化工学报 ›› 2024, Vol. 75 ›› Issue (9): 3152-3162.DOI: 10.11949/0438-1157.20240106
胡德政1,2( ), 王榕1, 王世栋1, 杨文菲1, 张宏伟1, 袁珮1,2(
), 王榕1, 王世栋1, 杨文菲1, 张宏伟1, 袁珮1,2( )
)
收稿日期:2024-01-23
修回日期:2024-04-24
出版日期:2024-09-25
发布日期:2024-10-10
通讯作者:
袁珮
作者简介:胡德政(1998—),男,硕士研究生,853491951@qq.com
基金资助:
Dezheng HU1,2( ), Rong WANG1, Shidong WANG1, Wenfei YANG1, Hongwei ZHANG1, Pei YUAN1,2(
), Rong WANG1, Shidong WANG1, Wenfei YANG1, Hongwei ZHANG1, Pei YUAN1,2( )
)
Received:2024-01-23
Revised:2024-04-24
Online:2024-09-25
Published:2024-10-10
Contact:
Pei YUAN
摘要:
C9石油树脂(C9PR)是由乙烯裂解副产物C9馏分聚合而成的热塑性树脂,经加氢改性后可以制得色度浅、抗氧化稳定性高、相容性好的高附加值氢化石油树脂(HC9PR)。但C9PR分子量大、空间位阻高且原料中含有硫化物等杂质易使催化剂中毒失活,因此加氢难度极大。为了同时实现C9PR的加氢和脱杂,采用化学还原法结合球磨法制备了非晶态NiP@γ-Al2O3催化剂。表征结果显示,该催化剂具有较大的比表面积、富含Ni δ+活性物种、较多的活性位数量,并对C
中图分类号:
胡德政, 王榕, 王世栋, 杨文菲, 张宏伟, 袁珮. 兼具加氢和脱硫活性的富含Ni δ+非晶态NiP@γ-Al2O3催化剂的构筑及其用于石油树脂加氢的性能研究[J]. 化工学报, 2024, 75(9): 3152-3162.
Dezheng HU, Rong WANG, Shidong WANG, Wenfei YANG, Hongwei ZHANG, Pei YUAN. Construction of amorphous NiP@γ-Al2O3 catalyst rich in Ni δ+ for petroleum resin hydrogenation with enhanced hydrogenation and desulfurization activity[J]. CIESC Journal, 2024, 75(9): 3152-3162.
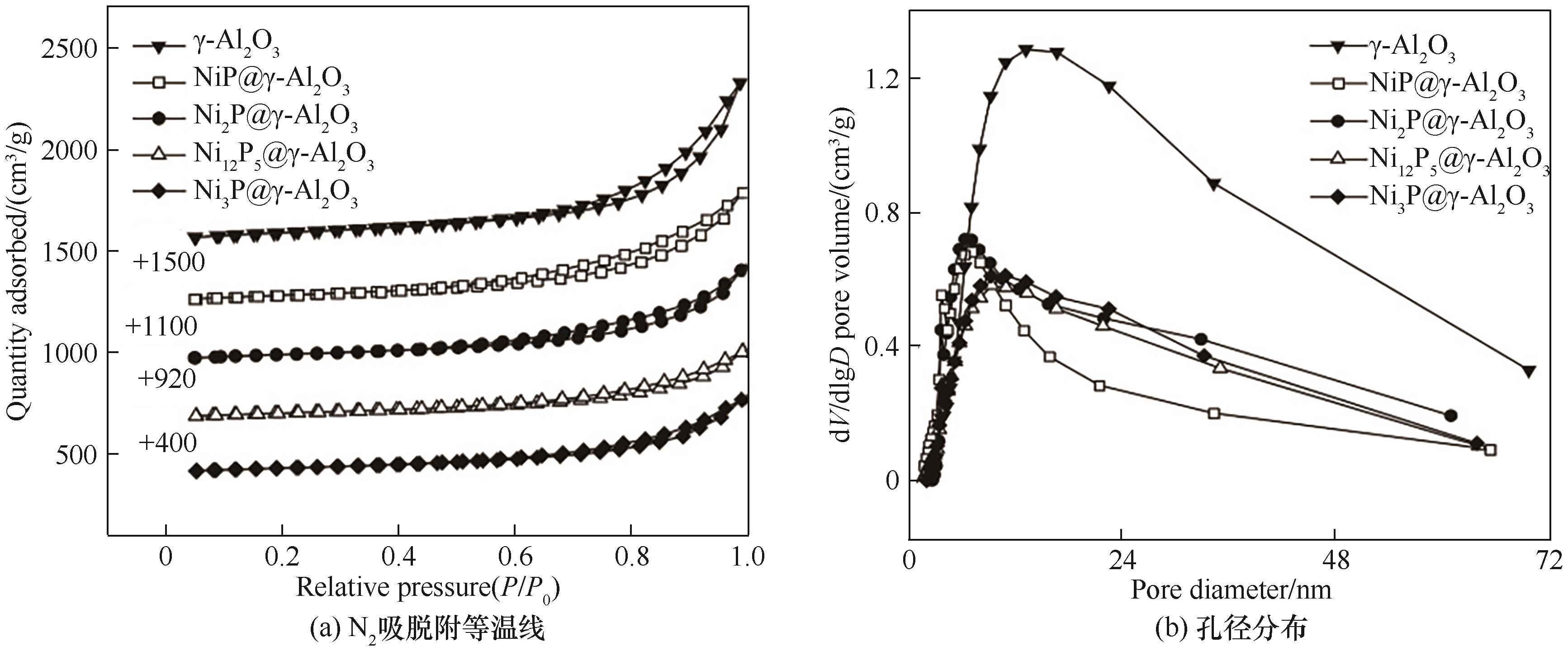
图2 γ-Al2O3、非晶态NiP@γ-Al2O3、不同晶型催化剂N2吸附-脱附等温曲线和孔径分布
Fig.2 N2 adsorption-desorption curves and pore size distributions of γ-Al2O3, amorphous NiP@γ-Al2O3 and different crystalline catalysts
| Sample | BET surface area/(m2/g) | Pore volume/(cm3/g) | Pore diameter/nm |
|---|---|---|---|
| γ-Al2O3 | 320 | 0.88 | 15.8 |
| 非晶态NiP@γ-Al2O3 | 255 | 0.47 | 6.9 |
| Ni2P@γ-Al2O3 | 241 | 0.53 | 6.3 |
| Ni12P5@γ-Al2O3 | 214 | 0.45 | 9.3 |
| Ni3P@γ-Al2O3 | 213 | 0.65 | 10.9 |
表1 γ-Al2O3载体、非晶态NiP@γ-Al2O3和不同晶型催化剂的织构性质
Table 1 Textural properties of γ-Al2O3, amorphous NiP@γ-Al2O3 and different crystalline catalysts
| Sample | BET surface area/(m2/g) | Pore volume/(cm3/g) | Pore diameter/nm |
|---|---|---|---|
| γ-Al2O3 | 320 | 0.88 | 15.8 |
| 非晶态NiP@γ-Al2O3 | 255 | 0.47 | 6.9 |
| Ni2P@γ-Al2O3 | 241 | 0.53 | 6.3 |
| Ni12P5@γ-Al2O3 | 214 | 0.45 | 9.3 |
