化工学报 ›› 2019, Vol. 70 ›› Issue (1): 128-135.DOI: 10.11949/j.issn.0438-1157.20180521
蔡惊涛1( ),李代禧1(
),李代禧1( ),刘宝林1,栾翰森2,郭柏松3,魏冬青4,王浩2
),刘宝林1,栾翰森2,郭柏松3,魏冬青4,王浩2
收稿日期:2018-05-21
修回日期:2018-10-23
出版日期:2019-01-05
发布日期:2019-01-05
通讯作者:
李代禧
作者简介:蔡惊涛(1993—),男,硕士,<email>maxcaijingtao1993@foxmail.com</email>|李代禧(1975—),男,副教授,<email>dxli75@126.com</email>
基金资助:
Jingtao CAI1( ),Daixi LI1(
),Daixi LI1( ),Baolin LIU1,Hansen LUAN2,Baisong GUO3,Dongqing WEI4,Hao WANG2
),Baolin LIU1,Hansen LUAN2,Baisong GUO3,Dongqing WEI4,Hao WANG2
Received:2018-05-21
Revised:2018-10-23
Online:2019-01-05
Published:2019-01-05
Contact:
Daixi LI
摘要:
由于尿素结晶呈白色针状,晶貌单一,且传统结晶工艺不可控,严重影响药物的一致性评价结果。采用分子动力学模拟方法,从分子水平上研究不同种类的添加剂对尿素晶体生长的调控作用,揭示添加剂对药物晶体生长的调控机制。结果表明:① 六种添加剂(海藻糖、蔗糖、葡萄糖、山梨醇、赖氨酸、精氨酸)在101.325 kPa、290 K下相较于无添加剂时都能不同程度地抑制(520)晶面的生长;② 添加剂对尿素晶面(520)的吸附能越负,其抑制晶面生长效果越好,其中海藻糖抑制(520)晶面生长效果最好;③ 添加剂分子携带基团的种类与数目决定与晶层的相互作用的强弱,特别是海藻糖、蔗糖双糖类分子含有8个羟基,与晶层上的尿素分子氢键相互作用强,与溶液层中的溶质形成竞争性吸附,能更好抑制(520)晶面生长。
中图分类号:
蔡惊涛, 李代禧, 刘宝林, 栾翰森, 郭柏松, 魏冬青, 王浩. 尿素(520)晶面可控结晶的分子动力学模拟[J]. 化工学报, 2019, 70(1): 128-135.
Jingtao CAI, Daixi LI, Baolin LIU, Hansen LUAN, Baisong GUO, Dongqing WEI, Hao WANG. Controllable crystallization of urea (520) crystal plane by molecular simulation[J]. CIESC Journal, 2019, 70(1): 128-135.
| Force field | a/? | b/? | c/? | α/(°) | β/(°) | γ/(°) |
|---|---|---|---|---|---|---|
| experiment[ | 5.662 | 5.662 | 4.716 | 90.0 | 90.0 | 90.0 |
| COMPASS | 5.558 | 5.558 | 4.636 | 90.0 | 90.0 | 90.0 |
表1 尿素晶胞参数
Table 1 Unit cell parameter of urea
| Force field | a/? | b/? | c/? | α/(°) | β/(°) | γ/(°) |
|---|---|---|---|---|---|---|
| experiment[ | 5.662 | 5.662 | 4.716 | 90.0 | 90.0 | 90.0 |
| COMPASS | 5.558 | 5.558 | 4.636 | 90.0 | 90.0 | 90.0 |
| System | Additives | N additive | N crystal | N urea | N water |
