化工学报 ›› 2024, Vol. 75 ›› Issue (11): 4274-4285.DOI: 10.11949/0438-1157.20240583
马烨玮1( ), 孙艳娜1, 高栋2, 王海彬2, 姚善泾1, 林东强1(
), 孙艳娜1, 高栋2, 王海彬2, 姚善泾1, 林东强1( )
)
收稿日期:2024-05-30
修回日期:2024-07-15
出版日期:2024-11-25
发布日期:2024-12-26
通讯作者:
林东强
作者简介:马烨玮(1999—),女,硕士研究生,mayewei2022@163.com
基金资助:
Yewei MA1( ), Yanna SUN1, Dong GAO2, Haibin WANG2, Shanjing YAO1, Dongqiang LIN1(
), Yanna SUN1, Dong GAO2, Haibin WANG2, Shanjing YAO1, Dongqiang LIN1( )
)
Received:2024-05-30
Revised:2024-07-15
Online:2024-11-25
Published:2024-12-26
Contact:
Dongqiang LIN
摘要:
连续流层析具有提高过程产率和介质利用率、降低生产成本等显著优势,在抗体药物生产中具有良好的应用前景。但是,连续流层析模式多样,影响因素众多,传统的基于实验的过程开发方法存在困难。将模型辅助方法引入到连续流层析亲和捕获过程,建立了模型辅助过程优化方法,系统比较了两柱、三柱和四柱连续捕获模式,确定了最佳模式和操作条件,并经实验验证。结果表明:模型预测与实验结果基本一致;与批次层析相比,四柱连续捕获的过程产率提高了27.2%,介质利用率提高了50.1%,且产品质量稳定。由此说明,模型辅助方法有助于确定最佳连续捕获模式和操作条件,促进过程优化,加速抗体药物连续生产过程的工业实现。
中图分类号:
马烨玮, 孙艳娜, 高栋, 王海彬, 姚善泾, 林东强. 模型辅助的单抗连续捕获工艺分析和过程优化[J]. 化工学报, 2024, 75(11): 4274-4285.
Yewei MA, Yanna SUN, Dong GAO, Haibin WANG, Shanjing YAO, Dongqiang LIN. Model-assisted process evaluation and optimization of continuous chromatography for antibody capture[J]. CIESC Journal, 2024, 75(11): 4274-4285.
| 步骤 | 溶液 | 保留时间/min | 体积/CV | 时间/min |
|---|---|---|---|---|
| 冲洗 | 0.025 mol/L Tris + 0.025 mol/L NaCl缓冲液(pH 7.7) | 6 | 4 | 24 |
| 淋洗 | 0.5 mol/L磷酸盐缓冲液(pH 6.0) | 6 | 5 | 30 |
| 平衡 | 0.025 mol/L Tris + 0.025 mol/L NaCl缓冲液(pH 7.7) | 6 | 3 | 18 |
| 洗脱 | 0.15 mol/L醋酸缓冲液(pH 2.8) | 6 | 4 | 24 |
| 再生 | 0.1 mol/L NaOH溶液 | 6 | 5 | 30 |
| 再平衡 | 0.025 mol/L Tris + 0.025 mol/L NaCl缓冲液(pH 7.7) | 6 | 3 | 18 |
表1 洗脱和再生过程的工艺参数
Table 1 Operation parameters of recovery and regeneration process units
| 步骤 | 溶液 | 保留时间/min | 体积/CV | 时间/min |
|---|---|---|---|---|