| Ni3P@γ-Al2O3 | 213 | 0.65 | 10.9 |
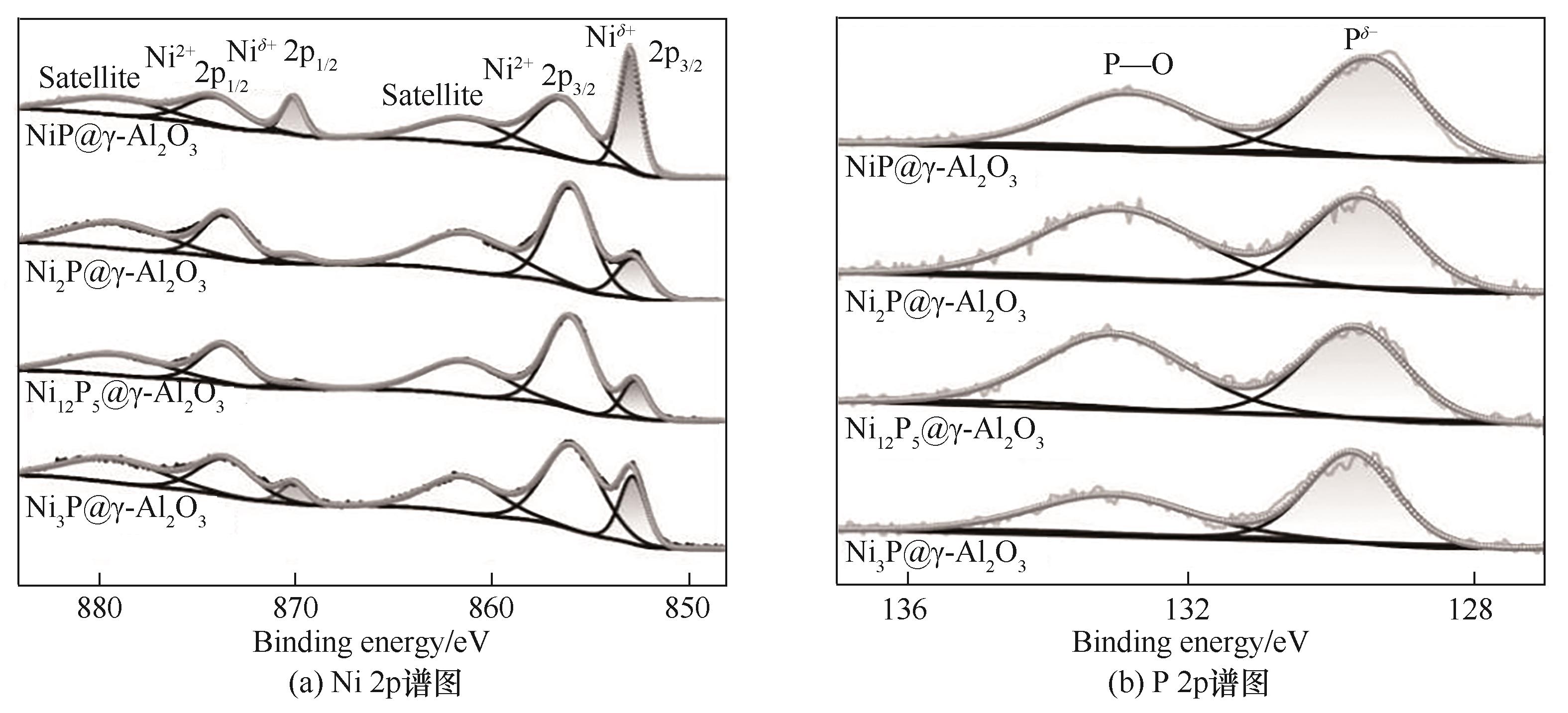
图3 非晶态NiP@γ-Al2O3和不同晶型磷化镍催化剂的Ni 2p和P 2p XPS谱图
Fig.3 Ni 2p XPS spectra and P 2p XPS spectra of amorphous NiP@γ-Al2O3 and different crystalline catalysts
| Sample | Ni species① | P species① | CO uptakes②/(μmol/g) | C2H4-TPD peak area③ | |||
|---|---|---|---|---|---|---|---|
| Ni δ+/% | Ni2+/% | Satellite/% | P δ-/% | P—O/% | |||
| 非晶态NiP@γ-Al2O3 | 36.51 | 37.44 | 26.06 | 58.44 | 41.56 | 21.67 | 1.00 |
| Ni2P@γ-Al2O3 | 12.06 | 45.76 | 42.17 | 47.15 | 52.85 | 11.43 | 0.69 |
| Ni12P5@γ-Al2O3 | 9.92 | 50.42 | 39.66 | 46.64 | 53.36 | 6.92 | 0.62 |
| Ni3P@γ-Al2O3 | 16.44 | 48.47 | 35.09 | 56.85 | 43.15 | 16.42 | 0.89 |
表2 非晶态NiP@γ-Al2O3,Ni2P@γ-Al2O3,Ni12P5@γ-Al2O3和Ni3P@γ-Al2O3催化剂的性质表征结果
Table 2 Properties of amorphous NiP@γ-Al2O3, Ni2P@γ-Al2O3, Ni12P5@γ-Al2O3 and Ni3P@γ-Al2O3 catalysts
| Sample | Ni species① | P species① | CO uptakes②/(μmol/g) | C2H4-TPD peak area③ | |||
|---|---|---|---|---|---|---|---|
| Ni δ+/% | Ni2+/% | Satellite/% | P δ-/% | P—O/% | |||
| 非晶态NiP@γ-Al2O3 | 36.51 | 37.44 | 26.06 | 58.44 | 41.56 | 21.67 | 1.00 |
| Ni2P@γ-Al2O3 | 12.06 | 45.76 | 42.17 | 47.15 | 52.85 | 11.43 | 0.69 |
| Ni12P5@γ-Al2O3 | 9.92 | 50.42 | 39.66 | 46.64 | 53.36 | 6.92 | 0.62 |
| Ni3P@γ-Al2O3 | 16.44 | 48.47 | 35.09 | 56.85 | 43.15 | 16.42 | 0.89 |
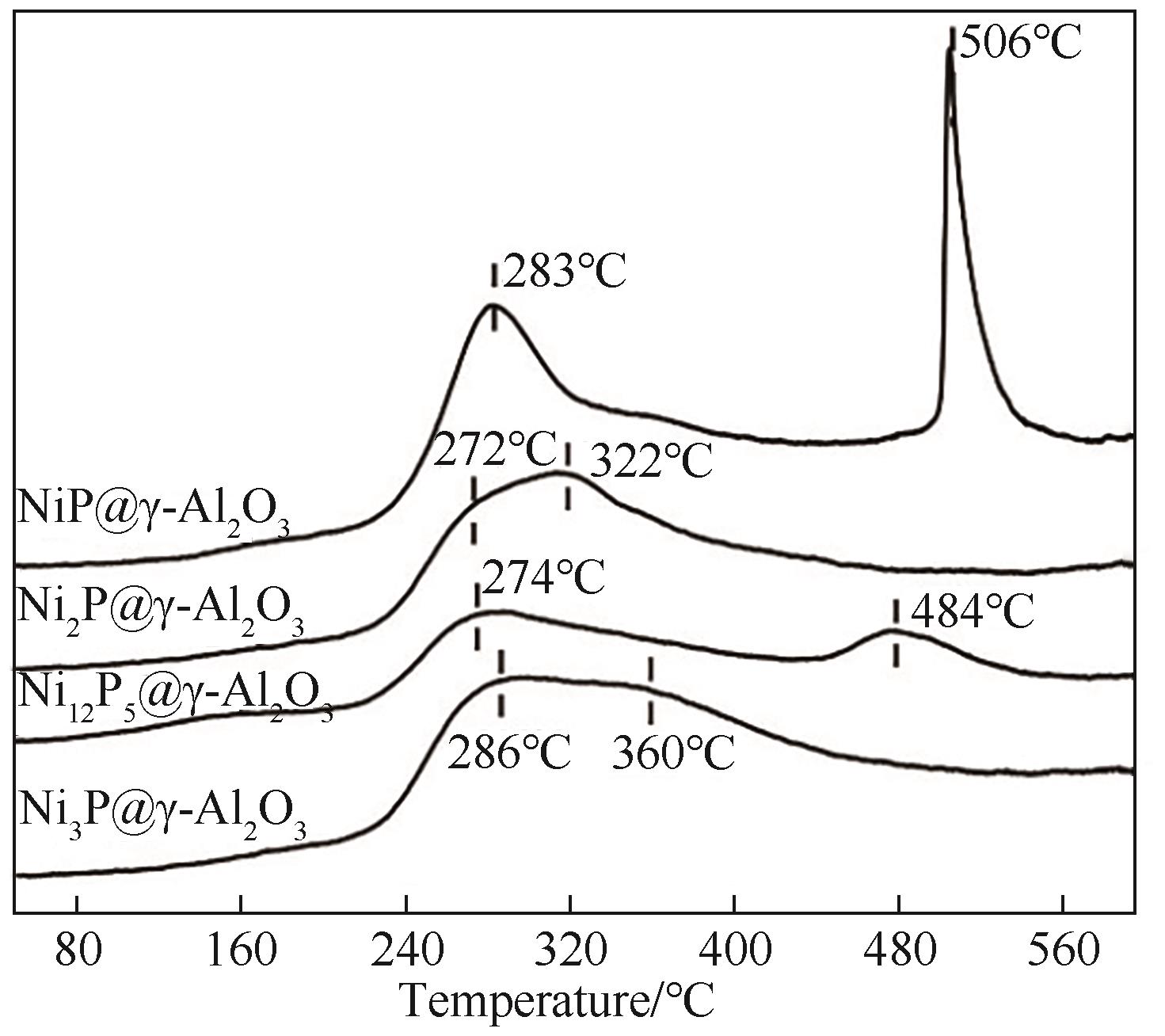
图4 非晶态NiP@γ-Al2O3,Ni2P@γ-Al2O3,Ni12P5@γ-Al2O3和Ni3P@γ-Al2O3催化剂的乙烯-TPD图
Fig.4 C2H4-TPD profiles of amorphous NiP@γ-Al2O3, Ni2P@γ- Al2O3, Ni12P5@γ-Al2O3 and Ni3P@γ-Al2O3 catalysts
| Sample | 溴值/ (g/100 g) | 色度 | 硫含量/ (μg/g) | 加氢度/% |
|---|---|---|---|---|
| C9PR | 5.06 | 5.6# | 122.80 | — |
| Blank | 3.65 | 4.3# | 102.80 | 27.86 |
| 非晶态NiP@γ-Al2O3 | 0.08 | 0.3# | 8.00 | 98.35 |
| Ni2P@γ-Al2O3 | 0.63 | 0.7# | 28.60 | 87.58 |
| Ni12P5@γ-Al2O3 | 0.94 | 2.7# | 28.53 | 81.37 |
| Ni3P@γ-Al2O3 | 0.30 | 1.0# | 28.02 | 94.05 |
表3 C9PR和HC9PR的溴值、Gardner色度、硫含量及加氢度数据
Table 3 The bromine value, Gardner color, sulfur content and hydrogenation degree of raw C9PR and HC9PR products
