化工学报 ›› 2024, Vol. 75 ›› Issue (12): 4576-4586.DOI: 10.11949/0438-1157.20240433
收稿日期:2024-04-22
修回日期:2024-06-22
出版日期:2024-12-25
发布日期:2025-01-03
通讯作者:
吴震
作者简介:郭磊磊(1998—),男,博士研究生,a13546547154@stu.xjtu.edu.cn
基金资助:
Leilei GUO( ), Zhen WU(
), Zhen WU( ), Fusheng YANG, Zaoxiao ZHANG
), Fusheng YANG, Zaoxiao ZHANG
Received:2024-04-22
Revised:2024-06-22
Online:2024-12-25
Published:2025-01-03
Contact:
Zhen WU
摘要:
设计了一种填充LaNi4.3Al0.7的流通式金属氢化物反应器,用于从含0.1%CO和39.9%CO2的混合气中分离提纯高纯氢,并系统实验研究了温度、压力等参数对装置分离纯化性能的影响。结果表明,吸氢过程中换热介质最佳温度约为120℃,旨在平衡高温下强抗中毒性和低温下强反应驱动力;混合气氢分压0.84 MPa被确定为较优工况。此外提出自产氢气吹扫以去除杂质并通过实验测试其性能,相比外加纯氢吹扫(主要通过黏性流促进杂质分离),自产氢气吹扫去除杂质效果更佳,其机理在于强化杂质表面脱附及分子扩散。通过自产氢气吹扫最终氢气纯度可达99.999%,氢气回收率达77.6%,展示了金属氢化物吸收分离法在氢分离提纯领域的广阔应用前景。
中图分类号:
郭磊磊, 吴震, 杨福胜, 张早校. 基于流通式金属氢化物反应器的氢高效分离提纯实验研究[J]. 化工学报, 2024, 75(12): 4576-4586.
Leilei GUO, Zhen WU, Fusheng YANG, Zaoxiao ZHANG. Experimental research on flow-through type metal hydride reactor running in by-product mixture for hydrogen purification[J]. CIESC Journal, 2024, 75(12): 4576-4586.
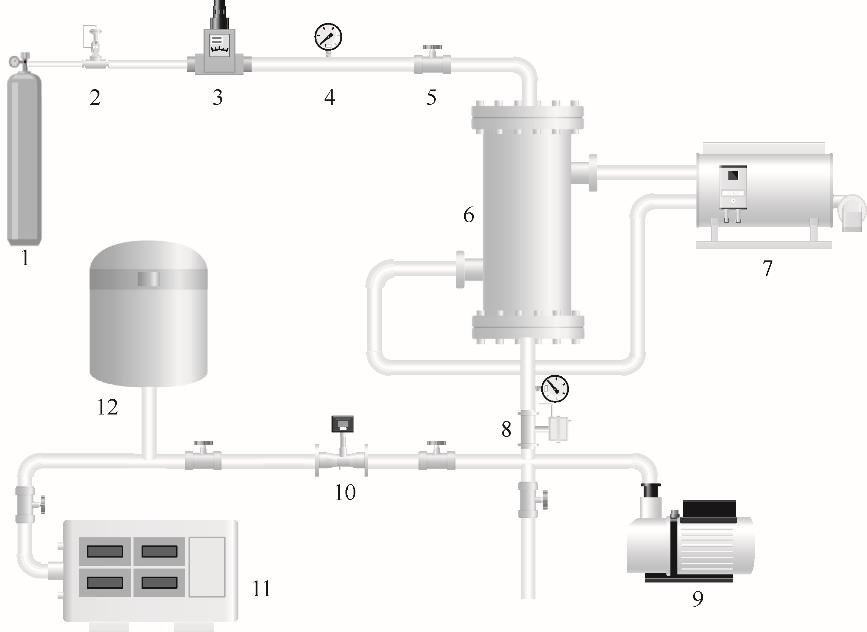
图1 金属氢化物吸收分离测试装置示意图1—气源;2—减压阀;3—流量控制器;4—压力变送器;5—阀门;6—MHAP反应器;7—换热装置;8—背压阀;9—真空泵;10—流量计;11—气相色谱仪;12—储氢罐
Fig.1 MH2P test equipment apparatus1—gas source;2—pressure regulator;3—flow controller;4—pressure transmitter;5—valve;6—MHAP reactor;7—heat transfer device;8—back pressure valve;9—vacuum pump;10—flow meter;11—gas chromatograph;12—hydrogen storage tank
| 设备 | 参数 |
