化工学报 ›› 2024, Vol. 75 ›› Issue (10): 3793-3803.DOI: 10.11949/0438-1157.20240423
收稿日期:2024-04-16
修回日期:2024-07-24
出版日期:2024-10-25
发布日期:2024-11-04
通讯作者:
殷俞
作者简介:李文宁(1999—),女,硕士研究生,lwn15703878253@163.com
基金资助:
Wenning LI1( ), Min LU1, Yu YIN2(
), Min LU1, Yu YIN2( )
)
Received:2024-04-16
Revised:2024-07-24
Online:2024-10-25
Published:2024-11-04
Contact:
Yu YIN
摘要:
在废水处理技术中,基于过氧单硫酸盐(PMS)的高级氧化工艺被认为是降解有机污染物中最高效且绿色的方法之一。在激活PMS的方法中,负载过渡金属的催化剂由于金属离子溶出量低而广受关注,但却面临金属物种易团聚引发的降解活性低的问题。将Co负载在还原氧化石墨烯(rGO)上,经简单方法分别合成了Co高分散的QSCo-rGO和Co团聚的AGCo-rGO。XRD、HRTEM和元素分布研究表明,QSCo-rGO中的Co未发生金属团聚现象,实现了高度分散;而AGCo-rGO中的Co以CoO纳米颗粒的形式团聚。在激活PMS降解苯酚的反应中,QSCo-rGO可在30 min内完成100%的降解,而AGCo-rGO需要90 min。此外,QSCo-rGO展现出较好的可重复性和使用广泛性。机理研究表明,催化剂QSCo-rGO中的Co高度分散,暴露出更多活性金属位点,其通过激活PMS产生非自由基1O2从而高活性降解苯酚。本文可为设计增强降解活性的高级氧化催化剂提供思路。
中图分类号:
李文宁, 陆敏, 殷俞. 钴高度分散于还原氧化石墨烯用于高级氧化降解有机污染物[J]. 化工学报, 2024, 75(10): 3793-3803.
Wenning LI, Min LU, Yu YIN. High dispersion of cobalt on the reduced graphene oxide for advanced oxidation degradation of organic pollutants[J]. CIESC Journal, 2024, 75(10): 3793-3803.
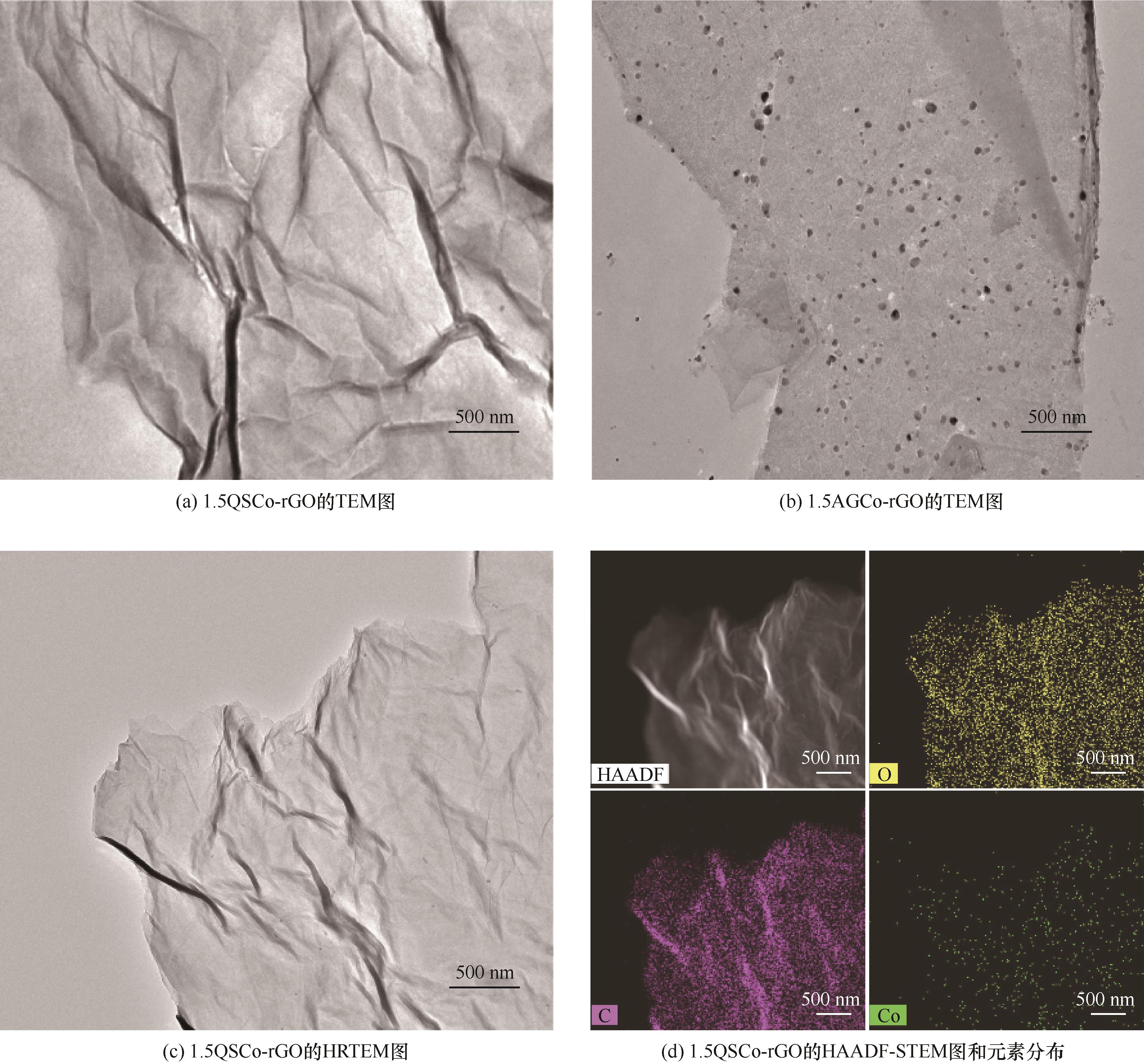
图1 1.5QSCo-rGO、1.5AGCo-rGO的TEM图;1.5QSCo-rGO的HRTEM和HAADF-STEM图像以及对应元素O、C、Co的分布
Fig.1 TEM images of 1.5QSCo-rGO, and 1.5AGCo-rGO; HRTEM and HAADF-STEM images and element mapping of corresponding O, C and Co elements of 1.5QSCo-rGO
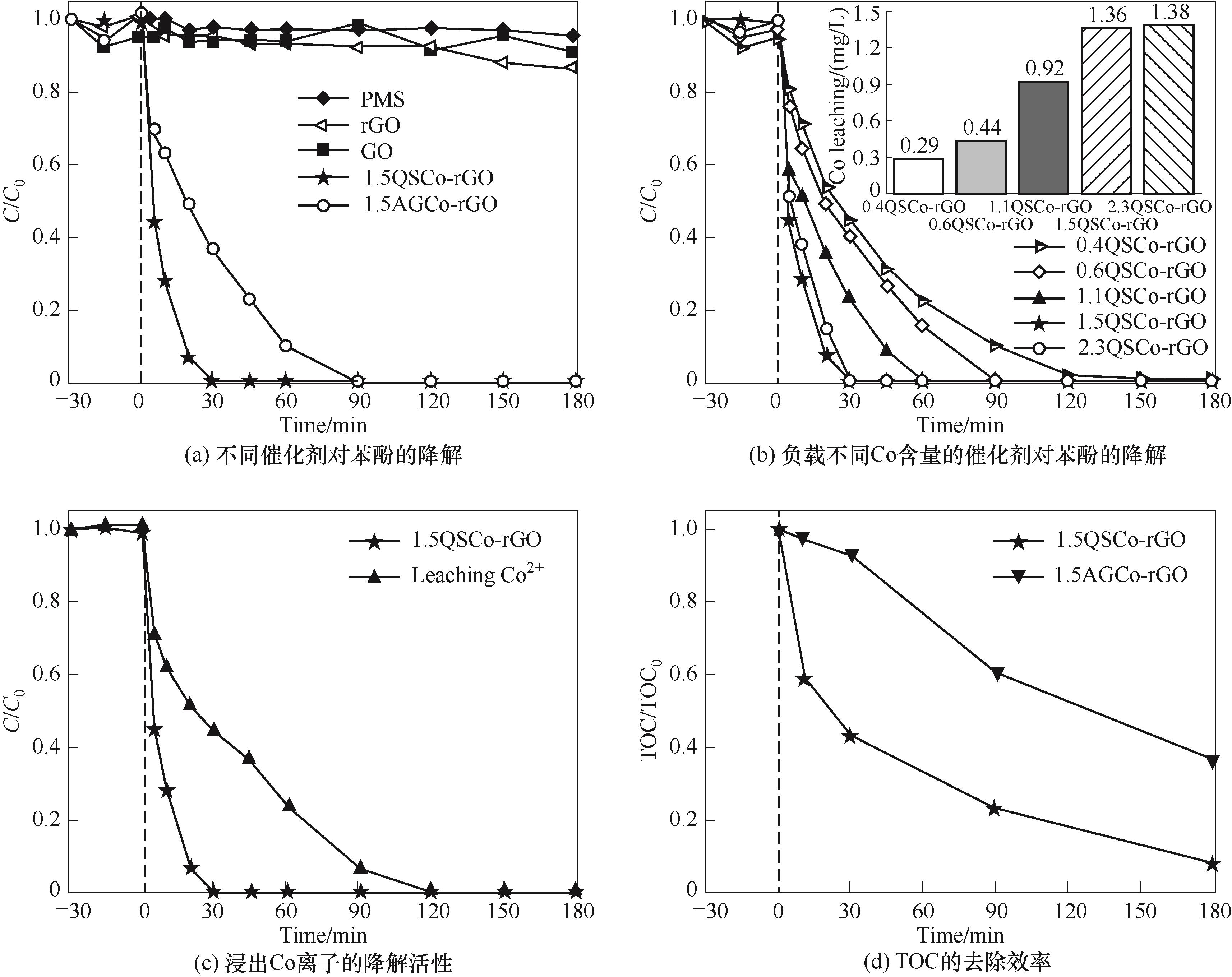
图4 不同催化剂对苯酚的降解作用和浸出Co离子的降解活性,以及TOC的去除效率
Fig.4 Degradation of phenol by different catalysts, degradation activity of leaching Co ions and removal efficiency of TOC
| 催化剂 | 反应条件 | 降解率 | 速率常数k/min-1 | 文献 | |||
|---|---|---|---|---|---|---|---|
| 温度/℃ | 苯酚浓度/(mg/L) | 催化剂浓度/(g/L) | 氧化剂(PMS) 浓度/(mmol/L) | ||||
| 1.5QSCo-rGO | 25 | 20 | 0.2 | 6.5 | 100%(30 min) | 0.131 | 本文 |
| Co/CoO@NC-1% | 25 | 20 | 0.1 | 1.63 | 100%(20 min) | 0.324 | [ |
| CoNP@NC/Co-SA | 25 | 20 | 0.08 | 0.49 | 91.6%(3 min) | 0.696 | [ |
| Co@NG-900 | 25 | 9.41 | 0.05 | 3 | 100%(12 min) | 0.397 | [ |
