化工学报 ›› 2024, Vol. 75 ›› Issue (5): 1750-1764.DOI: 10.11949/0438-1157.20240091
赵亭亭1( ), 鄢立祥1, 唐福利2, 肖敏之1, 谭烨1, 宋刘斌1(
), 鄢立祥1, 唐福利2, 肖敏之1, 谭烨1, 宋刘斌1( ), 肖忠良1(
), 肖忠良1( ), 李灵均3
), 李灵均3
收稿日期:2024-01-19
修回日期:2024-03-03
出版日期:2024-05-25
发布日期:2024-06-25
通讯作者:
宋刘斌,肖忠良
作者简介:赵亭亭(1994—),女,博士,讲师,zhaott_468@163.com
基金资助:
Tingting ZHAO1( ), Lixiang YAN1, Fuli TANG2, Minzhi XIAO1, Ye TAN1, Liubin SONG1(
), Lixiang YAN1, Fuli TANG2, Minzhi XIAO1, Ye TAN1, Liubin SONG1( ), Zhongliang XIAO1(
), Zhongliang XIAO1( ), Lingjun LI3
), Lingjun LI3
Received:2024-01-19
Revised:2024-03-03
Online:2024-05-25
Published:2024-06-25
Contact:
Liubin SONG, Zhongliang XIAO
摘要:
光辅助Li-CO2电池具有理论能量密度高、环境友好等特点,是下一代高比能电池系统的重要发展方向。然而,正极处CO2还原/析出反应存在动力学缓慢等问题,限制了Li-CO2电池发展。光辅助技术利用正极负载的光催化剂吸收光能,产生电子和空穴以驱动化学反应,有利于提升电池性能。本文阐述了光辅助Li-CO2电池的光化学原理及充放电反应机制,详细列举了正极光催化剂的设计策略及具体实例。通过深入探讨Li-CO2电池的光催化反应机理,进一步理解了光催化剂结构对电池性能的影响机制。此外,还讨论了光辅助Li-CO2电池的基本认识、当前面临的挑战以及对光催化剂发展前景的展望,可为新能源材料领域的技术研究提供参考,有助于推动Li-CO2电池的实用化进程。
中图分类号:
赵亭亭, 鄢立祥, 唐福利, 肖敏之, 谭烨, 宋刘斌, 肖忠良, 李灵均. 光辅助锂-二氧化碳电池催化剂的设计策略与反应机理研究进展[J]. 化工学报, 2024, 75(5): 1750-1764.
Tingting ZHAO, Lixiang YAN, Fuli TANG, Minzhi XIAO, Ye TAN, Liubin SONG, Zhongliang XIAO, Lingjun LI. Research progress on design strategies and reaction mechanisms of photo-assisted Li-CO2 battery catalysts[J]. CIESC Journal, 2024, 75(5): 1750-1764.

图4 光诱导放电过程的工作机制(a);In2S3@CNT/SS的能带图(b);ICS、In2S3NS/SS和CNT对紫外线的光电流响应(c)[40]
Fig.4 The working mechanism for the light-induced discharging process (a); Band diagram of the In2S3@CNT/SS (b); Photocurrent response to UV light of ICS, In2S3NS/SS, and CNT (c)[40]
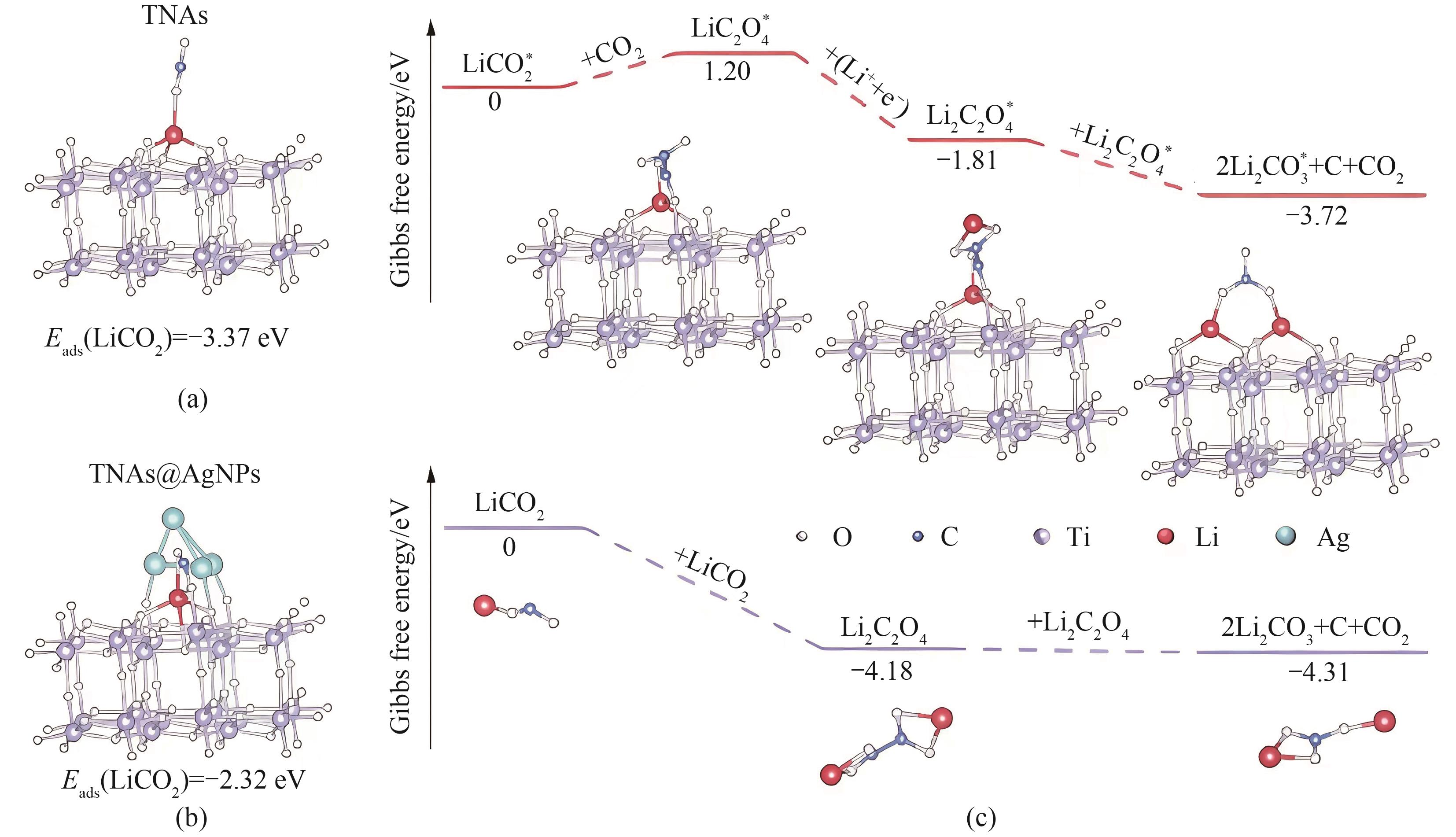
图5 中间LiCO2分子在TNA上的优化结构和吸附能(a);中间LiCO2分子在TNA@AgNPs上的优化结构和吸附能(b);电池表面和溶液介导反应途径的Gibbs自由能(c)[39]
Fig.5 Optimized structures and adsorption energy of intermediate LiCO2 molecules on the TNAs (a); Optimized structures and adsorption energy of intermediate LiCO2 molecules on the TNAs@AgNPs (b); Gibbs free energy of battery surface and solution-mediated reaction pathways (c)[39]

图6 基于TiO2 NAs/CT和RuO2-TiO2 NAs/CT阴极的Li-CO2电池中Li2CO3不同生长机制的示意图(a);Li2CO3在TiO2和RuO2表面上的优化结构和相应的结合能(b);吸附在TiO2和RuO2表面上的CO2或Li2CO3的电荷密度差Δρ(c)[38]
Fig.6 Schematic illustration of different growth mechanisms of Li2CO3 in the Li-CO2 batteries based on TiO2 NAs/CT and RuO2-TiO2 NAs/CT cathodes (a); The optimized structures and the corresponding binding energy of Li2CO3 on TiO2 and RuO2 surfaces (b); The differential charge density Δρ of CO2 or Li2CO3 adsorbed on TiO2 and RuO2 surfaces (c)[38]
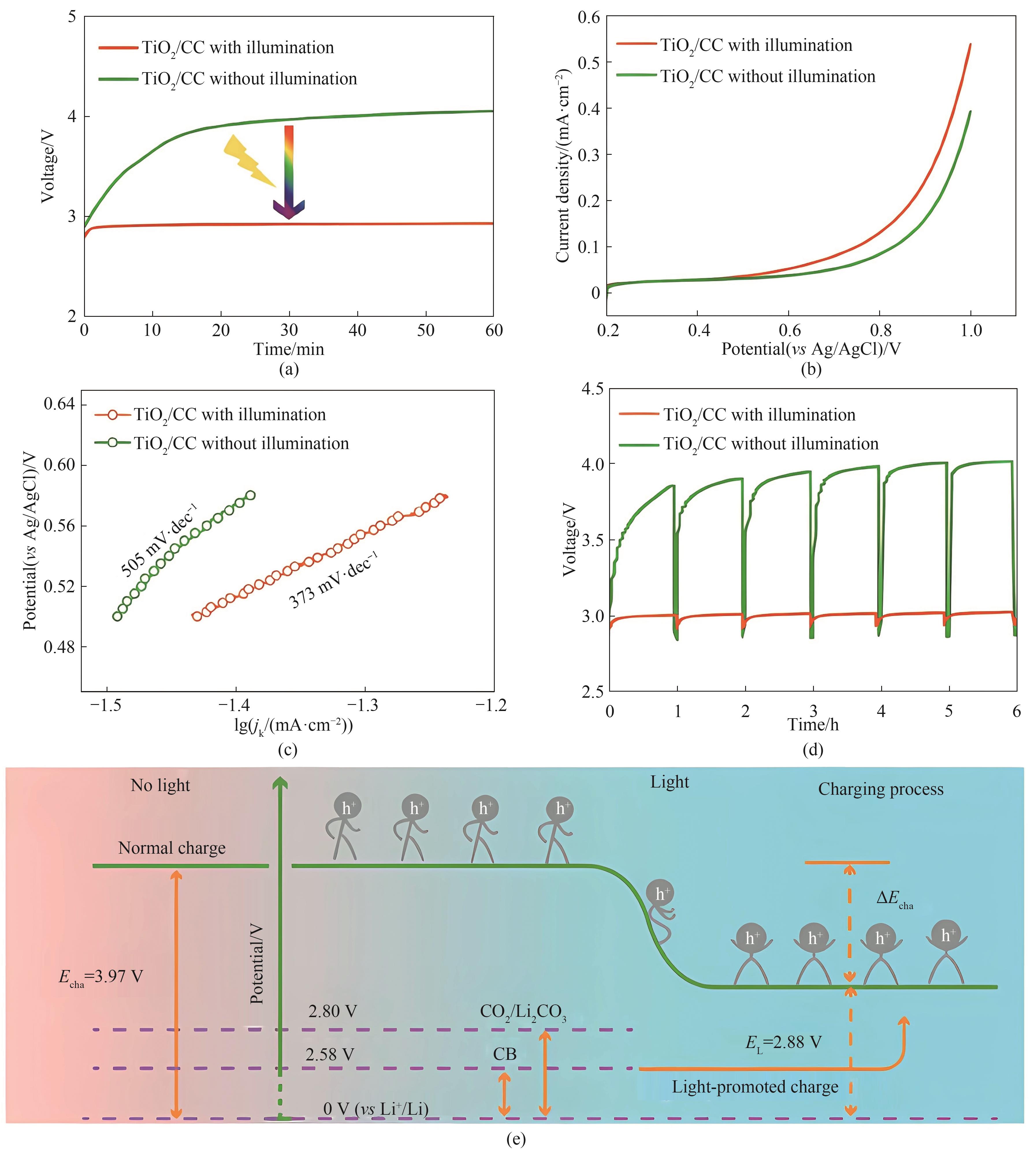
图7 使用TiO2/CC阴极的Li-CO2电池在照明和不照明情况下的充电曲线(a);在0.5 mol·L-1 CO2饱和LiTFSI/DME溶液中,扫描速率为5 mV·s-1,光照和不光照下TiO2/CC在CO2ER区域的LSV曲线(b);相应的Tafel曲线(c);采用TiO2/CC阴极的Li-CO2电池在光照和不光照下的恒流间歇滴定技术曲线(d);照明下Li-CO2电池充电电压降低的能量图(e)[36]