|---|---|
| 真空泵 | 抽气速率1 dm3/s,极限真空2 Pa |
| 换热装置 | 温度范围-40~300℃,冷却功率1.2 kW,加热功率3.0 kW |
| 背压阀 | 控制压力范围0~2.5 MPa |
| 信号采集仪34970A | 同时收集热电偶、直流/交流电压和电流信号 |
| 气相色谱仪GC2060 | 配置有高灵敏度热导检测器(TCD)和高稳定性氢火焰离子化检测器(FID) |
| 温度传感器PT100 | 测量温度范围-200~850℃, 精度A级,输出0~5 V电压 |
| 压力变送器 | 测量压力范围-0.1~5 MPa,输出4~20 mA电流 |
| 流量控制器 | 控制流量范围0~0.56 dm3/s,精度0.5% |
表1 MH2P测试装置中部分设备及参数
Table 1 Some equipment and parameters involved in this study
| 设备 | 参数 |
|---|---|
| 真空泵 | 抽气速率1 dm3/s,极限真空2 Pa |
| 换热装置 | 温度范围-40~300℃,冷却功率1.2 kW,加热功率3.0 kW |
| 背压阀 | 控制压力范围0~2.5 MPa |
| 信号采集仪34970A | 同时收集热电偶、直流/交流电压和电流信号 |
| 气相色谱仪GC2060 | 配置有高灵敏度热导检测器(TCD)和高稳定性氢火焰离子化检测器(FID) |
| 温度传感器PT100 | 测量温度范围-200~850℃, 精度A级,输出0~5 V电压 |
| 压力变送器 | 测量压力范围-0.1~5 MPa,输出4~20 mA电流 |
| 流量控制器 | 控制流量范围0~0.56 dm3/s,精度0.5% |
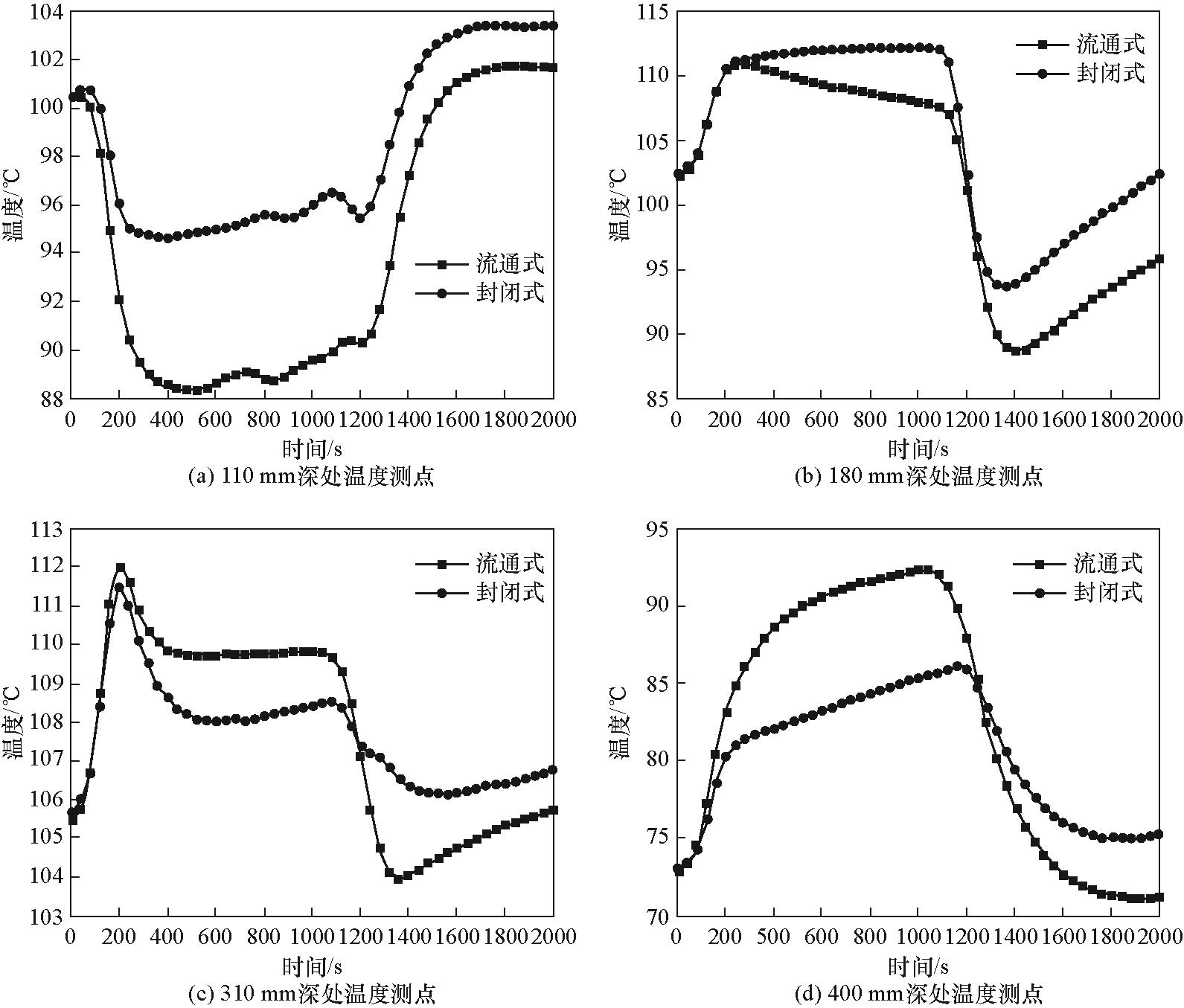
图5 流通式和封闭式MH2P反应器吸放氢过程温度变化
Fig.5 Temperature evolution of flow-through type MH2P reactor and discontinuous MH2P reactor in hydrogen absorption and desorption process
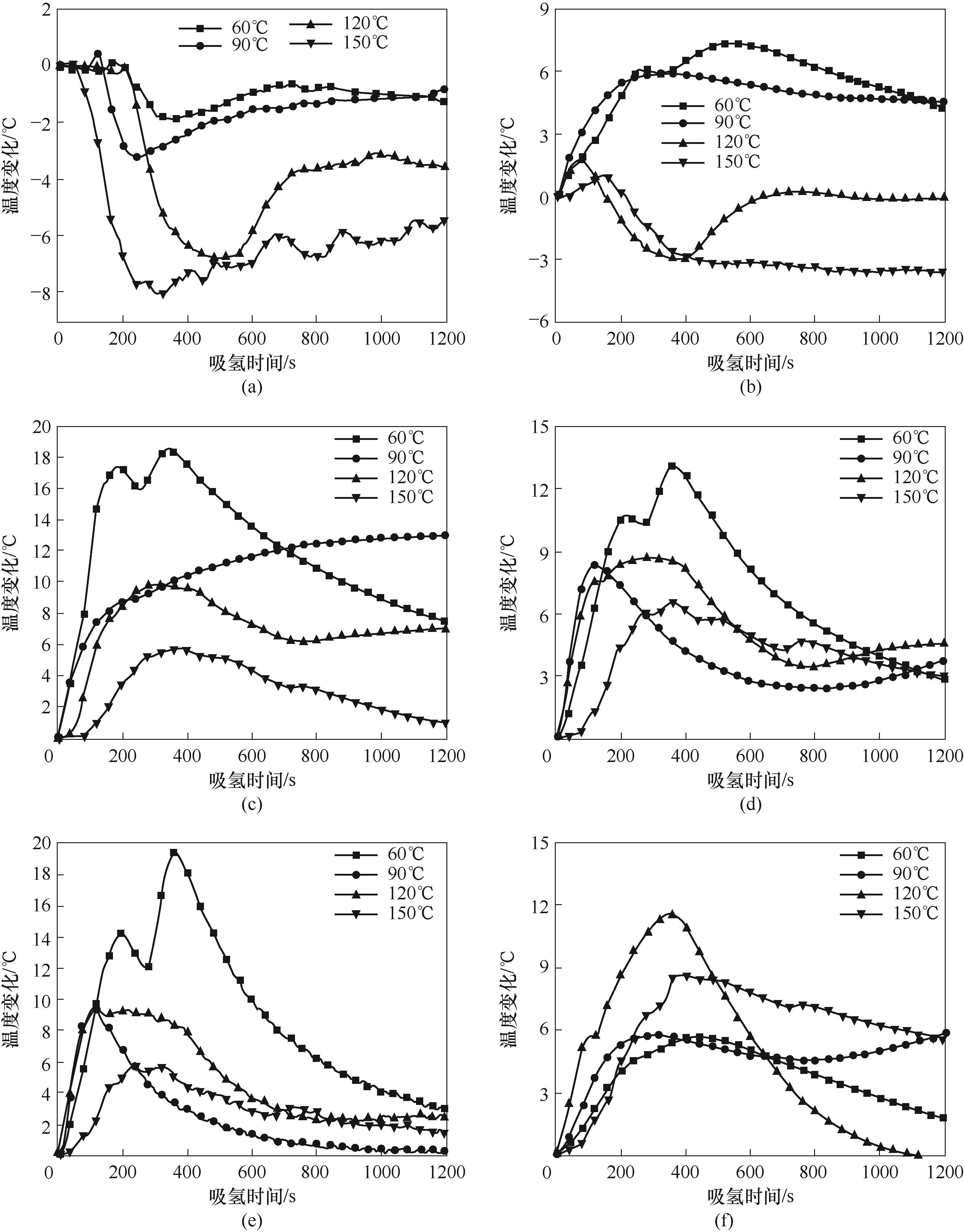
图6 110 mm(a)、150 mm(b)、180 mm(c)、240 mm(d)、310 mm(e)、400 mm(f)测点温度变化
Fig.6 Temperature evolution of measuring points at 110 mm (a), 150 mm (b), 180 mm (c), 240 mm (d), 310 mm (e) and 400 mm (f) in MH2P reactor
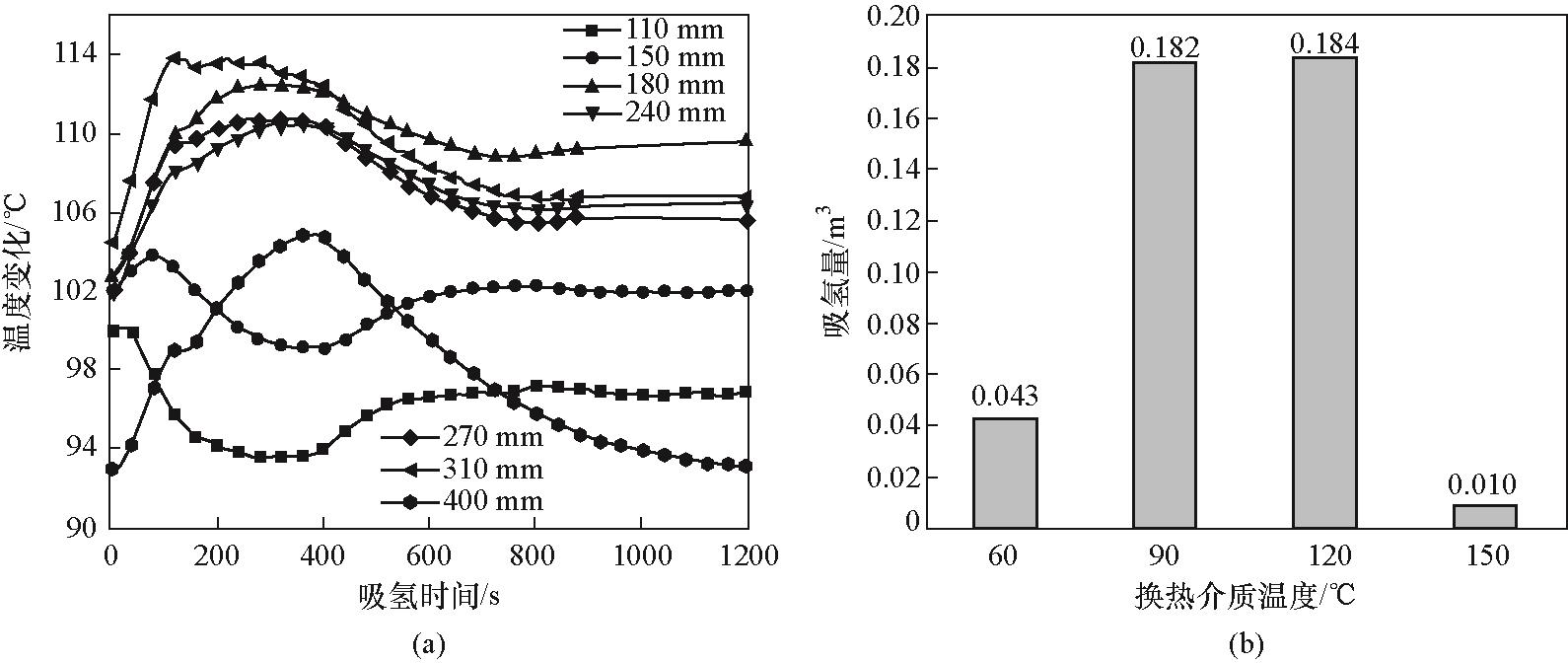
图7 (a)换热介质温度120℃下MH2P反应器内各测点温度变化;(b)吸氢2000 s、换热介质60~150℃温度范围下MH2P反应器吸氢量