| Se@NC-900 | 25 | 10 | 0.1 | 0.25 | 99.1%(30 min) | 0.169 | [ |
| CFGA | 25 | 20 | 0.2 | 3.25 | 100%(60 min) | 0.046 | [ |
| MnOOH-rGO | 30 | 23.5 | 0.5 | 0.625 | 100%(30 min) | not provided | [ |
| CNS6 | 25 | 10 | 0.05 | 3 | 100%(60 min) | 0.089 | [ |
| CoOOH/GO | 25 | 10 | 0.2 | 0.15 | 41%(5 min) | not provided | [ |
| Co-30/KCC-1 | 25 | 20 | 0.2 | 7.8 | 100%(9 min) | not provided | [ |
| Co2MnO4 | 25 | 50 | 0.2 | 6.5 | 100%(30 min) | 0.076 | [ |
| 10%Co3O4/CeO2 | 20 | 20 | 0.2 | 6.5 | 100%(50 min) | 0.0865 | [ |
| CoMgAl-LDH | 30 | 9.41 | 0.3 | 3 | 100%(60 min) | 0.051 | [ |
| 2.5%MnO x /GO | 25 | 75 | 0.4 | 6.5 | 28%(30 min) | not provided | [ |
表1 已报道催化材料对苯酚降解的催化性能比较
Table 1 Comparison of catalytic properties of reported catalytic materials for phenol degradation
| 催化剂 | 反应条件 | 降解率 | 速率常数k/min-1 | 文献 | |||
|---|---|---|---|---|---|---|---|
| 温度/℃ | 苯酚浓度/(mg/L) | 催化剂浓度/(g/L) | 氧化剂(PMS) 浓度/(mmol/L) | ||||
| 1.5QSCo-rGO | 25 | 20 | 0.2 | 6.5 | 100%(30 min) | 0.131 | 本文 |
| Co/CoO@NC-1% | 25 | 20 | 0.1 | 1.63 | 100%(20 min) | 0.324 | [ |
| CoNP@NC/Co-SA | 25 | 20 | 0.08 | 0.49 | 91.6%(3 min) | 0.696 | [ |
| Co@NG-900 | 25 | 9.41 | 0.05 | 3 | 100%(12 min) | 0.397 | [ |
| Se@NC-900 | 25 | 10 | 0.1 | 0.25 | 99.1%(30 min) | 0.169 | [ |
| CFGA | 25 | 20 | 0.2 | 3.25 | 100%(60 min) | 0.046 | [ |
| MnOOH-rGO | 30 | 23.5 | 0.5 | 0.625 | 100%(30 min) | not provided | [ |
| CNS6 | 25 | 10 | 0.05 | 3 | 100%(60 min) | 0.089 | [ |
| CoOOH/GO | 25 | 10 | 0.2 | 0.15 | 41%(5 min) | not provided | [ |
| Co-30/KCC-1 | 25 | 20 | 0.2 | 7.8 | 100%(9 min) | not provided | [ |
| Co2MnO4 | 25 | 50 | 0.2 | 6.5 | 100%(30 min) | 0.076 | [ |
| 10%Co3O4/CeO2 | 20 | 20 | 0.2 | 6.5 | 100%(50 min) | 0.0865 | [ |
| CoMgAl-LDH | 30 | 9.41 | 0.3 | 3 | 100%(60 min) | 0.051 | [ |
| 2.5%MnO x /GO | 25 | 75 | 0.4 | 6.5 | 28%(30 min) | not provided | [ |
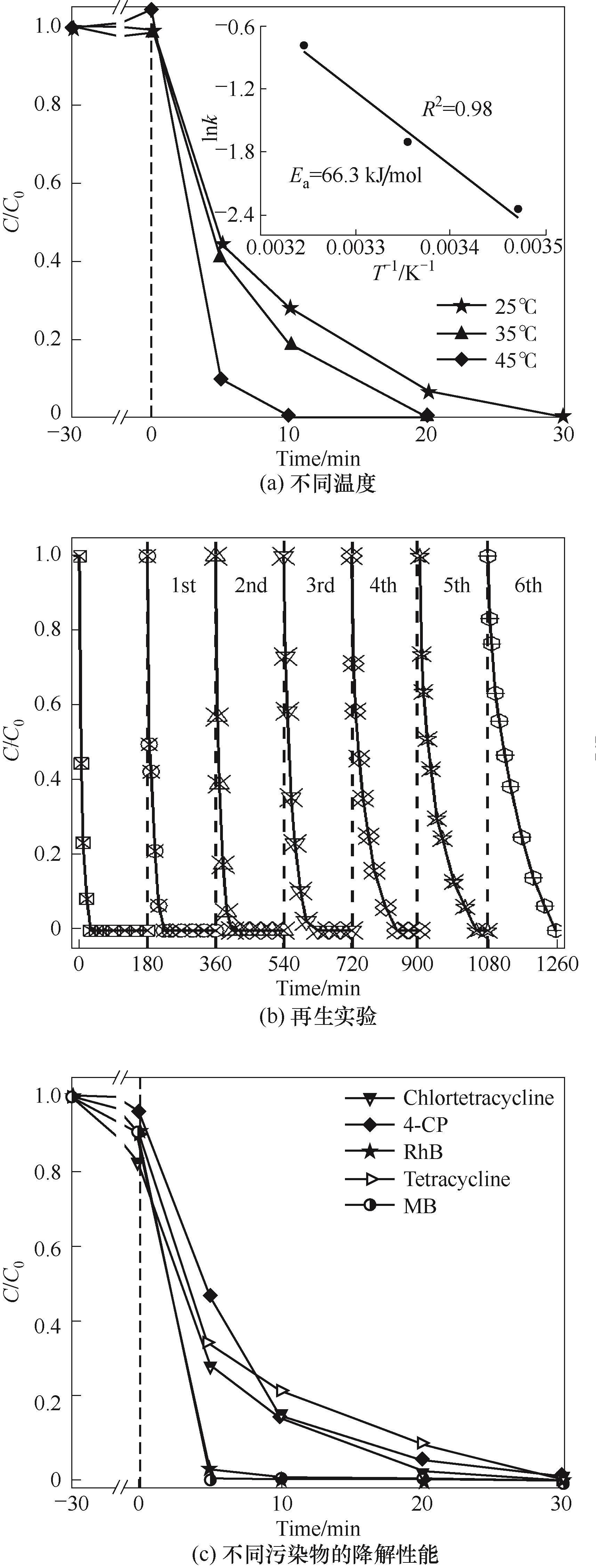
图5 不同温度对催化体系1.5QSCo-rGO/PMS的降解活性影响,以及稳定性与普适性