Fig.7 Charge curves of Li-CO2 batteries with TiO2/CC cathode with and without illumination (a); LSV curves measured in the CO2ER region of TiO2/CC with and without illumination in 0.5 mol·L-1 CO2-saturated LiTFSI/DME solution at a scan rate of 5 mV·s-1 (b); Corresponding Tafel curves (c); Galvanostatic intermittent titration technique curves obtained from Li-CO2 battery with TiO2/CC cathode with and without illumination (d); Schematic of energy diagrams for the reduced charge voltage of Li-CO2 battery under illumination (e)[36]
| 光催化剂 | 首次效率/% | 电流密度/(mA·cm-2) | 放电比容量/(mAh·cm-2) | 充电/放电电压平台/V | 循环次数(圈) | 文献 |
|---|---|---|---|---|---|---|
| TiO2/CC | 97.2 | 0.01 | 0.01 | 2.82/2.88 | 30 | [ |
| RuO2/TiO2 | 95.5 | 250 mA·g-1 | 1000 mAh·g-1 | 2.78/2.91 | 238 | [ |
| TiO2@Ag | 87.1 | 0.01 | 0.1 | 2.49/2.86 | 100 | [ |
| Cu2O/CNT | 85 | 100 mA·g-1 | 100 mAh·g-1 | 2.5/4.5 | 50 | [ |
| In2S3@CNT/SS | 98.1 | 0.01 | 0.01 | 3.14/3.2 | 25 | [ |
| CNT@C3N4 | 98.8 | 0.02 | 0.02 | 3.24/3.28 | 100 | [ |
| SiC/RGO | 84.4 | 20 mA·g-1 | 0.01 | 2.77/3.28 | 20 | [ |
| CoPc-Mn-O | 98.5 | 0.02 | — | 3.20/3.25 | 30 | [ |
| Au@TiO2 | 92.4 | 0.01 | 0.01 | 2.95/3.49 | 200 | [ |
| 混合相TiO2 | — | 0.025 | — | 2.2/3.0 | 52 | [ |
表1 光辅助Li-CO2电池的结构和性能
Table 1 Structure and performance of photo-assisted Li-CO2 batteries
| 光催化剂 | 首次效率/% | 电流密度/(mA·cm-2) | 放电比容量/(mAh·cm-2) | 充电/放电电压平台/V | 循环次数(圈) | 文献 |
|---|---|---|---|---|---|---|
| TiO2/CC | 97.2 | 0.01 | 0.01 | 2.82/2.88 | 30 | [ |
| RuO2/TiO2 | 95.5 | 250 mA·g-1 | 1000 mAh·g-1 | 2.78/2.91 | 238 | [ |
| TiO2@Ag | 87.1 | 0.01 | 0.1 | 2.49/2.86 | 100 | [ |
| Cu2O/CNT | 85 | 100 mA·g-1 | 100 mAh·g-1 | 2.5/4.5 | 50 | [ |
| In2S3@CNT/SS | 98.1 | 0.01 | 0.01 | 3.14/3.2 | 25 | [ |
| CNT@C3N4 | 98.8 | 0.02 | 0.02 | 3.24/3.28 | 100 | [ |
| SiC/RGO | 84.4 | 20 mA·g-1 | 0.01 | 2.77/3.28 | 20 | [ |
| CoPc-Mn-O | 98.5 | 0.02 | — | 3.20/3.25 | 30 | [ |
| Au@TiO2 | 92.4 | 0.01 | 0.01 | 2.95/3.49 | 200 | [ |
| 混合相TiO2 | — | 0.025 | — | 2.2/3.0 | 52 | [ |

图8 通过基于等离子体和激子的方法描绘等离子体半导体纳米材料的太阳光谱光热效应的示意图(a);Au@TiO2和TiO2表面温度升高的开/关响应(b);光照条件下TiO2(c)和Au@TiO2(d)球体表面温度升高的分布;Au@TiO2和TiO2球的I-T曲线(e);光照条件下TiO2 和Au@TiO2球表面的电近场分布(f);TiO2和Au@TiO2球在光照和无光照情况下的CDR过程的LSV曲线(g)[44]
Fig.8 Schematic depiction of the solar-spectrum photothermal effect of plasmonic semiconductor nanomaterials via plasmon-and exciton-based approaches (a); On/off response of the surface temperature increase of Au@TiO2 and TiO2 (b); Distribution of the surface temperature increase on TiO2 (c) and Au@TiO2 (d) spheres under the illumination conditions; I-T curves of Au@TiO2 and TiO2 spheres (e); Distribution of the electric near field under the illumination conditions on TiO2 and Au@TiO2 spheres surface (f); LSV curves for the CDR process of TiO2 and Au@TiO2 spheres with and without illumination (g)[44]
