00018 | 0.11 | 99.89 |
| 600 | -0.08145 | -0.08145 | 0.00018 | 0.11 | 99.89 |
| ? | ? | ? | ? | ? | ? |
| 10000 | -0.29473 | -0.29473 | 0.000199 | 0.09 | 99.91 |
表3 氦-4第二介电维里系数
Table 3 Second dielectric virial coefficient of helium-4
| T/K | B ε/ (cm6·mol-2) | [1/1] Padé/(cm6·mol-2) | σ/ (cm6·mol-2) | σ from V/% | σ from Δα ave/% |
|---|---|---|---|---|---|
| 25 | -0.01162 | -0.008735 | 0.000177 | 0.13 | 99.86 |
| 30 | -0.01179 | -0.01017 | 0.00018 | 0.13 | 99.87 |
| 35 | -0.01255 | -0.01153 | 0.00017 | 0.13 | 99.87 |
| 40 | -0.01353 | -0.01285 | 0.00017 | 0.13 | 99.87 |
| 45 | -0.01459 | -0.01411 | 0.00017 | 0.12 | 99.88 |
| 50 | -0.01568 | -0.01533 | 0.00017 | 0.12 | 99.88 |
| 60 | -0.01785 | -0.01764 | 0.00017 | 0.12 | 99.88 |
| 70 | -0.01995 | -0.01982 | 0.00017 | 0.12 | 99.88 |
| 80 | -0.02197 | -0.02188 | 0.00017 | 0.12 | 99.88 |
| 90 | -0.02391 | -0.02385 | 0.00017 | 0.12 | 99.88 |
| 100 | -0.02578 | -0.02573 | 0.00017 | 0.12 | 99.88 |
| 150 | -0.03417 | -0.03415 | 0.00017 | 0.11 | 99.89 |
| 200 | -0.04142 | -0.04141 | 0.00017 | 0.11 | 99.89 |
| 250 | -0.04787 | -0.04787 | 0.00018 | 0.11 | 99.89 |
| 273.15 | -0.05065 | -0.05065 | 0.00018 | 0.11 | 99.89 |
| 273.16 | -0.05065 | -0.05065 | 0.00018 | 0.11 | 99.89 |
| 293.15 | -0.05296 | -0.05296 | 0.00018 | 0.11 | 99.89 |
| 298.15 | -0.05353 | -0.05352 | 0.00018 | 0.11 | 99.89 |
| 300 | -0.05373 | -0.05373 | 0.00018 | 0.11 | 99.89 |
| 350 | -0.05913 | -0.05912 | 0.00018 | 0.11 | 99.89 |
| 400 | -0.06414 | -0.06414 | 0.00018 | 0.11 | 99.89 |
| 450 | -0.06884 | -0.06884 | 0.00018 | 0.11 | 99.89 |
| 500 | -0.07327 | -0.07326 | 0.00018 | 0.11 | 99.89 |
| 600 | -0.08145 | -0.08145 | 0.00018 | 0.11 | 99.89 |
| ? | ? | ? | ? | ? | ? |
| 10000 | -0.29473 | -0.29473 | 0.000199 | 0.09 | 99.91 |
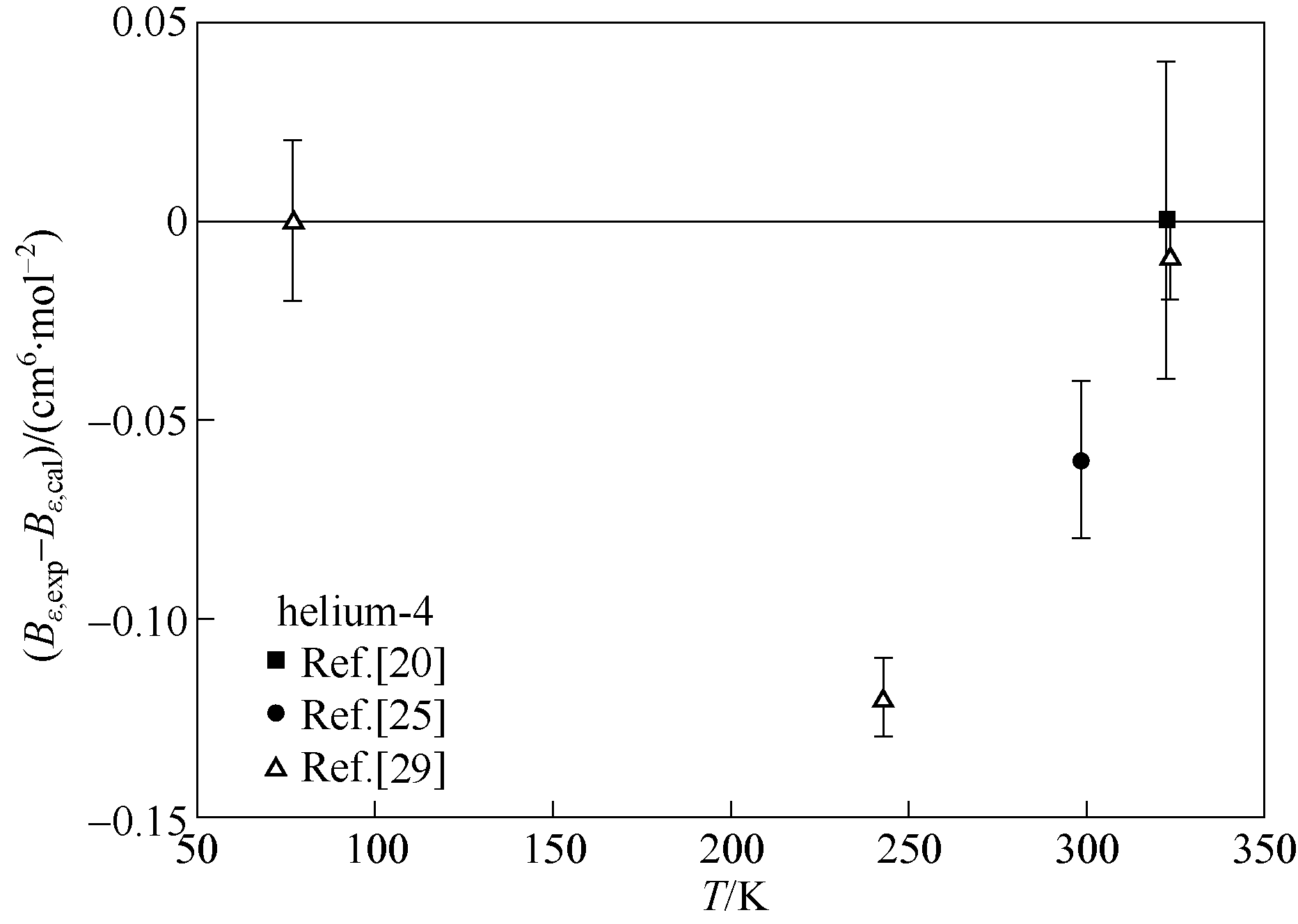
图1 氦-4第二介电维里系数实验值与本文理论计算结果对比
Fig.1 Comparisons of experimental second dielectric virial coefficient data of helium-4 with calculated values of this work
| 1 | Stock M , Davis R , de Mirandés E , et al . The revision of the SI—the result of three decades of progress in metrology[J]. Metrologia, 2019, 56(2): 022001. |
| 2 | Newell D B , Cabiati F , Fischer J , et al . The CODATA 2017 values of h, e, k, and NA for the revision of the SI[J]. Metrologia, 2018, 55(1): L13. |