| Sample | 溴值/ (g/100 g) | 色度 | 硫含量/ (μg/g) | 加氢度/% |
|---|---|---|---|---|
| C9PR | 5.06 | 5.6# | 122.80 | — |
| Blank | 3.65 | 4.3# | 102.80 | 27.86 |
| 非晶态NiP@γ-Al2O3 | 0.08 | 0.3# | 8.00 | 98.35 |
| Ni2P@γ-Al2O3 | 0.63 | 0.7# | 28.60 | 87.58 |
| Ni12P5@γ-Al2O3 | 0.94 | 2.7# | 28.53 | 81.37 |
| Ni3P@γ-Al2O3 | 0.30 | 1.0# | 28.02 | 94.05 |
| Sample | 溴值/ (g/100 g) | 色度 | 硫含量/ (μg/g) | 加氢度/ % |
|---|---|---|---|---|
| C9PR | 5.06 | 5.6# | 122.80 | — |
| 10% 非晶态NiP@γ-Al2O3 | 0.12 | 0.8# | 25.00 | 97.65 |
| 20% 非晶态NiP@γ-Al2O3 | 0.08 | 0.3# | 8.00 | 98.35 |
| 30% 非晶态NiP@γ-Al2O3 | 0.02 | 0.2# | 0 | 99.10 |
表4 C9PR与HC9PR的物性参数
Table 4 Characteristic of raw C9PR and HC9PR products
| Sample | 溴值/ (g/100 g) | 色度 | 硫含量/ (μg/g) | 加氢度/ % |
|---|---|---|---|---|
| C9PR | 5.06 | 5.6# | 122.80 | — |
| 10% 非晶态NiP@γ-Al2O3 | 0.12 | 0.8# | 25.00 | 97.65 |
| 20% 非晶态NiP@γ-Al2O3 | 0.08 | 0.3# | 8.00 | 98.35 |
| 30% 非晶态NiP@γ-Al2O3 | 0.02 | 0.2# | 0 | 99.10 |
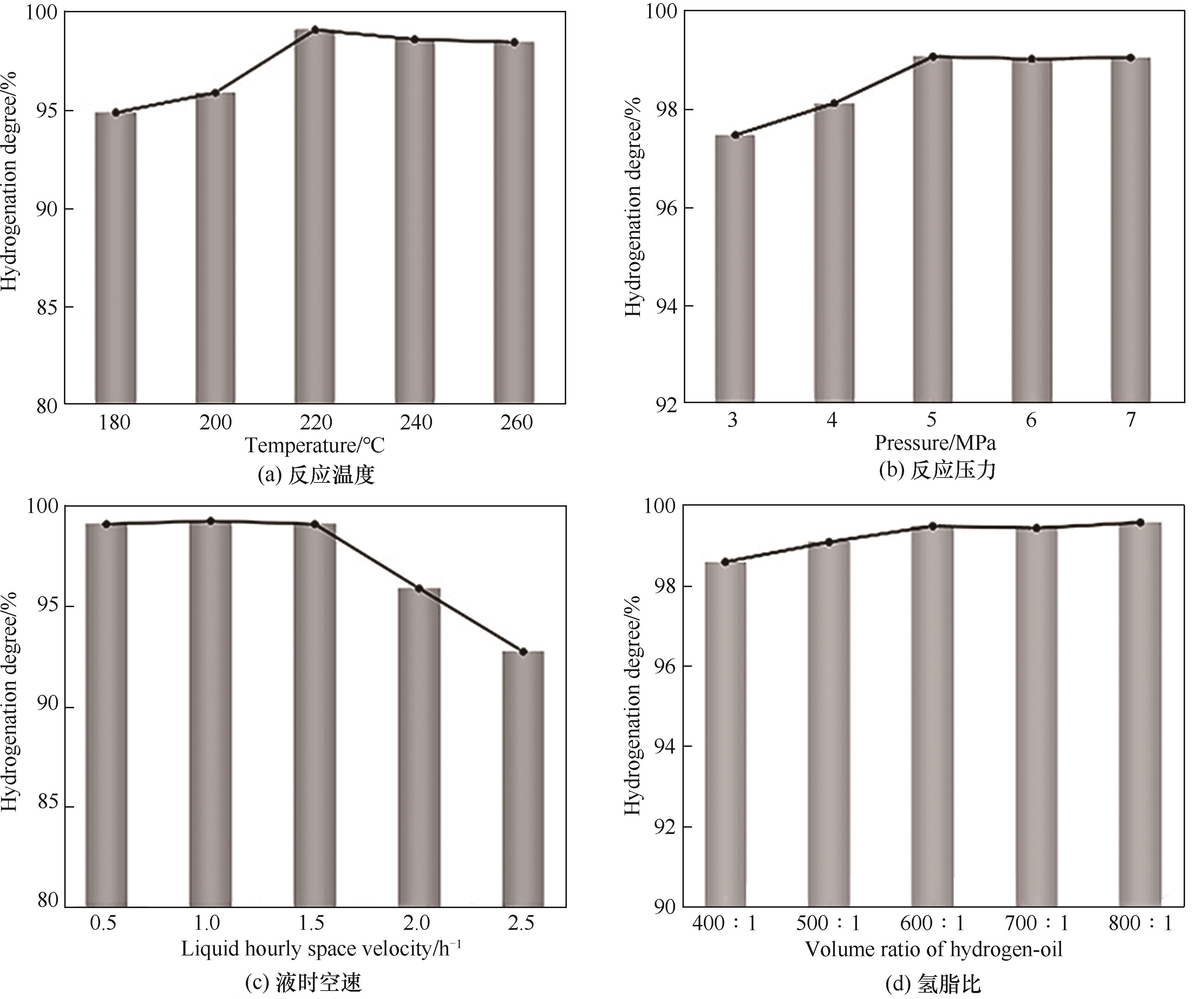
图9 不同反应条件对30%非晶态NiP@γ-Al2O3催化剂的C9PR加氢性能的影响(加氢反应条件固定,考察单一变量:温度220℃、压力5 MPa、液时空速1.5 h-1、氢脂比600∶1)
Fig.9 Effect of different reaction conditions on the C9PR hydrogenation performance of 30% amorphous NiP@γ-Al2O3 catalyst (hydrogenation reaction conditions were fixed and a single variable was examined: temperature of 220℃, pressure of 5 MPa, LHSV of 1.5 h-1, VRHO of 600∶1)
| Sample | Ni0/% | Ni δ+/% | Ni2+/% | Satellite/% | P δ-/% | P—O/% |
|---|---|---|---|---|---|---|
| fresh NiP@γ-Al2O3 | — | 39.96 | 34.26 | 25.78 | 53.68 | 46.32 |
| spent NiP@γ-Al2O3 | 4.58 | 23.89 | 36.87 | 34.66 | 40.53 | 59.47 |
表5 反应前后NiP@γ-Al2O3催化剂中的各物种组成占比
Table 5 Elemental composition ratio of fresh and spent NiP@γ-Al2O3 catalysts
| Sample | Ni0/% | Ni δ+/% | Ni2+/% | Satellite/% | P δ-/% | P—O/% |
|---|---|---|---|---|---|---|
| fresh NiP@γ-Al2O3 | — | 39.96 | 34.26 | 25.78 | 53.68 | 46.32 |
| spent NiP@γ-Al2O3 | 4.58 | 23.89 | 36.87 | 34.66 | 40.53 | 59.47 |
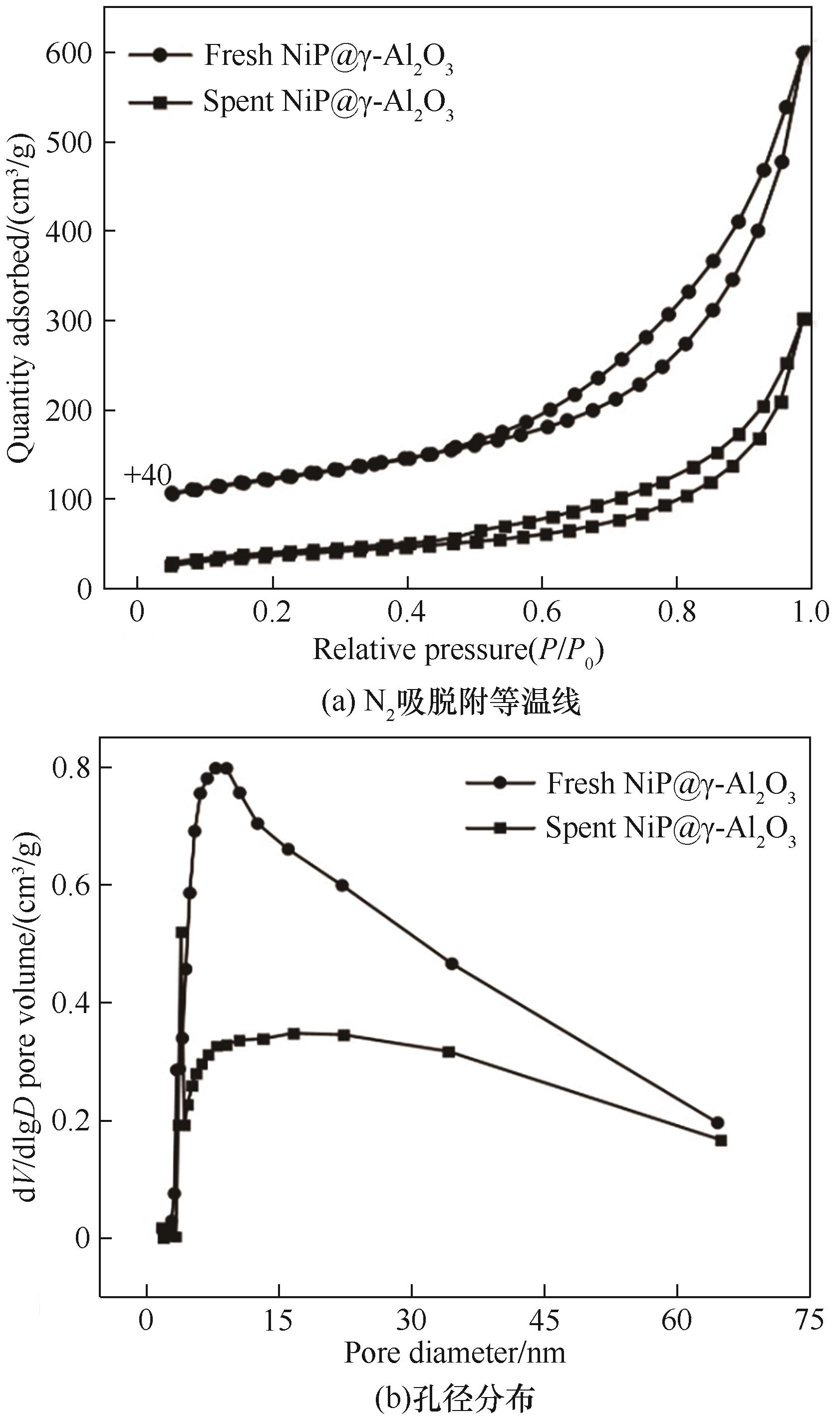