Fig.5 The effects of different temperatures on the degradation activity of catalytic system 1.5QSCo-rGO/PMS, and the stability and adaptability
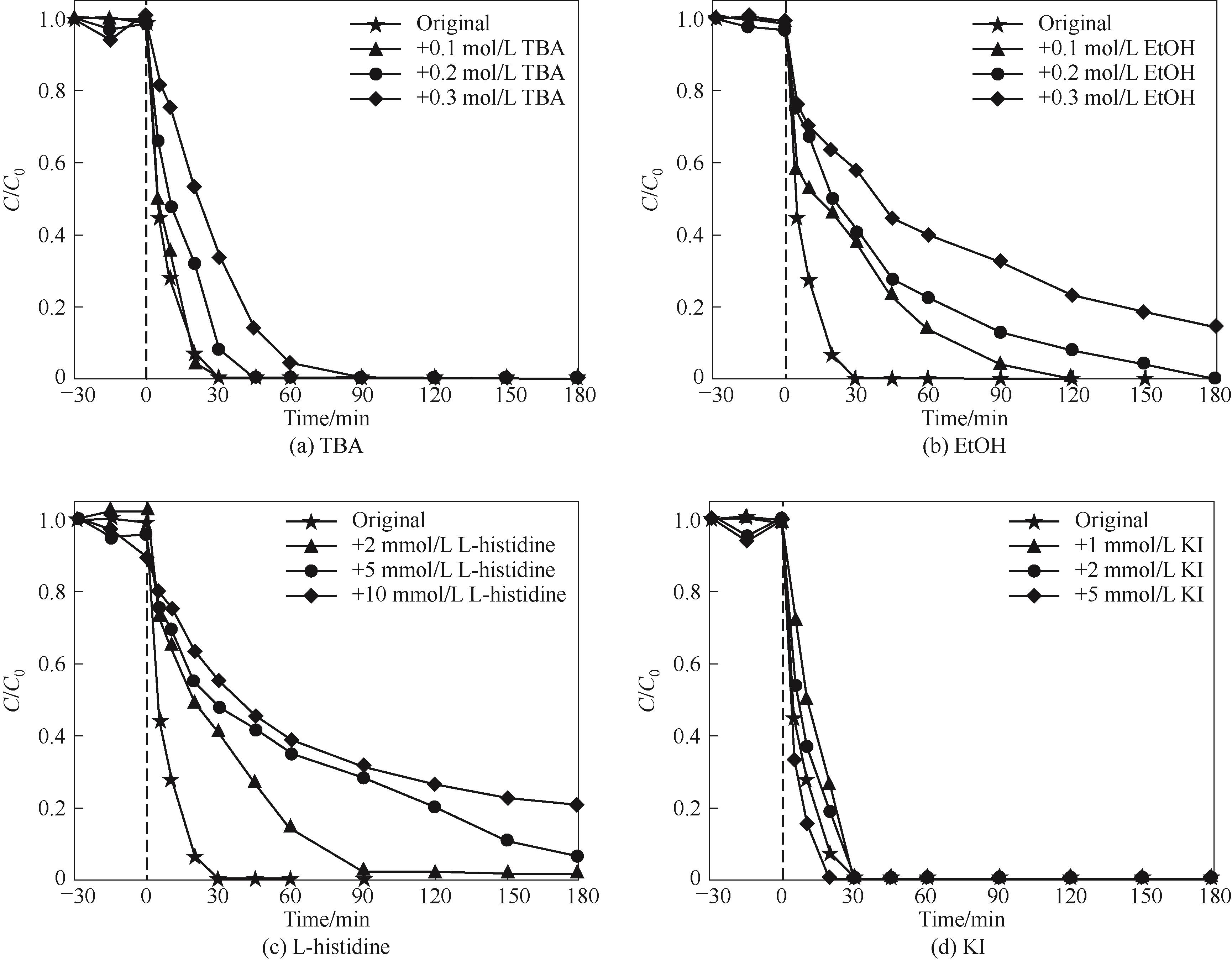
图6 不同用量的猝灭剂TBA、EtOH、L-histidine和KI对降解苯酚的抑制效果
Fig.6 Inhibitory effects of different dosages of quenching agents TBA, EtOH, L-histidine, and KI on the degradation of phenol
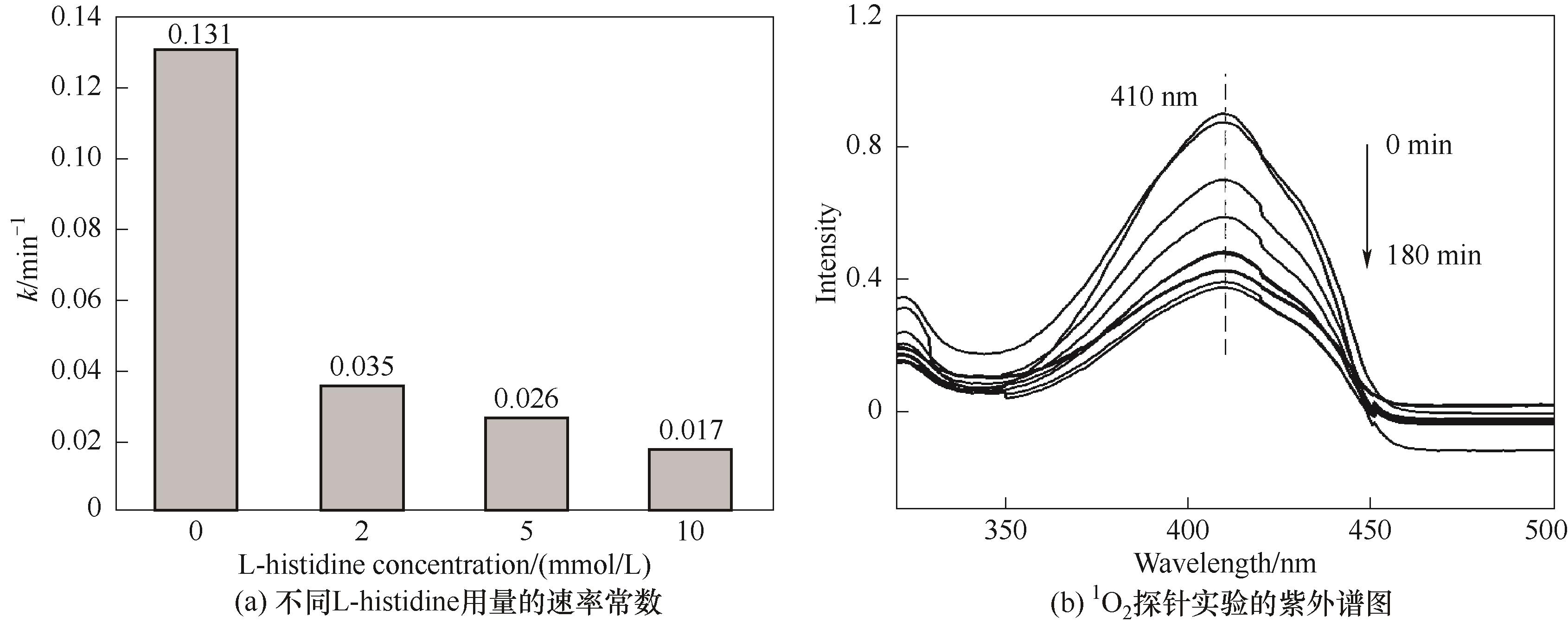
图7 不同猝灭剂L-histidine用量的速率常数k的变化,以及活性物种1O2的探针实验
Fig.7 Change of rate constant k with different dosage of quenching agent L-histidine, and probe experiments with active species 1O2
| 1 | Li X T, Wang J, Duan X G, et al. Fine-tuning radical/nonradical pathways on graphene by porous engineering and doping strategies[J]. ACS Catalysis, 2021, 11(8): 4848-4861. |
| 2 | Fan L S, Fujie K, Long T R, et al. Characteristics of draft tube gas-liquid-solid fluidized-bed bioreactor with immobilized living cells for phenol degradation[J]. Biotechnology and Bioengineering, 1987, 30(4): 498-504. |
| 3 | Farhan Hanafi M, Sapawe N. A review on the water problem associate with organic pollutants derived from phenol, methyl orange, and remazol brilliant blue dyes[J]. Materials Today: Proceedings, 2020, 31: A141-A150. |
| 4 | Zhao T T, Gao Y H, Yu T T, et al. Biodegradation of phenol by a highly tolerant strain Rhodococcus ruber C1: biochemical characterization and comparative genome analysis[J]. Ecotoxicology and Environmental Safety, 2021, 208: 111709. |
| 5 | You Y Y, He Z. Phenol degradation in iron-based advanced oxidation processes through ferric reduction assisted by molybdenum disulfide[J]. Chemosphere, 2023, 312: 137278. |
| 6 | Wang Y S, Qiu W, Lu X H, et al. Nitrilotriacetic acid-assisted Mn(Ⅱ) activated periodate for rapid and long-lasting degradation of carbamazepine: the importance of Mn(Ⅳ)-oxo species[J]. Water Research, 2023, 241: 120156. |