| 3 | Mohr P J , Newell D B , Taylor B N , et al . Data and analysis for the CODATA 2017 special fundamental constants adjustment[J]. Metrologia, 2018, 55(1): 125. |
| 4 | de Podesta M , Mark D F , Dymock R C , et al . Re-estimation of argon isotope ratios leading to a revised estimate of the Boltzmann constant[J]. Metrologia, 2017, 54(5): 683. |
| 5 | Feng X J , Zhang J T , Lin H , et al . Determination of the Boltzmann constant with cylindrical acoustic gas thermometry: new and previous results combined[J]. Metrologia, 2017, 54(5): 748. |
| 6 | Pitre L , Sparasci F , Risegari L , et al . New measurement of the Boltzmann constant k by acoustic thermometry of helium-4 gas[J]. Metrologia, 2017, 54(6): 856. |
| 7 | Gaiser C , Fellmuth B , Haft N , et al . Final determination of the Boltzmann constant by dielectric-constant gas thermometry[J]. Metrologia, 2017, 54(3): 280. |
| 8 | 王延儒, 马沛生 . 第二维里系数与温度的关联及Lennard-Jones和Stockmayer位能参数的推算[J]. 化工学报, 1988, 39(5): 608-623. |
| Wang Y R , Ma P S . Correlations of the second virial coefficient with temperature and determinations of Lennard-Jones and Stockmayer potential parameters[J]. Journal of Chemical Industry and Engineering (China), 1988, 39(5): 608-623. | |
| 9 | 刘强, 冯晓娟, 段远源 . 圆柱定程干涉法确定工质的第二维里系数[J]. 化工学报, 2013, 64(8): 2711-2717. |
| Liu Q , Feng X J , Duan Y Y . Determination of the second virial coefficient for working fluid using a cylindrical resonator[J]. CIESC Journal, 2013, 64(8): 2711-2717. | |
| 10 | 刘强, 冯晓娟, 段远源 . 气相声速测量偏差对导出热力学性质的影响[J]. 化工学报, 2014, 65(4): 1162-1168. |
| Liu Q , Feng X J , Duan Y Y . Effects of measurement deviation in gaseous speed of sound on derived thermodynamic properties[J]. CIESC Journal, 2014, 65(4): 1162-1168. | |
| 11 | 何茂刚, 郑雄, 张颖, 等 . 三种脂肪酸甲酯音速测量与相关热物理性质推算[J]. 化工学报, 2016, 67(12): 4922-4928. |
| He M G , Zheng X , Zhang Y , et al . Measurement of sound speed and derived properties of three FAMEs[J], CIESC Journal, 2016, 67(12): 4922-4928. | |
| 12 | Przybytek M , Cencek W , Jeziorski B , et al . Pair potential with submillikelvin uncertainties and nonadiabatic treatment of the halo state of the helium dimer[J]. Phys. Rev. Lett., 2017, 119(12): 123401. |
| 13 | Cencek W , Komasa J , Szalewicz K . Collision-induced dipole polarizability of helium dimer from explicitly correlated calculations[J]. J. Chem. Phys., 2011, 135(1): 014301. |