Fig.7 (a) Temperature evolution of measuring points in MH2P reactor at heat exchange medium temperature of 120℃; (b) Hydrogen absorption capacity of MH2P reactor at heat exchange medium temperature of 60—150℃ after 2000 s
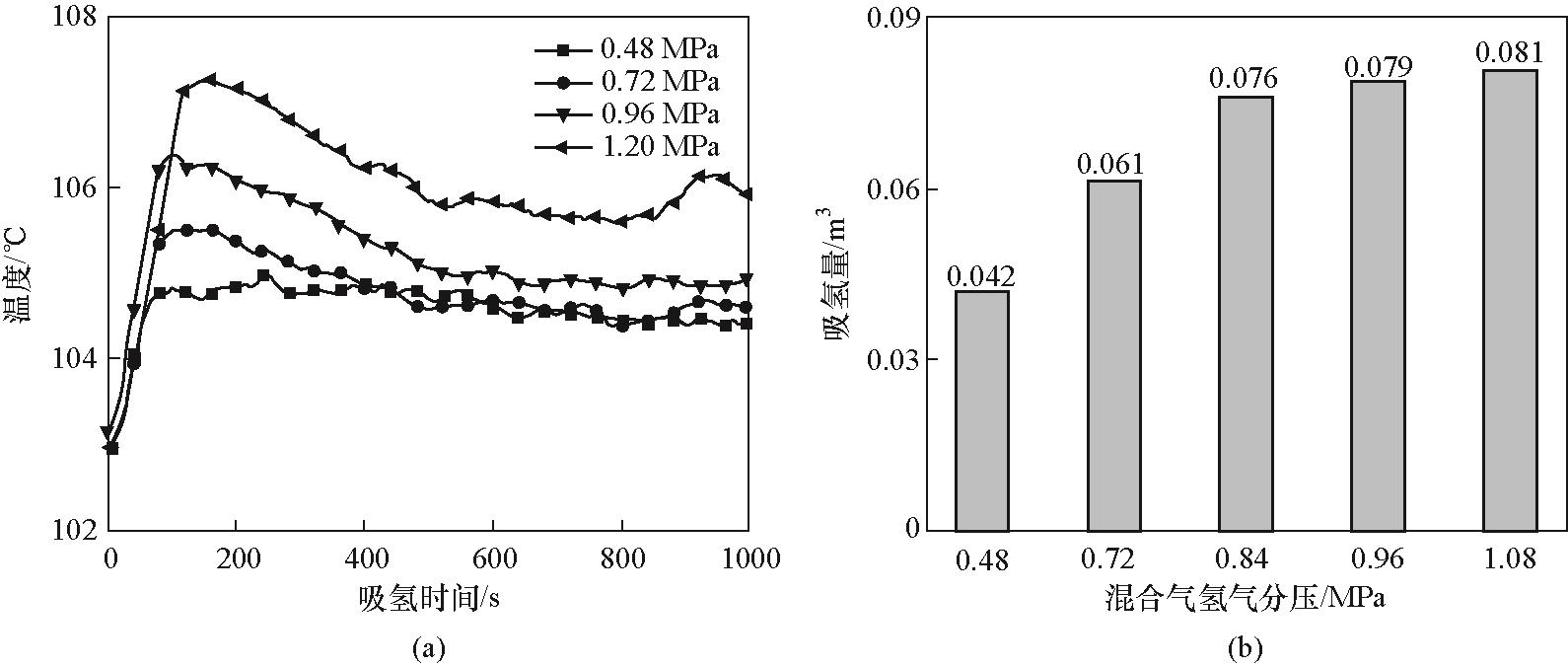
图8 (a)换热介质温度120℃、混合气中氢分压0.48~1.20 MPa范围下MH2P反应器240 mm测点温度变化;(b)换热介质温度120℃、吸氢2000 s、混合气中氢分压0.48~1.08 MPa范围下MH2P反应器吸氢量
Fig.8 (a) Temperature evolution of 240 mm measuring point in MH2P reactor at heat exchange medium temperature of 120℃ and pressure of 0.48—1.20 MPa; (b) Hydrogen absorption capacity of MH2P reactor at heat exchange medium temperature of 120℃, pressure of 0.48—1.08 MPa after 2000 s
| 1 | Martinez-Burgos W J, de Souza Candeo E, Pedroni Medeiros A B, et al. Hydrogen: current advances and patented technologies of its renewable production[J]. Journal of Cleaner Production, 2021, 286: 124970. |
| 2 | Edwards P P, Kuznetsov V L, David W I F, et al. Hydrogen and fuel cells: towards a sustainable energy future[J]. Energy Policy, 2008, 36(12): 4356-4362. |
| 3 | Baykara S Z. Hydrogen: a brief overview on its sources, production and environmental impact[J]. International Journal of Hydrogen Energy, 2018, 43(23): 10605-10614. |
| 4 | Dawood F, Anda M, Shafiullah G M. Hydrogen production for energy: an overview[J]. International Journal of Hydrogen Energy, 2020, 45(7): 3847-3869. |
| 5 | Qiao L, Kang Z X, Li Z L, et al. Crystalline porous material based membranes for hydrogen separation[J]. Fuel, 2024, 359: 130477. |