|---|---|---|---|---|---|
| A0 A1 | none trehalose | 0 40 | 252 252 | 1000 1000 | 1300 1295 |
| A2 | sucrose | 40 | 252 | 1000 | 1294 |
| A3 | sorbitol | 40 | 252 | 1000 | 1297 |
| A4 | glucose | 40 | 252 | 1000 | 1299 |
| A5 | L-lysine | 40 | 252 | 1000 | 1294 |
| A6 | L-arginine | 40 | 252 | 1000 | 1304 |
表2 不同种类添加剂溶液体系的组成
Table 2 Models and detail components for simulation
| System | Additives | N additive | N crystal | N urea | N water |
|---|---|---|---|---|---|
| A0 A1 | none trehalose | 0 40 | 252 252 | 1000 1000 | 1300 1295 |
| A2 | sucrose | 40 | 252 | 1000 | 1294 |
| A3 | sorbitol | 40 | 252 | 1000 | 1297 |
| A4 | glucose | 40 | 252 | 1000 | 1299 |
| A5 | L-lysine | 40 | 252 | 1000 | 1294 |
| A6 | L-arginine | 40 | 252 | 1000 | 1304 |
| Hkl | Multiplicity | d hkl /? | Distance/? | Total facet area/% |
|---|---|---|---|---|
| (110) (001) | 4 2 | 4.00 4.72 | 172.80 330.33 | 38.00 16.12 |
| (200) | 4 | 2.83 | 196.17 | 14.20 |
| (520) | 8 | 1.05 | 194.39 | 8.05 |
表3 EM法预测尿素在真空下主要晶面族
Table 3 Main crystal planes of urea predicted by the EM model
| Hkl | Multiplicity | d hkl /? | Distance/? | Total facet area/% |
|---|---|---|---|---|
| (110) (001) | 4 2 | 4.00 4.72 | 172.80 330.33 | 38.00 16.12 |
| (200) | 4 | 2.83 | 196.17 | 14.20 |
| (520) | 8 | 1.05 | 194.39 | 8.05 |
| Additives | Number (N ads ) | E′ ads /(kJ/mol) |
|---|---|---|
| urea trehalose | 421.69±3.375 78.75±0.687 | -4.46±0.380 -34.00±0.691 |
| sucrose | 80.79±1.123 | -33.38±0.384 |
| sorbitol | 112.82±1.496 | -20.33±0.441 |
| glucose | 136.88±2.188 | -16.68±0.476 |
| L-lysine | 119.11±3.393 | -5.66±0.256 |
| L-arginine | 97.25±0.648 | -5.39±0.230 |
表4 添加剂在(520)晶面上的吸附能
Table 4 Adsorption energy of each additive on the facet (520)
| Additives | Number (N ads ) | E′ ads /(kJ/mol) |
|---|---|---|