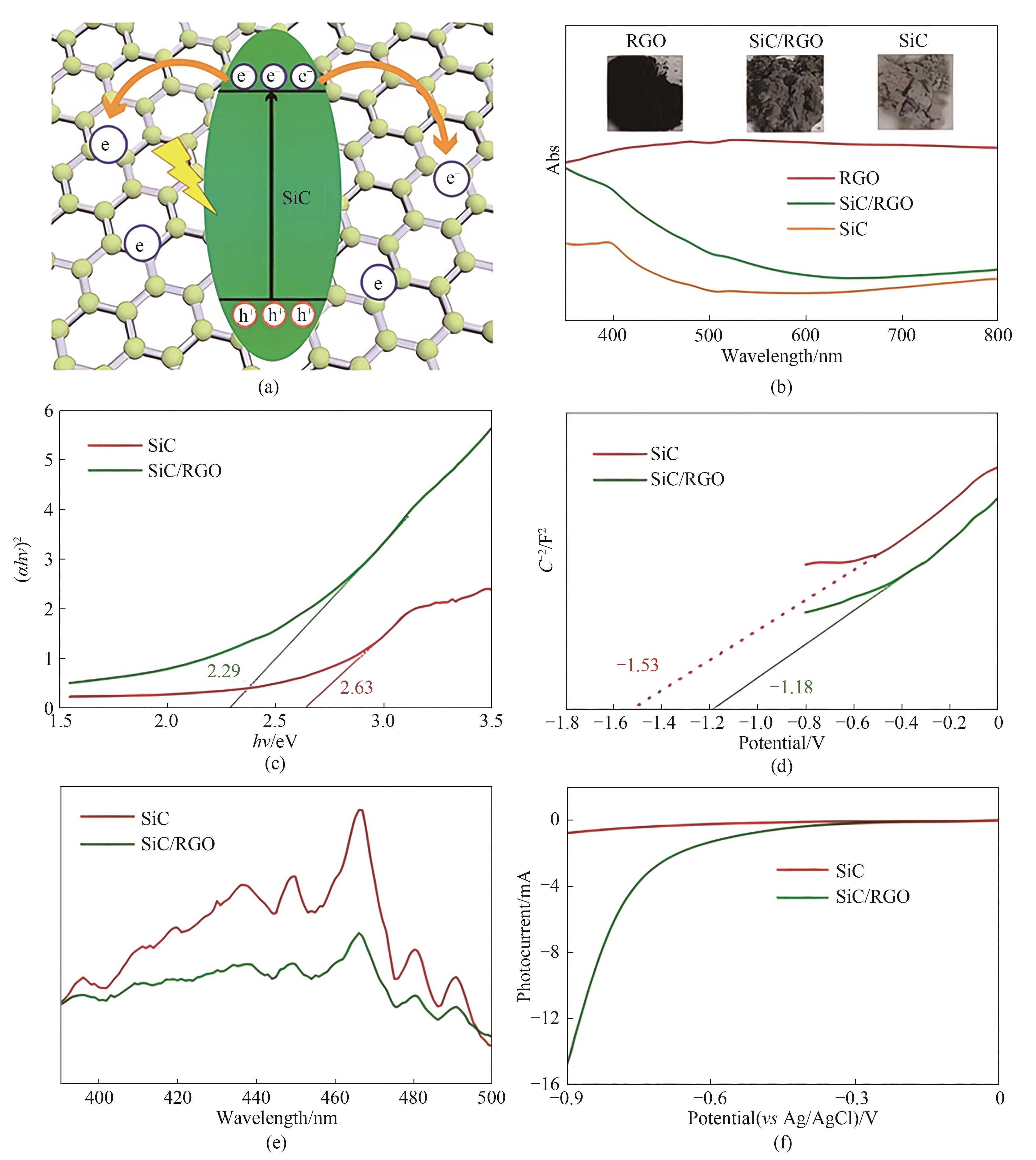
图9 SiC薄片和RGO的协同效应示意图(a); RGO、SiC/RGO和纯SiC的紫外可见漫反射光谱(b);Tauc图(c);Mott-Schottky图(d);室温PL光谱(e);SiC/RGO和SiC阴极在光照下的LSV曲线(f)[43]
Fig.9 Schematic diagram of the synergistic effect of SiC flakes and RGO (a); UV-Vis diffuse reflectance spectra of RGO, SiC/RGO and pure SiC (b); The Tauc plot (c); Mott-Schottky plots (d); Room-temperature PL spectra of SiC and SiC/RGO samples (e); LSV curves of SiC/RGO and SiC cathodes under illumination (f)[43]

图10 双场辅助Li-CO2电池的工作机制(a);首次循环的充/放电电压曲线(b);循环性能(c);采用TNAs和TNAs@AgNPs阴极的Li-CO2电池在光暗条件下的倍率能力(d);TNA@AgNPs正极在1.0 mAh·cm-2和2.0 mAh·cm-2下的充/放电曲线(e);双场辅助Li-CO2电池与一些基于电催化和光电催化机制的代表性Li-CO2电池的循环稳定性(f)和倍率性能(g)比较[39]
Fig.10 Working mechanism of the dual-field assisted Li-CO2 battery (a); Discharge/charge voltage profiles at the first cycle (b); Cycling performance (c); Rate capability of Li-CO2 batteries with TNAs and TNAs@AgNPs cathodes in light or dark (d); Discharge/charge curves of the TNAs@AgNPs cathode in light at 1.0 mAh·cm-2 and 2.0 mA·cm-2 (e); Comparison of cycling stability (f) and rate capability (g) of the dual-field assisted Li-CO2 battery with some representative Li-CO2 batteries based on the electrocatalysis and photo electrocatalysis mechanisms[39]
| 1 | Armand M, Tarascon J M. Building better batteries[J]. Nature, 2008, 451: 652-657. |
| 2 | Ding X B, Huang Q H, Xiong X H. Research and application of fast-charging graphite anodes for lithium-ion batteries[J]. Chinese Journal of Inorganic Chemistry, 2022, 38(11): 2204057. |
| 3 | Chen C, Zhang J M, Hu B R, et al. Dynamic gel as artificial interphase layer for ultrahigh-rate and large-capacity lithium metal anode[J]. Nature communications, 2023, 14(1): 4018. |
| 4 | Chiang Y M. Building a better battery[J]. MRS Bulletin, 2020, 45(3): 246-247. |
| 5 | Li M, Lu J, Chen Z W, et al. 30 years of lithium-ion batteries[J]. Advanced Materials, 2018: e1800561. |
| 6 | Ding X B, Huang H Y, Huang Q H, et al. Doping sites modulation of T-Nb2O5 to achieve ultrafast lithium storage[J]. Journal of Energy Chemistry, 2023, 77(2): 280-289. |
| 7 | Chen K, Yang D Y, Huang G, et al. Lithium-air batteries: air-electrochemistry and anode stabilization[J]. Accounts of Chemical Research, 2021, 54(3): 632-641. |
| 8 | Ohno S, Zeier W G. Toward practical solid-state lithium-sulfur batteries: challenges and perspectives[J]. Accounts of Materials Research, 2021, 2(10): 869-880. |
| 9 | Zhang L, Wang S, Wang Q, et al. Dendritic solid polymer electrolytes: a new paradigm for high-performance lithium-based batteries[J]. Advanced Materials, 2023, 35(35): e2303355. |
| 10 | Ma L, Yu T W, Tzoganakis E, et al. Fundamental understanding and material challenges in rechargeable nonaqueous Li-O2 batteries: recent progress and perspective[J]. Advanced Energy Materials, 2018, 8(22): 1800348. |