| 6 | Ali Abdelkareem M, Ayoub M, Al Najada R I, et al. Hydrogen from waste metals: recent progress, production techniques, purification, challenges, and applications[J]. Sustainable Horizons, 2024, 9: 100079. |
| 7 | Heinzel A, Vogel B, Hübner P. Reforming of natural gas—hydrogen generation for small scale stationary fuel cell systems[J]. Journal of Power Sources, 2002, 105(2): 202-207. |
| 8 | Farhana K, Shadate Faisal Mahamude A, Kadirgama K. Comparing hydrogen fuel cost of production from various sources — a competitive analysis[J]. Energy Conversion and Management, 2024, 302: 118088. |
| 9 | Nunes S P, Culfaz-Emecen P Z, Ramon G Z, et al. Thinking the future of membranes: perspectives for advanced and new membrane materials and manufacturing processes[J]. Journal of Membrane Science, 2020, 598: 117761. |
| 10 | Yousef A M, El-Maghlany W M, Eldrainy Y A, et al. New approach for biogas purification using cryogenic separation and distillation process for CO2 capture[J]. Energy, 2018, 156: 328-351. |
| 11 | Joshi A, Chaulamwar N, Tendolkar M. Design and development of PSA system for ultra purification of green hydrogen[C]//2023 IEEE International Transportation Electrification Conference (ITEC-India). Chennai, India: IEEE, 2023: 1-19. |
| 12 | 麻蓉, 张桥. PSA-低温甲醇洗-膜分离耦合的氢气分离系统建立与模拟[J]. 化工学报, 2023, 74(10): 4201-4207. |
| Ma R, Zhang Q. Establishment and simulation of hydrogen separation system coupled with PSA, rectisol and membrane separation[J]. CIESC Journal, 2023, 74(10): 4201-4207. | |