| 7 | Li Y Y, Zhang S G, Qin Y N, et al. Preparation of cobalt/hydrochar using the intrinsic features of rice hulls for dynamic carbamazepine degradation via efficient PMS activation[J]. Journal of Environmental Chemical Engineering, 2022, 10(6): 108659. |
| 8 | Li Z L, Zhang L, Wang L, et al. Engineering the electronic structure of two-dimensional MoS2 by Ni dopants for pollutant degradation[J]. Separation and Purification Technology, 2023, 314: 123637. |
| 9 | Zhang Q H, He D, Li X R, et al. Mechanism and performance of singlet oxygen dominated peroxymonosulfate activation on CoOOH nanoparticles for 2,4-dichlorophenol degradation in water[J]. Journal of Hazardous Materials, 2020, 384: 121350. |
| 10 | Wu Z L, Wang Y P, Xiong Z K, et al. Core-shell magnetic Fe3O4@Zn/Co-ZIFs to activate peroxymonosulfate for highly efficient degradation of carbamazepine[J]. Applied Catalysis B: Environmental, 2020, 277: 119136. |
| 11 | Yao Y J, Tao Z M, Hu H W, et al. In situ growth of iron incorporated Ni3S2 nanosheet on nickel foam in mediating electron transfer to peroxymonosulfate for pollutant abatement[J]. Journal of Environmental Sciences, 2025, 150: 704-718. |
| 12 | Yi Q Y, Tan J L, Liu W Y, et al. Peroxymonosulfate activation by three-dimensional cobalt hydroxide/graphene oxide hydrogel for wastewater treatment through an automated process[J]. Chemical Engineering Journal, 2020, 400: 125965. |
| 13 | Chen H Y, Lu J, Fedeyko J M, et al. Zeolite supported Pd catalysts for the complete oxidation of methane: a critical review[J]. Applied Catalysis A: General, 2022, 633: 118534. |
| 14 | Shen P F, Yin P, Zou Y T, et al. Ultra-fast piezocatalysts enabled by interfacial interaction of reduced graphene oxide/MoS2 heterostructures[J]. Advanced Materials, 2023, 35(18): e2212172. |
| 15 | 王永胜, 兰小林, 邱天, 等. 铜基石墨烯复合催化剂的合成与表征[J]. 化工学报, 2020, 71(6): 2889-2899. |
| Wang Y S, Lan X L, Qiu T, et al. Synthesis and characterization of copper-based graphene composite catalyst[J]. CIESC Journal, 2020, 71(6): 2889-2899. | |