| 11 | Xiao X, Zhang Z J, Yan A J, et al. Upgrading carbon utilization and green energy storage through oxygen-assisted lithium-carbon dioxide batteries[J]. Energy Storage Materials, 2024, 65: 103129. |
| 12 | Chen L, Zhou J W, Wang Y H, et al. Flexible, stretchable, water-/fire-proof fiber-shaped Li-CO2 batteries with high energy density[J]. Advanced Energy Materials, 2023, 13(1): 2202933. |
| 13 | Zhao W T, Yang Y, Deng Q H, et al. Toward an understanding of bimetallic MXene solid-solution in binder-free electrocatalyst cathode for advanced Li-CO2 batteries[J]. Advanced Functional Materials, 2022, 33(5): 1. |
| 14 | Sun X Y, Mu X W, Zheng W, et al. Binuclear Cu complex catalysis enabling Li-CO2 battery with a high discharge voltage above 3.0 V[J]. Nature Communications, 2023, 14: 536. |
| 15 | Li X L, Zhang J X, Qi G C, et al. Vertically aligned N-doped carbon nanotubes arrays as efficient binder-free catalysts for flexible Li-CO2 batteries[J]. Energy Storage Materials, 2021, 35: 148-156. |
| 16 | Zhou L J, Wang H, Zhang K, et al. Fast decomposition of Li2CO3/C actuated by single-atom catalysts for Li-CO2 batteries[J]. Science China Materials, 2021, 64(9): 2139-2147. |
| 17 | Liu B, Sun Y L, Liu L Y, et al. Recent advances in understanding Li-CO2 electrochemistry[J]. Energy & Environmental Science, 2019, 12(3): 887-922. |
| 18 | Lin J F, Ding J N, Wang H Z, et al. Boosting energy efficiency and stability of Li-CO2 batteries via synergy between Ru atom clusters and single-atom Ru-N4 sites in the electrocatalyst cathode[J]. Advanced Materials, 2022, 34(17): e2200559. |
| 19 | Xiao Y, Hu S L, Miao Y, et al. Recent progress in hot spot regulated strategies for catalysts applied in Li-CO2 batteries[J]. Small, 2024, 20(1): e2305009. |
| 20 | Lu B Y, Min Z W, Xiao X, et al. Recycled tandem catalysts promising ultralow overpotential Li-CO2 batteries[J]. Advanced Materials, 2024, 36(1): e2309264. |
| 21 | Chourasia A K, Shavez M, Naik K M, et al. Candle soot nanoparticles versus multiwalled carbon nanotubes as a high-performance cathode catalyst for Li-CO2Mars batteries for Mars exploration[J]. ACS Applied Energy Materials, 2023, 6(1): 378-386. |
| 22 | Zhou J W, Cheng J L, Wang B, et al. Flexible metal-gas batteries: a potential option for next-generation power accessories for wearable electronics[J]. Energy & Environmental Science, 2020, 13(7): 1933-1970. |
| 23 | Gowda S R, Brunet A, Wallraff G M, et al. Implications of CO2 contamination in rechargeable nonaqueous Li-O2 batteries[J]. The Journal of Physical Chemistry Letters, 2013, 4(2): 276-279. |
| 24 | Zhao Z W, Huang J, Peng Z Q. Achilles’ heel of lithium-air batteries: lithium carbonate[J]. Angewandte Chemie International Edition, 2018, 57(15): 3874-3886. |