| 13 | Wang P F, Chen Y Q, Teng Y, et al. A comprehensive review of hydrogen purification using a hydrate-based method[J]. Renewable and Sustainable Energy Reviews, 2024, 194: 114303. |
| 14 | Kudapa V K, Paliyal P S, Mondal A, et al. A critical review of fabrication strategies, separation techniques, challenges, and future prospects for the hydrogen separation membrane[J]. Fusion Science and Technology, 2024, 80(7): 803-825. |
| 15 | Aasadnia M, Mehrpooya M, Ghorbani B. A novel integrated structure for hydrogen purification using the cryogenic method[J]. Journal of Cleaner Production, 2021, 278: 123872. |
| 16 | Shabbani H J K, Othman M R, Al-Janabi S K, et al. H2 purification employing pressure swing adsorption process: parametric and bibliometric review[J]. International Journal of Hydrogen Energy, 2024, 50: 674-699. |
| 17 | Chen X Y, Wei L X, Deng L, et al. A review on the metal hydride based hydrogen purification and separation technology[J]. Applied Mechanics and Materials, 2013, 448/449/450/451/452/453: 3027-3036. |
| 18 | 李开宇, 刘桂莲. 储氢提纯和氢网络的耦合优化[J]. 化工学报, 2020, 71(3): 1143-1153. |
| Li K Y, Liu G L. Coupling optimization of hydrogen-storage based purification and hydrogen network[J]. CIESC Journal, 2020, 71(3): 1143-1153. | |