| urea trehalose | 421.69±3.375 78.75±0.687 | -4.46±0.380 -34.00±0.691 |
| sucrose | 80.79±1.123 | -33.38±0.384 |
| sorbitol | 112.82±1.496 | -20.33±0.441 |
| glucose | 136.88±2.188 | -16.68±0.476 |
| L-lysine | 119.11±3.393 | -5.66±0.256 |
| L-arginine | 97.25±0.648 | -5.39±0.230 |
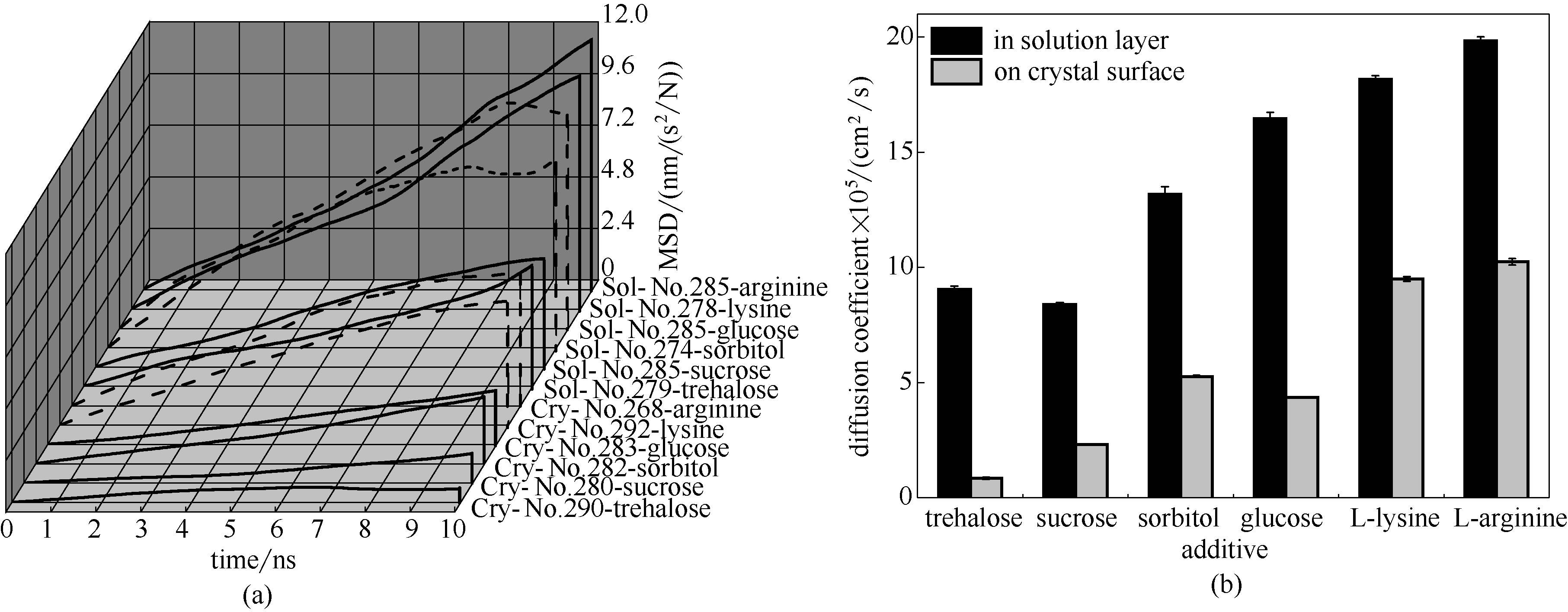
图6 分布在晶层上与溶液层中特定添加剂的均方根位移与扩散系数
Fig.6 Mean square displacement and diffusion coefficient of specific additives distributed on crystal face and in solution layer
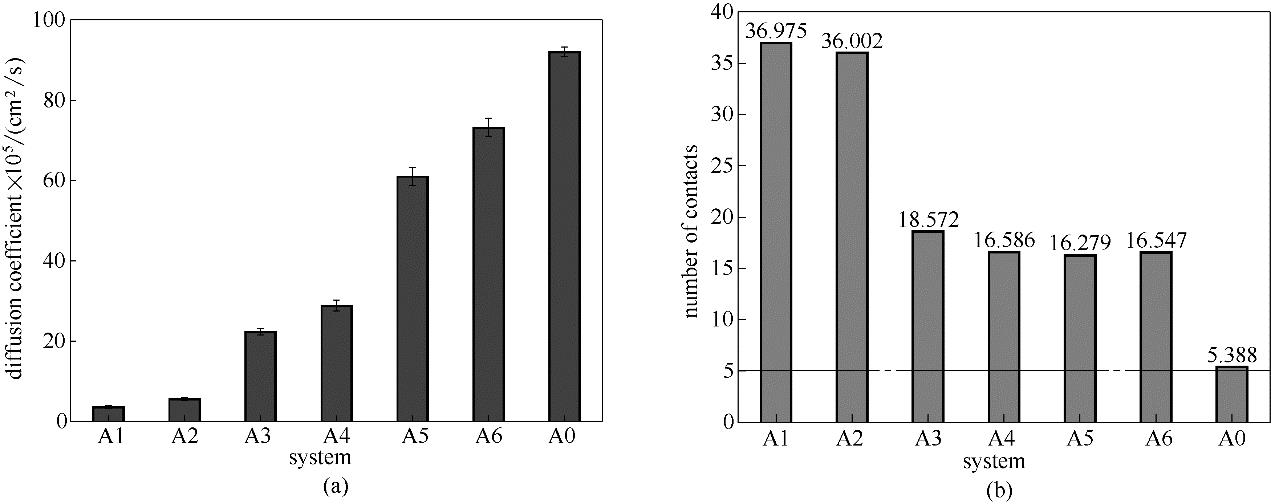
图7 各体系溶液层中尿素分子扩散系数与各特定添加剂周围聚集尿素分子数
Fig.7 Diffusion coefficient of urea molecular and urea number around each particular additive in each system solution layer
| 1 | Snyder R C , Veesler S , Doherty M F . The evolution of crystal shape during dissolution: predictions and experiments[J]. Crystal Growth & Design, 2008, 8(4): 1100-1101. |
| 2 | 宫海燕, 李彩虹, 王佩佩, 等 . 杂质对溶液结晶过程影响的研究进展[J]. 化学与生物工程, 2010, 27(3): 9-12. |
| Gong H Y , Li C H , Wang P P , et al . Research progress of influence of impurities on solution crystallization[J].Chemistry & Bioengineering, 2010, 27(3): 9-12 | |
| 3 | Mikami T , Sakuma T , Hirasawai I . CSD-controlled reactive crystallization of SrSO4 in the presence of polyethylenimine[J]. Chemical Engineering Research & Design, 2010, 88(9): 1200-1205. |
| 4 | 张相洋 . 杂质存在条件下的乳酸锌结晶行为研究[D]. 上海: 华东理工大学, 2010. |
| Zhang X Y . Crystallization behaviour of zinc lactate in presence of impurities[D]. Shanghai: East China University of Science and Technology, 2010. | |
| 5 | Zhang X , Qian G , Zhou X . Effects of different organic acids on solubility and metastable zone width of zinc lactate[J]. Journal of Chemical & Engineering Data, 2012, 57(57): 2963–2970. |
| 6 | Cashell C , Corcoran D , Hodnett B K . Effect of amino acid additives on the crystallization of L-glutamic acid[J]. Crystal Growth & Design, 2015, 5(2): 593-597. |