| 25 | Yang S X, He P, Zhou H S. Exploring the electrochemical reaction mechanism of carbonate oxidation in Li-air/CO2 battery through tracing missing oxygen[J]. Energy & Environmental Science, 2016, 9(5): 1650-1654. |
| 26 | Li J X, Zhang K, Wang B J, et al. Light-assisted metal-air batteries: progress, challenges, and perspectives[J]. Angewandte Chemie International Edition, 2022, 61(51): e202213026. |
| 27 | Savunthari K V, Chen C H, Chen Y R, et al. Effective Ru/CNT cathode for rechargeable solid-state Li-CO2 batteries[J]. ACS Applied Materials & Interfaces, 2021, 13(37): 44266-44273. |
| 28 | Guo C, Zhang F L, Han X, et al. Intrinsic descriptor guided noble metal cathode design for Li-CO2 battery[J]. Advanced Materials, 2023, 35(33): e2302325. |
| 29 | Wang Y F, Ji G J, Song L N, et al. A highly reversible lithium–carbon dioxide battery based on soluble oxalate[J]. ACS Energy Letters, 2023, 8(2): 1026-1034. |
| 30 | Fan L, Shen H M, Ji D X, et al. Biaxially compressive strain in Ni/Ru core/shell nanoplates boosts Li-CO2 batteries[J]. Advanced Materials, 2022, 34(30): e2204134. |
| 31 | Zhou J W, Wang T S, Chen L, et al. Boosting the reaction kinetics in aprotic lithium-carbon dioxide batteries with unconventional phase metal nanomaterials[J]. Proceedings of the National Academy of Sciences of the United States of America, 2022, 119(40): e2204666119. |
| 32 | Chen L, Zhou J W, Zhang J X, et al. Copper indium sulfide enables Li-CO2 batteries with boosted reaction kinetics and cycling stability[J]. Energy & Environmental Materials, 2023, 6(5): 12415. |
| 33 | Zhang Z, Yang C, Wu S S, et al. Exploiting synergistic effect by integrating ruthenium-copper nanoparticles highly co-dispersed on graphene as efficient air cathodes for Li-CO2 batteries[J]. Advanced Energy Materials, 2019, 9(8): 1802805. |
| 34 | Li J X, Zhang K, Zhao Y, et al. High-efficiency and stable Li-CO2 battery enabled by carbon nanotube/carbon nitride heterostructured photocathode[J]. Angewandte Chemie International Edition, 2022, 61(4): e202114612. |
| 35 | Wang J H, Li S, Chen Y F, et al. Phthalocyanine based metal-organic framework ultrathin nanosheet for efficient photocathode toward light-assisted Li-CO2 battery[J]. Advanced Functional Materials, 2022, 32(49): 2210259. |
| 36 | Wang X X, Guan D H, Li F, et al. A renewable light-promoted flexible LiCO2 battery with ultrahigh energy efficiency of 97.9%[J]. Small, 2021, 17(26): e2100642. |
| 37 | Zhu T, Wang S, Yu Z Q, et al. High-performance Li-CO2 battery based on carbon-free porous Ru@QNFs cathode[J]. Small, 2023, 19(33): e2301498. |