| 冲洗 | 0.025 mol/L Tris + 0.025 mol/L NaCl缓冲液(pH 7.7) | 6 | 4 | 24 |
| 淋洗 | 0.5 mol/L磷酸盐缓冲液(pH 6.0) | 6 | 5 | 30 |
| 平衡 | 0.025 mol/L Tris + 0.025 mol/L NaCl缓冲液(pH 7.7) | 6 | 3 | 18 |
| 洗脱 | 0.15 mol/L醋酸缓冲液(pH 2.8) | 6 | 4 | 24 |
| 再生 | 0.1 mol/L NaOH溶液 | 6 | 5 | 30 |
| 再平衡 | 0.025 mol/L Tris + 0.025 mol/L NaCl缓冲液(pH 7.7) | 6 | 3 | 18 |
| 模型参数 | 数值 |
|---|---|
| ε | 0.38 |
| εp | 0.52 |
| Qmax/(g/L) | 130 |
| Kd/(g/L) | 0.08 |
| Dax/(10-7 m2/s) | 30 |
| kf/(10-6 m/s) | 20 |
| Ds/(10-14 m2/s) | 0.5 |
| Dp/(10-12 m2/s) | 4.9 |
表2 蛋白A亲和层析模型参数汇总
Table 2 Model parameters for protein A affinity chromatography
| 模型参数 | 数值 |
|---|---|
| ε | 0.38 |
| εp | 0.52 |
| Qmax/(g/L) | 130 |
| Kd/(g/L) | 0.08 |
| Dax/(10-7 m2/s) | 30 |
| kf/(10-6 m/s) | 20 |
| Ds/(10-14 m2/s) | 0.5 |
| Dp/(10-12 m2/s) | 4.9 |
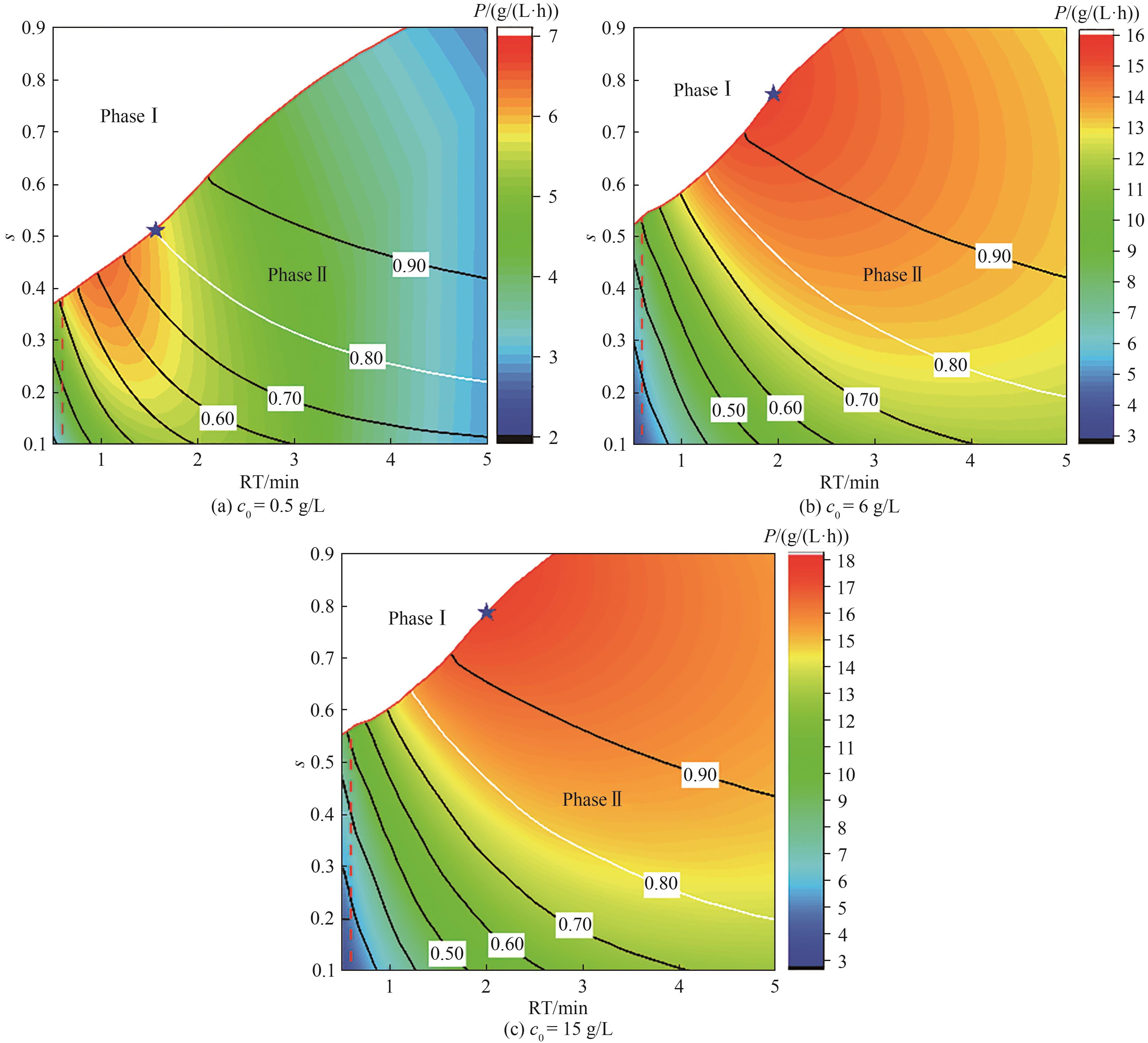
图3 CaptureSMB连续捕获过程性能比较[彩色等高线图为过程产率变化,黑色实线为介质利用率变化,红色虚线为上样保留时间(对应流速)上限,蓝色星点为最优操作点]
Fig.3 Process performance of CaptureSMB continuous capture(Color contour maps show the changes of productivity; Black-line contour maps show the changes of capacity utilization; Red dash lines represent the upper flow rate of the resin; Star points are the optimal operation points)
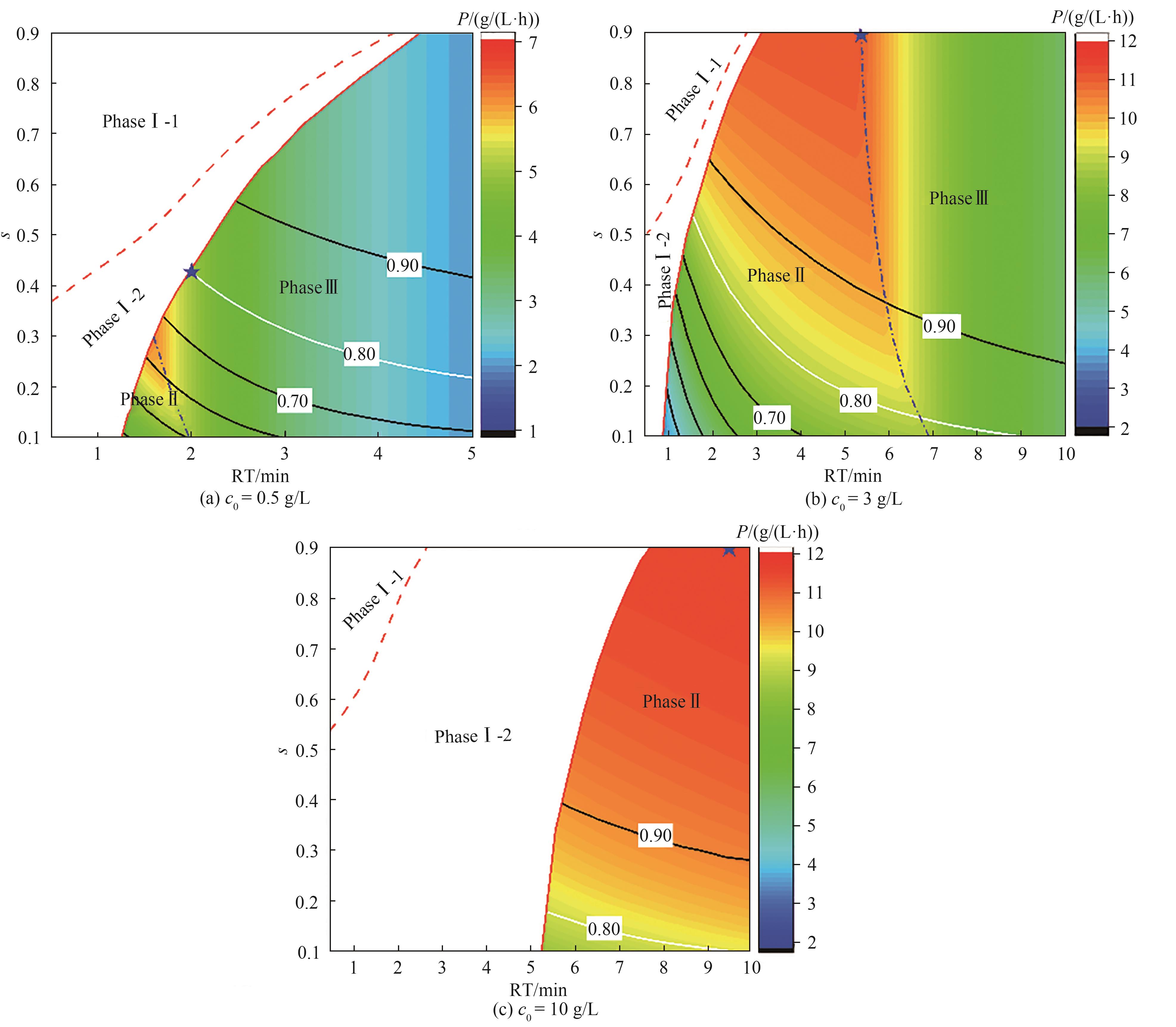
图4 3C-PCC连续捕获过程性能比较(彩色等高线图为过程产率变化,黑色实线为介质利用率变化,红色虚线阶段Ⅰ-1和阶段Ⅰ-2分界线,蓝色虚线为阶段Ⅱ和阶段Ⅲ分界线,蓝色星点为最优操作点)
Fig.4 Process performance of 3C-PCC continuous capture(Color contour maps show the changes of productivity; Black-line contour maps show the changes of capacity utilization; Red dash line represents the boundary of phase Ⅰ-1 and phase Ⅰ-2; Blue dash line represents the boundary of phase Ⅱ and phase Ⅲ; Star points are the optimal operation points)