| 19 | Borzone E M, Blanco M V, Meyer G O, et al. Cycling performance and hydriding kinetics of LaNi5 and LaNi4.73Sn0.27 alloys in the presence of CO[J]. International Journal of Hydrogen Energy, 2014, 39(20): 10517-10524. |
| 20 | Schweppe F, Martin M, Fromm E. Hydrogen absorption of LaNi5 powders precovered with O2, CO, H2S, CO2 or N2 [J]. Journal of Alloys and Compounds, 1997, 253: 511-514. |
| 21 | Dunikov D, Borzenko V, Malyshenko S. Influence of impurities on hydrogen absorption in a metal hydride reactor[J]. International Journal of Hydrogen Energy, 2012, 37(18): 13843-13848. |
| 22 | Miura S, Fujisawa A, Ishida M. A hydrogen purification and storage system using metal hydride[J]. International Journal of Hydrogen Energy, 2012, 37(3): 2794-2799. |
| 23 | Wang H G, Liu Y F, Zhang J. Hydrogen purification by Mg alloy hydrogen adsorbent[J]. Adsorption, 2022, 28(1): 85-95. |
| 24 | Lototskyy M, Modibane K D, Williams M, et al. Application of surface-modified metal hydrides for hydrogen separation from gas mixtures containing carbon dioxide and monoxide[J]. Journal of Alloys and Compounds, 2013, 580: 382-385. |
| 25 | Modibane K D, Williams M, Lototskyy M, et al. Poisoning-tolerant metal hydride materials and their application for hydrogen separation from CO2/CO containing gas mixtures[J]. International Journal of Hydrogen Energy, 2013, 38(23): 9800-9810. |
| 26 | Fujisawa A, Miura S, Mitsutake Y, et al. Simulation study of hydrogen purification using metal hydride[J]. Journal of Alloys and Compounds, 2013, 580: 423-426. |