| 14 | Hellmann R , Bich E , Vogel E . Ab initio potential energy curve for the neon atom pair and thermophysical properties of the dilute neon gas (Ⅰ): Neon-neon interatomic potential and rovibrational spectra[J]. Mol. Phys., 2008, 106(1): 133-140. |
| 15 | Hättig C , Cacheiro J L , Fernández B , et al . Ab initio calculation of the refractivity and hyperpolarizability second virial coefficients of neon gas[J]. Mol. Phys., 2003, 101(13): 1983-1995. |
| 16 | Patkowski K , Szalewicz K . Argon pair potential at basis set and excitation limits[J]. J. Chem. Phys., 2010, 133(9): 094304. |
| 17 | Vogel E , Jäger B , Hellmann R , et al . Ab initio pair potential energy curve for the argon atom pair and thermophysical properties for the dilute argon gas (Ⅱ): Thermophysical properties for low-density argon[J]. Mol. Phys., 2010, 108(24): 3335-3352. |
| 18 | Moszynski R , Heijmen T G , Wormer P E , et al . Ab initio collision-induced polarizability, polarized and depolarized Raman spectra, and second dielectric virial coefficient of the helium diatom[J]. J. Chem. Phys., 1996, 104(18): 6997-7007. |
| 19 | Johnston D , Oudemans G , Cole R . Dielectric constants of imperfect gases (Ⅰ): Helium, argon, nitrogen, and methane[J]. J. Chem. Phys., 1960, 33(5): 1310-1317. |
| 20 | Orcutt R H , Cole R H . Dielectric constants of imperfect gases (Ⅲ): Atomic gases, hydrogen, and nitrogen[J]. J. Chem. Phys., 1967, 46(2): 697-702. |
| 21 | Kerr E C , Sherman R H . Dielectric polarizability of helium-3[C]// Proc. 11th Int. Conf. Low Temp. Phys. St Andrew: University of St Andrew, 1969: 236-239. |
| 22 | Kerr E C , Sherman R H . The molar polarizability of 3He at low temperatures and its density dependence[J]. J. Low Temp. Phys., 1970, 3(5): 451-461. |
| 23 | Bose T K , Cole R . Dielectric and pressure virial coefficients of imperfect gases (Ⅱ): CO2-argon mixtures[J]. J. Chem. Phys., 1970, 52(1): 140-147. |
| 24 | Kirouac S , Bose T K . Polarizability and dielectric properties of helium[J]. J. Chem. Phys., 1976, 64(4): 1580-1582. |
| 25 | Vidal D , Lallemand M . Evolution of the Clausius-Mossotti function of noble gases and nitrogen, at moderate and high density, near room temperature[J]. J. Chem. Phys., 1976, 64(11): 4293-4302. |