| 16 | Shahzad A, Jawad A, Ifthikar J, et al. The hetero-assembly of reduced graphene oxide and hydroxide nanosheets as superlattice materials in PMS activation[J]. Carbon, 2019, 155: 740-755. |
| 17 | Shen C C, Wang Y, Fu J. Urchin-like Co3O4 anchored on reduced graphene oxide with enhanced performance for peroxymonosulfate activation in ibuprofen degradation[J]. Journal of Environmental Management, 2022, 307: 114572. |
| 18 | Zou L J, Xiao X, Chu C H, et al. Facile synthesis of porous CoFe2O4/graphene aerogel for catalyzing efficient removal of organic pollutants[J]. Science of the Total Environment, 2021, 775: 143398. |
| 19 | Liu X Y, Yu H R, Ji J H, et al. Graphene oxide-supported three-dimensional cobalt-nickel bimetallic sponge-mediated peroxymonosulfate activation for phenol degradation[J]. ACS ES&T Engineering, 2021, 1(12): 1705-1714. |
| 20 | Hummers W S, Offeman R E. Preparation of graphitic oxide[J]. Journal of the American Chemical Society, 1958, 80(6): 1339. |
| 21 | Chen Z L, Yang L X, Liu X T, et al. Enhanced oxygen activation over self-supporting Cu2+ doped Co3O4 nanoneedle arrays for efficient HCHO oxidation at room temperature[J]. Separation and Purification Technology, 2024, 338: 126542. |
| 22 | Singh M, T P R, Golda A S, et al. Selective vanadium etching and in-situ formation of δ-Bi2O3 on m-BiVO4 with g-C3N4 nanosheets for photocatalytic degradation of antibiotic tetracycline[J]. Journal of Cleaner Production, 2024, 442: 140921. |
| 23 | Rad T S, Khataee A, Rahim Pouran S. Synergistic enhancement in photocatalytic performance of Ce(Ⅳ) and Cr(Ⅲ) co-substituted magnetite nanoparticles loaded on reduced graphene oxide sheets[J]. Journal of Colloid and Interface Science, 2018, 528: 248-262. |
| 24 | Zhang W, Li M, Luo J W, et al. Modulating the coordination environment of Co single-atom catalysts through sulphur doping to efficiently enhance peroxymonosulfate activation for degradation of carbamazepine[J]. Chemical Engineering Journal, 2023, 474: 145377. |