图12 反应前后30%非晶态NiP@γ-Al2O3催化剂的N2吸附-脱附等温曲线和孔径分布
Fig.12 N2 adsorption-desorption curves and pore size distributions of fresh and spent NiP@γ-Al2O3 catalysts
| Sample | BET surface area/(m2/g) | Pore volume/(cm3/g) | Pore diameter/nm |
|---|---|---|---|
| fresh NiP@γ-Al2O3 | 230 | 0.47 | 7.9 |
| spent NiP@γ-Al2O3 | 128 | 0.30 | 3.9/7.9 |
表6 反应前后30%非晶态NiP@γ-Al2O3催化剂的织构性质
Table 6 Textural properties of fresh and spent NiP@γ-Al2O3 catalysts
| Sample | BET surface area/(m2/g) | Pore volume/(cm3/g) | Pore diameter/nm |
|---|---|---|---|
| fresh NiP@γ-Al2O3 | 230 | 0.47 | 7.9 |
| spent NiP@γ-Al2O3 | 128 | 0.30 | 3.9/7.9 |
| 1 | Jiang M, Wei X J, Chen X P, et al. C9 petroleum resin hydrogenation over a PEG1000-modified nickel catalyst supported on a recyclable fluid catalytic cracking catalyst residue[J]. ACS Omega, 2020, 5(32): 20291-20298. |
| 2 | Zhang W G, Qiu L, Liu J P, et al. Modification mechanism of C9 petroleum resin and its influence on SBS modified asphalt[J]. Construction and Building Materials, 2021, 306: 124740. |
| 3 | 何志山, 韦小杰, 陈小鹏, 等. β-环糊精修饰Ni/SFCC强化C9石油树脂催化加氢性能[J]. 石油炼制与化工, 2020, 51(6): 62-71. |
| He Z S, Wei X J, Chen X P, et al. β-Cyclodextrin modified Ni/SFCC to enhance catalytic hydrogenation of C9 petroleum resin[J]. Petroleum Processing and Petrochemicals, 2020, 51(6): 62-71. | |
| 4 | 俞陆军, 江大好, 徐娇, 等. C9石油树脂显色原因及其加氢改性的研究[J]. 石油化工, 2012, 41(10): 1181-1185. |
| Yu L J, Jiang D H, Xu J, et al. Color cause and hydrogenation modification of C9 petroleum resin[J]. Petrochemical Technology, 2012, 41(10): 1181-1185. | |
| 5 | Yu L J, Jiang D H, Xu J, et al. Two-stage hydrogenation modification of C9 petroleum resin over NiWS/γ-Al2O3 and PdRu/γ-Al2O3 catalysts in series[J]. China Petroleum Processing & Petrochemical Technology, 2012, 14(3): 83-89. |
| 6 | Suwaid M A, Varfolomeev M A, Al-muntaser A A, et al. In-situ catalytic upgrading of heavy oil using oil-soluble transition metal-based catalysts[J]. Fuel, 2020, 281: 118753. |