| 27 | Blinov D V, Borzenko V I, Kazakov A N. Metal hydride flow-through hydrogen purification method for renewable energy production and storage[C]//2018 International Multi-Conference on Industrial Engineering and Modern Technologies (FarEastCon). Vladivostok, Russia: IEEE, 2018: 1-6. |
| 28 | Blinov D V, Borzenko V I, Dunikov D O, et al. Experimental investigations of thermal processes in the flow-throw hydrogen purification reactor[J]. Journal of Physics: Conference Series, 2018, 1128: 012120. |
| 29 | Dunikov D, Blinov D. Extraction of hydrogen from a lean mixture with methane by metal hydride[J]. International Journal of Hydrogen Energy, 2020, 45(16): 9914-9926. |
| 30 | Yang F S, Chen X Y, Wu Z, et al. Experimental studies on the poisoning properties of a low-plateau hydrogen storage alloy LaNi4.3Al0.7 against CO impurities[J]. International Journal of Hydrogen Energy, 2017, 42(25): 16225-16234. |
| 31 | Wu Z, Guo L L, Yao J, et al. Absorption of poisoned hydrogen from metal hydride under CO+H2 mixture gas for the production of clean, high purity hydrogen[J]. Journal of Cleaner Production, 2022, 365: 132751. |
| 32 | Hao P X, Wang X Y, Li S, et al. Warm hydrogen direct adsorptive separation and purification with highly CO/H2S-tolerant rare earth alloys[J]. Applications in Energy and Combustion Science, 2020, 1: 100004. |
| [1] | 李彦熹, 王晔春, 谢向东, 王进芝, 王江, 周煜, 潘盈秀, 丁文涛, 郭烈锦. 蜗壳式多通道气液旋流分离器结构优化及分离特性研究[J]. 化工学报, 2024, 75(8): 2875-2885. |