| 25 | Zou L J, Zhu X Y, Lu L, et al. Bimetal organic framework/graphene oxide derived magnetic porous composite catalyst for peroxymonosulfate activation in fast organic pollutant degradation[J]. Journal of Hazardous Materials, 2021, 419: 126427. |
| 26 | Zou H Y, Wang H T, Sun H Q, et al. Single-atom cobalt catalysts encapsulating cobalt nanoparticles with built-in electric field for ultrafast and lasting peroxymonosulfate activation[J]. ACS ES&T Water, 2024, 4(6): 2433-2444. |
| 27 | Yu X Y, Wang L J, Wang X, et al. Enhanced nonradical catalytic oxidation by encapsulating cobalt into nitrogen doped graphene: highlight on interfacial interactions[J]. Journal of Materials Chemistry A, 2021, 9(11): 7198-7207. |
| 28 | Chai Y D, Dai H L, Zhan P, et al. Selective degradation of organic micropollutants by activation of peroxymonosulfate by Se@NC: role of Se doping and nonradical pathway mechanism[J]. Journal of Hazardous Materials, 2023, 452: 131202. |
| 29 | Zhao H X, Cao Y, Liu Y Q, et al. Efficient degradation of phenol by MnOOH-rGO composite with high peroxymonosulfate utilization efficiency[J]. Chemosphere, 2023, 336: 139200. |
| 30 | Ren S Y, Xu X, Zhu Z S, et al. Catalytic transformation of microplastics to functional carbon for catalytic peroxymonosulfate activation: conversion mechanism and defect of scavenging[J]. Applied Catalysis B: Environmental, 2024, 342: 123410. |
| 31 | Afzal S, Pan K, Duan D D, et al. Heterogeneous activation of peroxymonosulfate with cobalt incorporated fibrous silica nanospheres for the degradation of organic pollutants in water[J]. Applied Surface Science, 2021, 542: 148674. |
| 32 | Yue L J, Hao L Y, Zhang J K, et al. Oxygen-enriched vacancy Co2MnO4 spinel catalyst activated peroxymonosulfate for degradation of phenol: non-radical dominated reaction pathway[J]. Journal of Water Process Engineering, 2023, 53: 103807. |