| 7 | Sae-Ma N, Praserthdam P, Panpranot J, et al. Color improvment of C9 hydrocarbon resin by hydrogenation over 2% Pd/γ-alumina catalyst: effect of degree of aromatic rings hydrogenation[J]. Journal of Applied Polymer Science, 2010, 117(5): 2862-2869. |
| 8 | Petrukhina N N, Korchagina S A, Khan O I, et al. Hydrogenation of polymeric petroleum resins in the presence of unsupported sulfide catalysts synthesized from water-soluble precursors[J]. Petroleum Chemistry, 2018, 58(14): 1192-1197. |
| 9 | Petrukhina N N, Zakharyan E M, Korchagina S A, et al. Hydrogenation of petroleum resins in the presence of supported sulfide catalysts[J]. Petroleum Chemistry, 2018, 58(1): 48-55. |
| 10 | Wu C H, Chen X P, Tang L Q, et al. Rationally constructing a nano MOF-derived Ni and CQD embedded N-doped carbon nanosphere for the hydrogenation of petroleum resin at low temperature[J]. ACS Applied Materials & Interfaces, 2021, 13(9): 10855-10869. |
| 11 | Liu Q H, Yang J T, Zhang H W, et al. Tuning the properties of Ni-based catalyst via La incorporation for efficient hydrogenation of petroleum resin[J]. Chinese Journal of Chemical Engineering, 2022, 45: 41-50. |
| 12 | Oyama S T, Wang X, Lee Y K, et al. Effect of phosphorus content in nickel phosphide catalysts studied by XAFS and other techniques[J]. Journal of Catalysis, 2002, 210(1): 207-217. |
| 13 | Zhao H Y, Oyama S T, Freund H J, et al. Nature of active sites in Ni2P hydrotreating catalysts as probed by iron substitution[J]. Applied Catalysis B: Environmental, 2015, 164: 204-216. |
| 14 | Oyama S T, Wang X, Lee Y K, et al. Active phase of Ni2P/SiO2 in hydroprocessing reactions[J]. Journal of Catalysis, 2004, 221(2): 263-273. |
| 15 | Jiang L, Feng F, Jiang D H, et al. Highly active and stable Ni2P/SiO2 catalyst for hydrogenation of C9 petroleum resin[J]. China Petroleum Processing & Petrochemical Technology, 2016, 18(1): 36-43. |
| 16 | Wang R, Sun H M, Liang M X, et al. Flower-like nickel phosphide catalyst for petroleum resin hydrogenation with enhanced catalytic activity, hydrodesulfurization ability and stability[J]. Chemical Engineering Science, 2022, 264: 118180. |