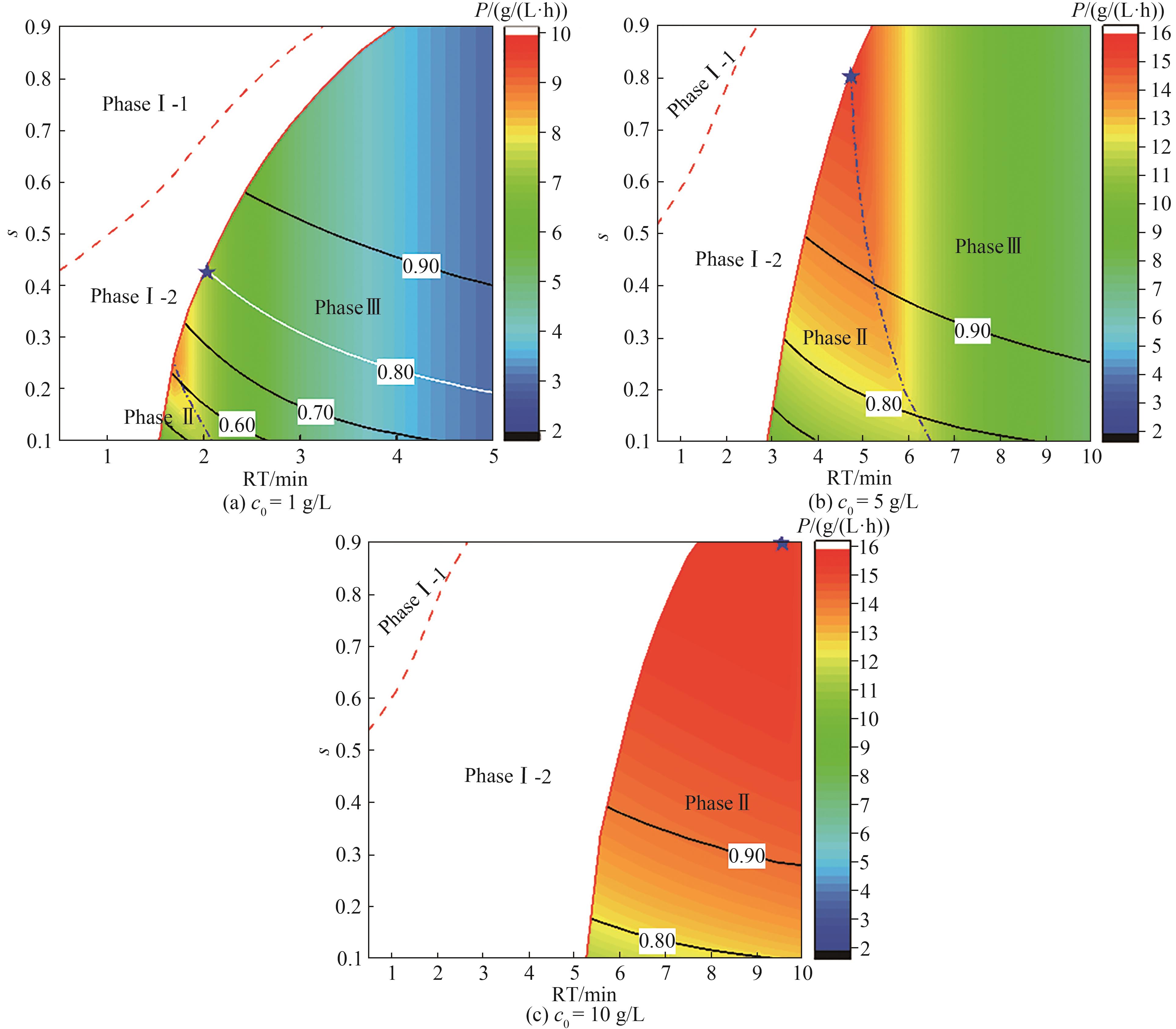
图5 4C-PCC连续捕获过程性能比较(彩色等高线图为过程产率变化,黑色实线为介质利用率变化,红色虚线阶段Ⅰ-1和阶段Ⅰ-2分界线,蓝色虚线为阶段Ⅱ和阶段Ⅲ分界线,蓝色星点为最优操作点)
Fig.5 Process performance of 4C-PCC continuous capture(Color contour maps show the changes of productivity; Black-line contour maps show the changes of capacity utilization; Red dash line represents the boundary of phase Ⅰ-1 and phase Ⅰ-2; Blue dash line represents the boundary of phase Ⅱ and phase Ⅲ; Star points are the optimal operation points)
| 项目 | 批次层析 | 连续捕获 |
|---|---|---|
| 纯度/% | 97.3 | 99.3 |
| 收率/% | 92.2 | 91.1 |
| 聚集体/% | 2.2 | 0.7 |
| HCP LRV | 2.33 | 2.57 |
| hcDNA LRV | 4.04 | 2.36 |
| 过程产率/(g/(L·h)) | 11.17 | 14.21 |
| 介质利用率/% | 59.9 | 89.9 |
| 缓冲液消耗/(L/g) | 0.55 | 0.35 |
表3 批次与连续的分离效果比较
Table 3 Comparison of batch and continuous separation performance
| 项目 | 批次层析 | 连续捕获 |
|---|---|---|
| 纯度/% | 97.3 | 99.3 |
| 收率/% | 92.2 | 91.1 |
| 聚集体/% | 2.2 | 0.7 |
| HCP LRV | 2.33 | 2.57 |
| hcDNA LRV | 4.04 | 2.36 |
| 过程产率/(g/(L·h)) | 11.17 | 14.21 |
| 介质利用率/% | 59.9 | 89.9 |
| 缓冲液消耗/(L/g) | 0.55 | 0.35 |
| 1 | David L, Schwan P, Lobedann M, et al. Side-by-side comparability of batch and continuous downstream for the production of monoclonal antibodies[J]. Biotechnology and Bioengineering, 2020, 117(4): 1024-1036. |
| 2 | Mahal H, Branton H, Farid S S. End-to-end continuous bioprocessing: impact on facility design, cost of goods, and cost of development for monoclonal antibodies[J]. Biotechnology and Bioengineering, 2021, 118(9): 3468-3485. |
| 3 | Rathore A S, Thakur G, Kateja N. Continuous integrated manufacturing for biopharmaceuticals: a new paradigm or an empty promise?[J]. Biotechnology and Bioengineering, 2023, 120(2): 333-351. |