| 26 | Lallemand M , Vidal D . Variation of the polarizability of noble gases with density[J]. J. Chem. Phys., 1977, 66(11): 4776-4780. |
| 27 | Gugan D , Michel G . Dielectric constant gas thermometry from 4.2 to 27.1 K[J]. Metrologia, 1980, 16(4): 149. |
| 28 | Bose T K , Boudjarane K , Huot J , et al . Refractivity virial coefficients of C2H4 and C2H4-Ar mixtures[J]. J. Chem. Phys., 1988, 89(12): 7435-7440. |
| 29 | Huot J , Bose T K . Experimental determination of the dielectric virial coefficients of atomic gases as a function of temperature[J]. J. Chem. Phys., 1991, 95(4): 2683-2687. |
| 30 | White M , Gugan D . Direct measurements of the dielectric virial coefficients of 4He between 3 K and 18 K[J]. Metrologia, 1992, 29(1): 37. |
| [1] | 何晓崐, 刘锐, 薛园, 左然. MOCVD生长AlN单晶薄膜的气相和表面化学反应综述[J]. 化工学报, 2023, 74(7): 2800-2813. |
| [2] | 李晨曦, 刘永峰, 张璐, 刘海峰, 宋金瓯, 何旭. O2/CO2氛围下正庚烷的燃烧机理研究[J]. 化工学报, 2023, 74(5): 2157-2169. |
| [3] | 陈科, 杜理, 曾英, 任思颖, 于旭东. 四元体系LiCl+MgCl2+CaCl2+H2O 323.2 K相平衡研究及计算[J]. 化工学报, 2023, 74(5): 1896-1903. |
| [4] | 姚晓宇, 沈俊, 李健, 李振兴, 康慧芳, 唐博, 董学强, 公茂琼. 流体气液临界参数测量方法研究进展[J]. 化工学报, 2023, 74(5): 1847-1861. |
| [5] | 毛元敬, 杨智, 莫松平, 郭浩, 陈颖, 罗向龙, 陈健勇, 梁颖宗. C6~C10烷醇的SAFT-VR Mie状态方程参数回归及其热物性研究[J]. 化工学报, 2023, 74(3): 1033-1041. |
| [6] | 程文婷, 李杰, 徐丽, 程芳琴, 刘国际. AlCl3·6H2O在FeCl3、CaCl2、KCl及KCl–FeCl3溶液中溶解度的实验及预测[J]. 化工学报, 2023, 74(2): 642-652. |
| [7] | 杨松涛, 李东洋, 牛玉清, 李鑫钢, 康绍辉, 李洪, 叶开凯, 周志全, 高鑫. 氟化物势能函数和热力学性质的分子模拟研究进展[J]. 化工学报, 2022, 73(9): 3828-3840. |
| [8] | 俞夏琪, 冯格, 赵金燕, 李嘉远, 邓声威, 郑靖楠, 李雯雯, 王亚秋, 沈榄, 刘旭, 徐威威, 王建国, 王式彬, 姚子豪, 毛成立. 基体(TDI-TMP-T313)与氧化剂(AP)相互作用的第一性原理研究[J]. 化工学报, 2022, 73(8): 3511-3517. |
| [9] | 孙哲, 金华强, 李康, 顾江萍, 黄跃进, 沈希. 基于知识数据化表达的制冷空调系统故障诊断方法[J]. 化工学报, 2022, 73(7): 3131-3144. |
| [10] | 刘晓涯, 王金超, 刘莹, 马敬环. 水合肼制氢纳米催化剂改性制备及机理研究进展[J]. 化工学报, 2022, 73(7): 2819-2834. |
| [11] | 任嘉辉, 刘豫, 刘朝, 刘浪, 李莹. 基于分子指纹和拓扑指数的工质临界温度理论预测[J]. 化工学报, 2022, 73(4): 1493-1500. |
| [12] | 孙铭泽, 马宁, 李浩然, 姜海峰, 洪文鹏, 牛晓娟. 中低温超临界CO2及其混合工质布雷顿循环热力学分析[J]. 化工学报, 2022, 73(3): 1379-1388. |
| [13] | 李怀旭, 孙晓岩, 陶少辉, 夏力, 项曙光. 基于分子热力学性质和密度峰聚类的脱硫汽油集总[J]. 化工学报, 2022, 73(12): 5449-5460. |
| [14] | 高腾飞, 李国选, 雷志刚. 从催化裂化柴油中分离联苯的溶剂筛选:实验和计算热力学[J]. 化工学报, 2022, 73(12): 5314-5323. |
| [15] | 任思颖, 于旭东, 罗军, 冯霞, 赵志星, 姚智豪. 298.2 K四元体系Li+, K+, |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备 11010102001995号
京公网安备 11010102001995号