| 38 | Wang C Z, Shang Y, Lu Y C, et al. Photoinduced homogeneous RuO2 nanoparticles on TiO2 nanowire arrays: a high-performance cathode toward flexible Li-CO2 batteries[J]. Journal of Power Sources, 2020, 475: 228703. |
| 39 | Zhang K, Li J X, Zhai W J, et al. Boosting cycling stability and rate capability of Li-CO2 batteries via synergistic photoelectric effect and plasmonic interaction[J]. Angewandte Chemie International Edition, 2022, 61(17): e202201718. |
| 40 | Guan D H, Wang X X, Li M L, et al. Light/electricity energy conversion and storage for a hierarchical porous In2S3@CNT/SS cathode towards a flexible Li-CO2 battery[J]. Angewandte Chemie International Edition, 2020, 59(44): 19518-19524. |
| 41 | Ma Y, Wang X L, Jia Y S, et al. Titanium dioxide-based nanomaterials for photocatalytic fuel generations[J]. Chemical Reviews, 2014, 114(19): 9987-10043. |
| 42 | Jena A, Hsieh H C, Thoka S, et al. Curtailing the overpotential of Li-CO2 batteries with shape-controlled Cu2O as cathode: effect of illuminating the cathode[J]. ChemSusChem, 2020, 13(10): 2719-2725. |
| 43 | Li Z, Li M L, Wang X X, et al. In situ fabricated photo-electro-catalytic hybrid cathode for light-assisted lithium-CO2 batteries[J]. Journal of Materials Chemistry A, 2020, 8(29): 14799-14806. |
| 44 | Guan D H, Wang X X, Li F, et al. All-solid-state photo-assisted Li-CO2 battery working at an ultra-wide operation temperature[J]. ACS Nano, 2022, 16(8): 12364-12376. |
| 45 | Long L Z, Ding Y Y, Liang N N, et al. A carbon-free and free-standing cathode from mixed-phase TiO2 for photo-assisted Li-CO2 battery[J]. Small, 2023, 19(27): e2300519. |
| 46 | Zhao Z W, Wang E K, Wang J W, et al. Kinetics of the CO2 reduction reaction in aprotic Li–CO2 batteries: a model study[J]. Journal of Materials Chemistry A, 2021, 9(6): 3290-3296. |
| 47 | Yang C, Guo K K, Yuan D W, et al. Unraveling reaction mechanisms of Mo2C as cathode catalyst in a Li-CO2 battery[J]. Journal of the American Chemical Society, 2020, 142(15): 6983-6990. |
| 48 | Xu S M, Das S K, Archer L A. The Li–CO2 battery: a novel method for CO2 capture and utilization[J]. RSC Advances, 2013, 3(18): 6656-6660. |
| 49 | Wang S, Song H C, Zhu T, et al. An ultralow-charge-overpotential and long-cycle-life solid-state Li-CO2 battery enabled by plasmon-enhanced solar photothermal catalysis[J]. Nano Energy, 2022, 100: 107521. |
| 50 | Pipes R, Bhargav A, Manthiram A. Nanostructured anatase titania as a cathode catalyst for Li-CO2 batteries[J]. ACS Applied Materials & Interfaces, 2018, 10(43): 37119-37124. |
| 51 | Xu Y Y, Gong H, Ren H, et al. Highly efficient Cu-porphyrin-based metal-organic framework nanosheet as cathode for high-rate Li-CO2 battery[J]. Small, 2022, 18(45): e2203917. |
| 52 | Gong H, Wang T, Xue H R, et al. Photo-enhanced lithium oxygen batteries with defective titanium oxide as both photo-anode and air electrode[J]. Energy Storage Materials, 2018, 13: 49-56. |