| 4 | 鲁伟, 应国清, 杨晓明, 等. 单克隆抗体工业生产中蛋白A亲和层析步骤的成本分析[J]. 高校化学工程学报, 2023, 37(2): 276-284. |
| Lu W, Ying G Q, Yang X M, et al. Cost analysis of protein A affinity chromatography in industrial production of monoclonal antibody[J]. Journal of Chemical Engineering of Chinese Universities, 2023, 37(2): 276-284. | |
| 5 | FDA. Quality considerations for continuous manufacturing[EB/OL]. [2024-05-30]. . |
| 6 | ICH. Continuous manufacturing of drug substances and drug products Q13[EB/OL]. [2024-05-30]. . |
| 7 | 史策, 虞骥, 高栋, 等. 单抗制备的过程模拟和经济性分析[J]. 化工学报, 2018, 69(7): 3198-3207. |
| Shi C, Yu J, Gao D, et al. Process simulation and economic evaluation of monoclonal antibody production[J]. CIESC Journal, 2018, 69(7): 3198-3207. | |
| 8 | Bielser J M, Wolf M, Souquet J, et al. Perfusion mammalian cell culture for recombinant protein manufacturing — a critical review[J]. Biotechnology Advances, 2018, 36(4): 1328-1340. |
| 9 | Matanguihan C, Wu P. Upstream continuous processing: recent advances in production of biopharmaceuticals and challenges in manufacturing[J]. Current Opinion in Biotechnology, 2022, 78: 102828. |
| 10 | 高宗晔, 史策, 姚善泾, 等. 双柱连续流层析亲和分离抗体的过程设计与应用[J]. 高校化学工程学报, 2019, 33(1): 117-127. |
| Gao Z Y, Shi C, Yao S J, et al. Process design and application of twin-column continuous chromatography for antibody affinity separation[J]. Journal of Chemical Engineering of Chinese Universities, 2019, 33(1): 117-127. | |
| 11 | Nicoud R M. The amazing ability of continuous chromatography to adapt to a moving environment[J]. Industrial & Engineering Chemistry Research, 2014, 53(10): 3755-3765. |
| 12 | 荆淑莹, 史策, 姚善泾, 等. 连续流层析及用于抗体分离的新进展[J]. 高校化学工程学报, 2021, 35(1): 1-12. |