| 17 | Yang W F, Wu S Z, Hu C J, et al. In-situ synthesis of Ni2P/Al2O3 catalyst with liquid-phase phosphidation for enhancing hydrogenation and desulfurization performance in C9 petroleum resin[J]. Chemical Engineering Science, 2024, 286: 119648. |
| 18 | Moreau L M, Ha D H, Zhang H T, et al. Defining crystalline/amorphous phases of nanoparticles through X-ray absorption spectroscopy and X-ray diffraction: the case of nickel phosphide[J]. Chemistry of Materials, 2013, 25(12): 2394-2403. |
| 19 | Li X Z, Wu K L, Ye Y, et al. Gas-assisted growth of boron-doped nickel nanotube arrays: rapid synthesis, growth mechanisms, tunable magnetic properties, and super-efficient reduction of 4-nitrophenol[J]. Nanoscale, 2013, 5(9): 3648-3653. |
| 20 | Bastakoti B P, Munkaila S, Guragain S. Micelles template for the synthesis of hollow nickel phosphate nanospheres[J]. Materials Letters, 2019, 251: 34-36. |
| 21 | Chen Y D, Li C M, Zhou J Y, et al. Metal phosphides derived from hydrotalcite precursors toward the selective hydrogenation of phenylacetylene[J]. ACS Catalysis, 2015, 5(10): 5756-5765. |
| 22 | Gan Y, Wang C, Chen X, et al. High conductivity Ni12P5 nanowires as high-rate electrode material for battery-supercapacitor hybrid devices[J]. Chemical Engineering Journal, 2020, 392: 123661. |
| 23 | Laursen A, Wexler R B, Whitaker M J, et al. Climbing the volcano of electrocatalytic activity while avoiding catalyst corrosion: Ni3P, a hydrogen evolution electrocatalyst stable in both acid and alkali[J]. ACS Catalysis, 2018, 8(5): 4408-4419. |
| 24 | Wang F, Ma J Z, He G Z, et al. Nanosize effect of Al2O3 in Ag/Al2O3 catalyst for the selective catalytic oxidation of ammonia[J]. ACS Catalysis, 2018, 8(4): 2670-2682. |
| 25 | Feng J T, Lin Y J, Evans D G, et al. Enhanced metal dispersion and hydrodechlorination properties of a Ni/Al2O3 catalyst derived from layered double hydroxides[J]. Journal of Catalysis, 2009, 266(2): 351-358. |