| 53 | Baek K, Jeon W C, Woo S, et al. Synergistic effect of quinary molten salts and ruthenium catalyst for high-power-density lithium-carbon dioxide cell[J]. Nature Communications, 2020, 11: 456. |
| 54 | Ahmadiparidari A, Warburton R E, Majidi L, et al. A long-cycle-life lithium-CO2 battery with carbon neutrality[J]. Advanced Materials, 2019, 31(40): 1902518. |
| 55 | Guo Q, Zhou C Y, Ma Z B, et al. Fundamentals of TiO2 photocatalysis: concepts, mechanisms, and challenges[J]. Advanced Materials, 2019, 31(50): e1901997. |
| 56 | Hu J Y, Su C B, Li R J, et al. High-performance Li-CO2 batteries enabled by synergistic interaction of iron dopant-modulated catalysts and nitrogen-modified substrates[J]. Journal of Alloys and Compounds, 2024, 976: 173146. |
| 57 | Liu J, Ma N K, Wu W, et al. Recent progress on photocatalytic heterostructures with full solar spectral responses[J]. Chemical Engineering Journal, 2020, 393: 124719. |
| 58 | Low J, Yu J G, Jaroniec M, et al. Heterojunction photocatalysts[J]. Advanced Materials, 2017, 29(20): 1601694. |
| 59 | Jin Y C, Liu Y, Song L, et al. Interfacial engineering in hollow NiS2/FeS2-NSGA heterostructures with efficient catalytic activity for advanced Li-CO2 battery[J]. Chemical Engineering Journal, 2022, 430: 133029. |
| 60 | Sun Z M, Wang D, Lin L, et al. Ultrathin hexagonal boron nitride as a van der Waals’ force initiator activated graphene for engineering efficient non-metal electrocatalysts of Li-CO2 battery[J]. Nano Research, 2022, 15(2): 1171-1177. |
| 61 | Cheng Z B, Wu Z Y, Tang Y Y, et al. Cationic metal-organic framework derived ruthenium-copper nano-alloys in porous carbon to catalytically boost the cycle life of Li-CO2 batteries[J]. Nanoscale, 2022, 14(40): 15073-15078. |
| 62 | Han J R, Wu H Y, Song R L, et al. Defect-rich porous carbon as a metal-free catalyst for high-performance Li-CO2 batteries[J]. Electrochimica Acta, 2024, 477: 143779. |
| 63 | Qie L, Lin Y, Connell J W, et al. Highly rechargeable lithium-CO2 batteries with a boron- and nitrogen-codoped holey-graphene cathode[J]. Angewandte Chemie International Edition, 2017, 56(24): 6970-6974. |
| 64 | 王禹婷, 杨天怡, 章应辉. 卟啉框架材料在光催化领域的应用[J]. 应用化学, 2020, 37(6): 611-619. |
| Wang Y T, Yang T Y, Zhang Y H. Application of porphyrin-based framework materials on photocatalysis[J]. Chinese Journal of Applied Chemistry, 2020, 37(6): 611-619. | |
| 65 | 焦帅, 杨磊, 武婷婷, 等. 混合盐模板法制备超级电容器用氮掺杂分级多孔碳纳米片[J]. 化工学报, 2021, 72(5): 2869-2877. |
| Jiao S, Yang L, Wu T T, et al. Synthesis of nitrogen doped hierarchically porous carbon nanosheets for supercapacitor by mixed salt template[J]. CIESC Journal, 2021, 72(5): 2869-2877. | |