| Jing S Y, Shi C, Yao S J, et al. Progress on continuous chromatography and its application in antibody separation[J]. Journal of Chemical Engineering of Chinese Universities, 2021, 35(1): 1-12. | |
| 13 | ChromaCon. Contichrom CUBE-versatile benchtop process development tool[EB/OL]. [2024-05-30]. . |
| 14 | Cytica. ÄKTA pcc and BioProcess pcc continuous chromatography systems[EB/OL]. [2024-05-30]. . |
| 15 | Sartorius. Continuous Chromatography-BIOSMB PD and BIOSMBProcess[EB/OL]. [2024-05-30]. . |
| 16 | Novasep. BioSC continuous chromatography for biologics[EB/OL]. [2024-05-30]. . |
| 17 | Semba. Octave 10 chromatography system[EB/OL]. [2024-05-30]. . |
| 18 | Mahajan E, George A, Wolk B. Improving affinity chromatography resin efficiency using semi-continuous chromatography[J]. Journal of Chromatography A, 2012, 1227: 154-162. |
| 19 | Angarita M, Müller-Späth T, Baur D, et al. Twin-column CaptureSMB: a novel cyclic process for protein A affinity chromatography[J]. Journal of Chromatography A, 2015, 1389: 85-95. |
| 20 | Gao Z Y, Zhang Q L, Shi C, et al. Antibody capture with twin-column continuous chromatography: effects of residence time, protein concentration and resin[J]. Separation and Purification Technology, 2020, 253: 117554. |
| 21 | Lin D Q, Zhang Q L, Yao S J. Model-assisted approaches for continuous chromatography: current situation and challenges[J]. Journal of Chromatography A, 2021, 1637: 461855. |
| 22 | Shi C, Chen X J, Jiao B, et al. Model-assisted process design for better evaluation and scaling up of continuous downstream bioprocessing[J]. Journal of Chromatography A, 2022, 1683: 463532. |