| 26 | Du W C, Zheng L P, Li X W, et al. Plate-like Ni-Mg-Al layered double hydroxide synthesized via a solvent-free approach and its application in hydrogenolysis of D-sorbitol[J]. Applied Clay Science, 2016, 123: 166-172. |
| 27 | Liu T, Li A R, Wang C B, et al. Interfacial electron transfer of Ni2P-NiP2 polymorphs inducing enhanced electrochemical properties[J]. Advanced Materials, 2018, 30(46): e1803590. |
| 28 | Xu J, Yang N J, Yu S Y, et al. Ultra-high energy density supercapacitors using a nickel phosphide/nickel/titanium carbide nanocomposite capacitor electrode[J]. Nanoscale, 2020, 12(25): 13618-13625. |
| 29 | Wang Z K, Wang S Y, Ma L X, et al. Water-induced formation of Ni2P-Ni12P5 interfaces with superior electrocatalytic activity toward hydrogen evolution reaction[J]. Small, 2021, 17(6): 2006770. |
| 30 | Fu H Q, Zhang L, Wang C W, et al. 1D/1D hierarchical nickel sulfide/phosphide nanostructures for electrocatalytic water oxidation[J]. ACS Energy Letters, 2018, 3(9): 2021-2029. |
| 31 | Li S L, Ma R G, Hu J C, et al. Coordination environment tuning of nickel sites by oxyanions to optimize methanol electro-oxidation activity[J]. Nature Communications, 2022, 13(1): 2916. |
| 32 | Ihsan-Ul-Haq M, Huang H, Cui J, et al. Chemical interactions between red P and functional groups in NiP3/CNT composite anodes for enhanced sodium storage[J]. Journal of Materials Chemistry A, 2018, 6(41): 20184-20194. |
| 33 | Ge X H, Dou M Y, Cao Y Q, et al. Mechanism driven design of trimer Ni1Sb2 site delivering superior hydrogenation selectivity to ethylene[J]. Nature Communications, 2022, 13: 5534. |
| 34 | Shimura K, Yoshida S, Oikawa H, et al. Impacts of Ni-loading method on the structure and the catalytic activity of NiO/SiO2-Al2O3 for ethylene oligomerization[J]. Catalysts, 2023, 13(9): 1303. |
| 35 | Sun H M, Yang J T, Zhang H W, et al. Hierarchical flower-like NiCu/SiO2 bimetallic catalysts with enhanced catalytic activity and stability for petroleum resin hydrogenation[J]. Industrial & Engineering Chemistry Research, 2021, 60(15): 5432-5442. |