| 33 | Gao Q, Cui Y C, Wang S J, et al. Efficient activation of peroxymonosulfate by Co-doped mesoporous CeO2 nanorods as a heterogeneous catalyst for phenol oxidation[J]. Environmental Science and Pollution Research, 2021, 28(22): 27852-27863. |
| 34 | Liao Z W, Zhu J Y, Jawad A, et al. Degradation of phenol using peroxymonosulfate activated by a high efficiency and stable CoMgAl-LDH catalyst[J]. Materials, 2019, 12(6): 968. |
| 35 | Saputra E, Pinem J A, Budihardjo M A, et al. Carbon-supported manganese for heterogeneous activation of peroxymonosulfate for the decomposition of phenol in aqueous solutions[J]. Materials Today Chemistry, 2020, 16: 100268. |
| 36 | Yao Y J, Cai Y M, Lu F, et al. Magnetic recoverable MnFe2O4 and MnFe2O4-graphene hybrid as heterogeneous catalysts of peroxymonosulfate activation for efficient degradation of aqueous organic pollutants[J]. Journal of Hazardous Materials, 2014, 270: 61-70. |
| 37 | Qian J, Mi X H, Chen Z J, et al. Efficient emerging contaminants (EM) decomposition via peroxymonosulfate (PMS) activation by Co3O4/carbonized polyaniline (CPANI) composite: characterization of tetracycline (TC) degradation property and application for the remediation of EM-polluted water body[J]. Journal of Cleaner Production, 2023, 405: 137023. |
| 38 | Zhang J, Ma Y L, Sun Y G, et al. Reduced porous 2D Co3O4 enhanced peroxymonosulfate activation to form multi-reactive oxygen species: the key role of oxygen vacancies[J]. Separation and Purification Technology, 2024, 330: 125409. |
| 39 | 岳敏, 王璟, 韩玉泽, 等. 盐助溶液燃烧法制备MnFe2O4催化过一硫酸盐降解双酚A[J]. 化工学报, 2020, 71(12): 5589-5598. |
| Yue M, Wang J, Han Y Z, et al. Degradation of bisphenol A by peroxymonosulfate activated by MnFe2O4 prepared by salt-assisted solution combustion synthesis[J]. CIESC Journal, 2020, 71(12): 5589-5598. | |
| 40 | Yang L, Jiao Y, Xu X M, et al. Superstructures with atomic-level arranged perovskite and oxide layers for advanced oxidation with an enhanced non-free radical pathway[J]. ACS Sustainable Chemistry & Engineering, 2022, 10(5): 1899-1909. |