| 23 | Narayanan H, Luna M, Sokolov M, et al. Hybrid models based on machine learning and an increasing degree of process knowledge: application to capture chromatographic step[J]. Industrial & Engineering Chemistry Research, 2021, 60(29): 10466-10478. |
| 24 | Chopda V, Gyorgypal A, Yang O, et al. Recent advances in integrated process analytical techniques, modeling, and control strategies to enable continuous biomanufacturing of monoclonal antibodies[J]. Journal of Chemical Technology & Biotechnology, 2022, 97(9): 2317-2335. |
| 25 | Guo J, Jin M, Kanani D. Optimization of single-column batch and multicolumn continuous protein A chromatography and performance comparison based on mechanistic model[J]. Biotechnology Journal, 2020, 15(10): e2000192. |
| 26 | Shi C, Zhang Q L, Jiao B, et al. Process development and optimization of continuous capture with three-column periodic counter-current chromatography[J]. Biotechnology and Bioengineering, 2021, 118(9): 3313-3322. |
| 27 | Shi C, Gao Z Y, Zhang Q L, et al. Model-based process development of continuous chromatography for antibody capture: a case study with twin-column system[J]. Journal of Chromatography A, 2020, 1619: 460936. |
| 28 | Sun Y N, Shi C, Zhang Q L, et al. Model-based process development and evaluation of twin-column continuous capture processes with protein A affinity resin[J]. Journal of Chromatography A, 2020, 1625: 461300. |
| 29 | Sun Y N, Shi C, Zhang Q L, et al. Comparison of protein A affinity resins for twin-column continuous capture processes: process performance and resin characteristics[J]. Journal of Chromatography A, 2021, 1654: 462454. |
| 30 | Sun Y N, Shi C, Zhong X Z, et al. Model-based evaluation and model-free strategy for process development of three-column periodic counter-current chromatography[J]. Journal of Chromatography A, 2022, 1677: 463311. |