| [2] | 黄志鸿, 周利, 柴士阳, 吉旭. 耦合加氢装置优化的多周期氢网络集成[J]. 化工学报, 2024, 75(5): 1951-1965. |
| [3] | 陈彦松, 阮达, 刘渊博, 郑通, 张帅帅, 马学虎. 微通道换热器拓扑结构优化与性能研究[J]. 化工学报, 2024, 75(3): 823-835. |
| [4] | 李文俊, 赵中阳, 倪震, 周灿, 郑成航, 高翔. 基于气-液传质强化的湿法烟气脱硫CFD模拟研究[J]. 化工学报, 2024, 75(2): 505-519. |
| [5] | 郑群, 陈培强, 王长富, 熊春华, 徐万里, 阮曼. 海水激活电池电解液流动特性分析[J]. 化工学报, 2024, 75(12): 4770-4779. |
| [6] | 弓志超, 李双喜, 李方俊, 黄泽盛, 肖可应. 开启式艉轴唇形密封结构参数多目标优化及性能分析[J]. 化工学报, 2024, 75(12): 4689-4701. |
| [7] | 从文杰, 黄嘉雯, 范小强, 杨遥, 王靖岱, 阳永荣. 高压法LDPE管式反应器的结构优化方法[J]. 化工学报, 2024, 75(10): 3557-3567. |
| [8] | 余洋, 罗祎青, 魏荣辉, 张文慧, 袁希钢. 考虑节点中断风险的弹性供应链设计方法[J]. 化工学报, 2024, 75(1): 338-353. |
| [9] | 陈哲文, 魏俊杰, 张玉明. 超临界水煤气化耦合SOFC发电系统集成及其能量转化机制[J]. 化工学报, 2023, 74(9): 3888-3902. |
| [10] | 齐聪, 丁子, 余杰, 汤茂清, 梁林. 基于选择吸收纳米薄膜的太阳能温差发电特性研究[J]. 化工学报, 2023, 74(9): 3921-3930. |
| [11] | 文兆伦, 李沛睿, 张忠林, 杜晓, 侯起旺, 刘叶刚, 郝晓刚, 官国清. 基于自热再生的隔壁塔深冷空分工艺设计及优化[J]. 化工学报, 2023, 74(7): 2988-2998. |
| [12] | 江锦波, 彭新, 许文烜, 门日秀, 刘畅, 彭旭东. 泵出型螺旋槽油气密封泄漏特性及参数影响研究[J]. 化工学报, 2023, 74(6): 2538-2554. |
| [13] | 孙永尧, 高秋英, 曾文广, 王佳铭, 陈艺飞, 周永哲, 贺高红, 阮雪华. 面向含氮油田伴生气提质利用的膜耦合分离工艺设计优化[J]. 化工学报, 2023, 74(5): 2034-2045. |
| [14] | 刘尚豪, 贾胜坤, 罗祎青, 袁希钢. 基于梯度提升决策树的三组元精馏流程结构最优化[J]. 化工学报, 2023, 74(5): 2075-2087. |
| [15] | 周必茂, 许世森, 王肖肖, 刘刚, 李小宇, 任永强, 谭厚章. 烧嘴偏转角度对气化炉渣层分布特性的影响[J]. 化工学报, 2023, 74(5): 1939-1949. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备 11010102001995号
京公网安备 11010102001995号