| 41 | 闫新龙, 黄志刚, 胡清勋, 等. Cu/Co掺杂多孔炭活化过硫酸盐降解水中硝基酚研究[J]. 化工学报, 2023, 74(3): 1102-1112. |
| Yan X L, Huang Z G, Hu Q X, et al. Catalytic nitrophenol degradation via peroxymonosulfate activation over Cu/Co doped porous carbon[J]. CIESC Journal, 2023, 74(3): 1102-1112. |
| [1] | 张佳颖, 王聪, 王雅君. CNT-Co/Bi2O3催化剂光催化协同过硫酸盐活化高效降解四环素[J]. 化工学报, 2024, 75(9): 3163-3175. |
| [2] | 王冉, 王焕, 熊晓云, 关慧敏, 郑云锋, 陈彩琳, 秦玉才, 宋丽娟. FCC催化剂传质强化活性位利用效率的可视化分析[J]. 化工学报, 2024, 75(9): 3198-3209. |
| [3] | 王树振, 王玉婷, 马梦茜, 张巍, 向江南, 鲁海莹, 王琰, 范彬彬, 郑家军, 代卫炯, 李瑞丰. 两步晶化合成ZSM-22分子筛及其临氢异构反应性能[J]. 化工学报, 2024, 75(9): 3176-3187. |
| [4] | 刘亚超, 谭晓杰, 李旭东, 王瑞, 王慧, 韩璇, 赵青山. DES合成高活性CoCO3纳米片及析氧反应性能研究[J]. 化工学报, 2024, 75(9): 3320-3328. |
| [5] | 胡术刚, 田国庆, 刘文娟, 徐广飞, 刘华清, 张建, 王艳龙. 纳米零价铁的制备及氧化还原技术的应用进展[J]. 化工学报, 2024, 75(9): 3041-3055. |
| [6] | 张梦婷, 王书林, 桑熙, 元兴昊, 徐刚. 人工Cu-TM1459金属酶催化不对称迈克尔加成反应[J]. 化工学报, 2024, 75(9): 3255-3265. |
| [7] | 刘旭升, 李泽洋, 杨宇森, 卫敏. 电催化二氧化碳还原制备气态产物的研究进展[J]. 化工学报, 2024, 75(7): 2385-2408. |
| [8] | 罗莉, 陈文尧, 张晶, 钱刚, 周兴贵, 段学志. 氧化铝结构与表面性质调控及其催化甲醇脱水制二甲醚性能研究[J]. 化工学报, 2024, 75(7): 2522-2532. |
| [9] | 王寅, 初鹏飞, 刘虎, 吕静, 黄守莹, 王胜平, 马新宾. 不同pH铝溶胶对二甲醚羰基化成型丝光沸石催化剂性能的影响[J]. 化工学报, 2024, 75(7): 2533-2543. |
| [10] | 杨露, 刘聪聪, 孟彤彤, 张博远, 杨腾飞, 邓文安, 王晓斌. 分散型催化剂在煤/重油共炼体系中的加氢抑焦作用[J]. 化工学报, 2024, 75(7): 2556-2564. |
| [11] | 王天闻, 闫肃, 赵梦园, 杨天让, 刘建国. 固体氧化物电池空气电极铬中毒机理及抗铬性能研究进展[J]. 化工学报, 2024, 75(6): 2091-2108. |
| [12] | 丁禹, 杨昌泽, 李军, 孙会东, 商辉. 原子尺度钼系加氢脱硫催化剂的研究进展与展望[J]. 化工学报, 2024, 75(5): 1735-1749. |
| [13] | 赵亭亭, 鄢立祥, 唐福利, 肖敏之, 谭烨, 宋刘斌, 肖忠良, 李灵均. 光辅助锂-二氧化碳电池催化剂的设计策略与反应机理研究进展[J]. 化工学报, 2024, 75(5): 1750-1764. |
| [14] | 莫锦洪, 韩雪, 朱毅翔, 李菁, 王旭裕, 纪红兵. Pt-Ga/CeO2-ZrO2-Al2O3脱氢裂解双功能催化剂用于正丁烷催化制烯烃研究[J]. 化工学报, 2024, 75(5): 1855-1869. |
| [15] | 张林, 张子怡, 李勇, 童少平. Fe-MOF-74前体制备铁-碳/氮复合材料及其活化过硫酸盐性能[J]. 化工学报, 2024, 75(5): 1882-1889. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备 11010102001995号
京公网安备 11010102001995号