| 31 | 史策. 模型辅助的连续流层析过程开发和抗体分离应用研究[D]. 杭州: 浙江大学, 2021. |
| Shi C. Model-assisted process development of continuous chromatography and its applications for antibody separation[D]. Hangzhou: Zhejiang University, 2021. |
| [1] | 向昕辰, 鲁丹, 赵影, 姚之侃, 寇瑞强, 郑丹军, 周志军, 张林. 聚酰胺纳滤膜表面季铵化提高荷正电性及其锂镁分离性能[J]. 化工学报, 2025, (): 1-10. |
| [2] | 袁梦星, 孙琳, 罗雄麟. 多效蒸发海水淡化系统变量相关性分析与全周期操作优化[J]. 化工学报, 2024, (): 1-15. |
| [3] | 袁玲雅, 张滢. 中国光伏产业发展及其资源环境影响[J]. 化工学报, 2024, 75(S1): 14-24. |
| [4] | 花敬贤, 罗宇荣, 顾亚伟, 吴婷婷, 潘宜昌, 邢卫红. 超薄取向ZIF-8膜的制备及乙烯/乙烷高效分离[J]. 化工学报, 2024, (): 1-14. |
| [5] | 高冰冰, 许诺, 白云翔, 张春芳, 杨永强, 董亮亮. 氦气分离聚合物膜[J]. 化工学报, 2024, (): 1-17. |
| [6] | 胡嘉朗, 姜明源, 金律铭, 张永刚, 胡鹏, 纪红兵. 机器学习辅助MOFs高通量计算筛选及气体分离研究进展[J]. 化工学报, 2024, (): 1-24. |
| [7] | 李季, 王佳才, 马永强, 袁海斌, 简路明, 姜基灿, 朱家骅. 湿法磷酸含氟尾气高效资源化利用热力学分析与工程实践[J]. 化工学报, 2024, (): 1-9. |
| [8] | 赵静, 刘公平, 金万勤, 徐南平. 限域传质分离膜的精密构筑与应用[J]. 化工学报, 2024, 75(11): 3857-3869. |
| [9] | 顾天宇, 陈献富, 王思琪, 徐鹏, 邱鸣慧, 范益群. 膜技术在杜仲有效成分分离纯化中的应用研究进展[J]. 化工学报, 2024, 75(11): 4005-4019. |
| [10] | 董正亚, 朱晓晶, 贾竞夫, 张杰, 郑卓韬, 刘晓霖, 武志林. 超声微反应器系统的放大及其在纳米材料制备中的应用[J]. 化工学报, 2024, 75(11): 4095-4119. |
| [11] | 王茂先, 孙启典, 付哲, 华放, 纪晔, 程易. 分子水平动力学模型和机器学习方法相结合研究废弃塑料热解[J]. 化工学报, 2024, 75(11): 4320-4332. |
| [12] | 冯咪, 张杰, 吕兴梅. 基于胆碱类离子液体的高纯甲壳素一步提取分离[J]. 化工学报, 2024, 75(11): 4286-4297. |
| [13] | 唐文娜, 刘宏臣, 米晓天, 秋列维, 杨梅, 陈光文. 嵌入式筛孔微通道内液滴生成规律研究[J]. 化工学报, 2024, 75(11): 4188-4195. |
| [14] | 阮见, 李双, 温正慧. 自动化与智能化在流动化学中的应用[J]. 化工学报, 2024, 75(11): 4120-4140. |
| [15] | 王璐遥, 张广勇, 于海鑫, 张轩诚, 黄岩, 赵玉潮. 聚全氟乙丙烯中空纤维复合膜的制备及其染料/无机盐分离性能研究[J]. 化工学报, 2024, 75(11): 4309-4319. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备 11010102001995号
京公网安备 11010102001995号