| 36 | Chen D, Wang L L, Chen X P, et al. A Ni-based catalyst with polyvinyl pyrrolidone as a dispersant supported in a pretreated fluid catalytic cracking catalyst residue for C9 petroleum resin (C9 PR) hydrogenation[J]. Royal Society Open Science, 2018, 5(5): 172052. |
| 37 | Bai Z X, Chen X, Li C, et al. Preparation of supported palladium catalyst from hydrotalcite-like compound for dicyclopentadiene resin hydrogenation[J]. Molecular Catalysis, 2020, 484: 110728. |
| [1] | 杨露, 刘聪聪, 孟彤彤, 张博远, 杨腾飞, 邓文安, 王晓斌. 分散型催化剂在煤/重油共炼体系中的加氢抑焦作用[J]. 化工学报, 2024, 75(7): 2556-2564. |
| [2] | 黄志鸿, 周利, 柴士阳, 吉旭. 耦合加氢装置优化的多周期氢网络集成[J]. 化工学报, 2024, 75(5): 1951-1965. |
| [3] | 丁禹, 杨昌泽, 李军, 孙会东, 商辉. 原子尺度钼系加氢脱硫催化剂的研究进展与展望[J]. 化工学报, 2024, 75(5): 1735-1749. |
| [4] | 王成秀, 宋大山, 李之辉, 杨潇, 蓝兴英, 高金森, 徐春明. Geldart C类脱硫灰颗粒在环流耦合提升管内稳定流动特性[J]. 化工学报, 2024, 75(4): 1485-1496. |
| [5] | 韩宇, 周乐, 张鑫, 罗勇, 孙宝昌, 邹海魁, 陈建峰. 高黏附性Pd/SiO2/NF整体式催化剂的制备及加氢性能研究[J]. 化工学报, 2024, 75(4): 1533-1542. |
| [6] | 杨欣, 王文, 徐凯, 马凡华. 高压氢气加注过程中温度特征仿真分析[J]. 化工学报, 2023, 74(S1): 280-286. |
| [7] | 曹跃, 余冲, 李智, 杨明磊. 工业数据驱动的加氢裂化装置多工况切换过渡状态检测[J]. 化工学报, 2023, 74(9): 3841-3854. |
| [8] | 杨绍旗, 赵淑蘅, 陈伦刚, 王晨光, 胡建军, 周清, 马隆龙. Raney镍-质子型离子液体体系催化木质素平台分子加氢脱氧制备烷烃[J]. 化工学报, 2023, 74(9): 3697-3707. |
| [9] | 杨菲菲, 赵世熙, 周维, 倪中海. Sn掺杂的In2O3催化CO2选择性加氢制甲醇[J]. 化工学报, 2023, 74(8): 3366-3374. |
| [10] | 陈宇豪, 陈晓平, 马吉亮, 梁财. 市政污泥回转窑焚烧气态污染物排放特性研究[J]. 化工学报, 2023, 74(5): 2170-2178. |
| [11] | 白天昊, 王晓雯, 杨梦滋, 段新伟, 米杰, 武蒙蒙. 类水滑石衍生锌基氧化物高温煤气脱硫过程中COS释放行为及其抑制研究[J]. 化工学报, 2023, 74(4): 1772-1780. |
| [12] | 张江淮, 赵众. 碳三加氢装置鲁棒最小协方差约束控制及应用[J]. 化工学报, 2023, 74(3): 1216-1227. |
| [13] | 梁梦欣, 郭艳, 王世栋, 张宏伟, 袁珮, 鲍晓军. 氮化碳负载钯催化剂的制备及对SBS选择性催化加氢性能的研究[J]. 化工学报, 2023, 74(2): 766-775. |
| [14] | 高善彬, 李梦晨, 于宏悦, 沈雨歌, 乔亮, 迟克彬, 史得军. Pt/ZSM-22催化剂酸性原位调控及加氢异构性能[J]. 化工学报, 2023, 74(10): 4164-4172. |
| [15] | 黄宽, 马永德, 蔡镇平, 曹彦宁, 江莉龙. 油脂催化加氢转化制备第二代生物柴油研究进展[J]. 化工学报, 2023, 74(1): 380-396. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备 11010102001995号
京公网安备 11010102001995号