| 66 | 后振中, 彭龙贵, 李颖, 等. 分级多孔聚吡咯膜的界面自组装合成与电化学电容性[J]. 化工学报, 2018, 69(9): 4121-4128. |
| Hou Z Z, Peng L G, Li Y, et al. Interfacial self-assembly synthesis and electrochemical capacitance of hierarchical porous polypyrrole films[J]. CIESC Journal, 2018, 69(9): 4121-4128. | |
| 67 | Hu C, Tu S C, Tian N, et al. Photocatalysis enhanced by external fields[J]. Angewandte Chemie International Edition, 2021, 60(30): 16309-16328. |
| 68 | Sun Z H, Yin H, Liu K L, et al. Machine learning accelerated calculation and design of electrocatalysts for CO2 reduction[J]. SmartMat, 2022, 3(1): 68-83. |
| 69 | Liu J W, Luo W Z, Wang L, et al. Toward excellence of electrocatalyst design by emerging descriptor-oriented machine learning[J]. Advanced Functional Materials, 2022, 32(17): 2110748. |
| 70 | Németh K, Srajer G. CO2/oxalate cathodes as safe and efficient alternatives in high energy density metal–air type rechargeable batteries[J]. RSC Advances, 2014, 4(4): 1879-1885. |
| 71 | Qiao Y, Yi J, Wu S C, et al. Li-CO2 electrochemistry: a new strategy for CO2 fixation and energy storage[J]. Joule, 2017, 1(2): 359-370. |
| 72 | Su L W, Zhou Z, Qin X, et al. CoCO3 submicrocube/graphene composites with high lithium storage capability[J]. Nano Energy, 2013, 2(2): 276-282. |
| [1] | 咸国义, 陈立芳, 漆志文. 基于DFT的环己酮肟液相贝克曼重排机理研究[J]. 化工学报, 2024, 75(1): 302-311. |
| [2] | 叶凯, 刘香华, 姜月, 于颖, 赵亚飞, 庄烨, 郑进保, 陈秉辉. 低温等离子体协同CeO2/13X催化降解甲苯[J]. 化工学报, 2021, 72(7): 3706-3715. |
| [3] | 朱倩倩, 靳海波, 郭晓燕, 何广湘, 马磊, 张荣月, 谷庆阳, 杨索和. H2O2/乙腈体系下MgO催化环己酮Baeyer-Villiger绿色氧化合成ε-己内酯的研究[J]. 化工学报, 2021, 72(5): 2638-2646. |
| [4] | 王琴, 徐会金, 韩兴超, 赵长颖. MgO/Mg(OH)2热化学储热反应的第一性原理研究[J]. 化工学报, 2021, 72(3): 1242-1252. |
| [5] | 王刚,段学志,袁渭康,周兴贵. 钛硅分子筛TS-1催化环氧丙烷异构反应的机理探究[J]. 化工学报, 2021, 72(10): 5150-5158. |
| [6] | 张红, 唐留. p型掺杂剂Cp2Mg在MOCVD气相中的反应机理研究[J]. 化工学报, 2020, 71(7): 3000-3008. |
| [7] | 金燕, 杨倩, 赵文斌, 胡宝山. 石墨烯化学气相沉积法可控制备的催化反应体系研究[J]. 化工学报, 2020, 71(6): 2564-2585. |
| [8] | 王鲁丰, 钱鑫, 邓丽芳, 袁浩然. 氮气电化学合成氨催化剂研究进展[J]. 化工学报, 2019, 70(8): 2854-2863. |
| [9] | 侯莲霞, 袁兆平, 乔鸿昌, 周静红, 周兴贵. Ni-W2C催化葡萄糖氢解制备低碳二元醇反应机理研究[J]. 化工学报, 2019, 70(4): 1390-1400. |
| [10] | 梁天水, 王宗莹, 高坤, 李润婉, 王铮, 钟委, 赵军. 基于cup burner的含铁基添加剂超细水雾灭火有效性分析[J]. 化工学报, 2019, 70(3): 1236-1242. |
| [11] | 张弋, 李建波, 王泉海, 卢啸风. 新型双流化床炉内NOx生成特性数值模拟[J]. 化工学报, 2018, 69(4): 1703-1713. |
| [12] | 赵海谦, 高杏存, 刘城昊, 王忠华, 周伟, 高继慧. 基于Russell机理分光光度法检测液相体系·OH浓度的改进[J]. 化工学报, 2017, 68(7): 2805-2811. |
| [13] | 李术艳, 孙莉娜, 沈淑君, 程天行, 程双华, 陈久喜. 二芳基二硫醚与硝基芳烃的反应[J]. 化工学报, 2017, 68(6): 2394-2398. |
| [14] | 张红梅, 林枫, 任铭琪, 李金莲, 郝玉兰, 吴红军, 赵晶莹, 赵亮, 贺永殿. 小分子烃类蒸汽热裂解自由基机理模型研究方法的探讨[J]. 化工学报, 2017, 68(4): 1423-1433. |
| [15] | 张光洁, 耿艳楼, 安华良, 赵新强, 王延吉. Zn(OAc)2/AC催化固定床上CO2与1,2-丙二醇合成碳酸丙烯酯及其反应机理[J]. 化工学报, 2015, 66(2): 567-575. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备 11010102001995号
京公网安备 11010102001995号