| 7 | Salvalaglio M , Vetter T , Giberti F , et al . Uncovering molecular details of urea crystal growth in the presence of additives[J]. Journal of the American Chemical Society, 2012, 134(41): 17221-17233. |
| 8 | 黄炳荣, 贺友平, 苏根博 . 尿素晶体生长研究[J]. 人工晶体学报, 1986, 15(2): 85-89. |
| Huang B R , He Y P , Su G B . Growth studies of urea crystals[J]. Journal of Synthetic Crystals, 1986, 15(2): 85-89. | |
| 9 | Piana S , Reyhani M , Gale J D . Simulating micrometre-scale crystal growth from solution[J]. Nature, 2005, 438(7064): 70-73. |
| 10 | 李代禧, 栾瀚森, 郭柏松, 等 . 实用计算药剂学——有效的药剂学工具[J]. 中国医药工业杂志, 2017, 48(12): 1673-1684. |
| Li D X , Luan H S , Guo B S , et al . Applied computational pharmaceutics: an efficient pharmaceutical tool[J]. Chinese Journal of Pharmaceuticals, 2017, 48(12): 1673-1684. | |
| 11 | Rapaport D C . Molecular dynamics simulation[J]. Computing in Science & Engineering, 2002, 1(1): 70-71. |
| 12 | Allen F H , Davies J E . The Cambridge Structural Database: a quarter of a million crystal structures and rising[J]. Acta Crystallographica, 2002, 58(3): 380–388. |
| 13 | Sun H . COMPASS: An ab initio force-field optimized for condensed-phase applications over view with details on alkane and benzene compounds[J]. Journal of Physical Chemistry B, 1998, 102(38): 7338-7364. |
| 14 | Sklar N , Senko M E , Post B . Thermal effects in urea: the crystal structure at 140°C and at room temperature[J]. Acta Crystallographica, 2010, 14(7): 716-720. |
| 15 | Massaro F R , Moret M , Bruno M , et al . Equilibrium and growth morphology of oligoacenes: periodic bond chains (PBC) analysis of tetracene crystal[J]. Crystal Growth & Design, 2011, 11(10): 4639-4646. |
| 16 | Hartman P , Bennema P . The attachment energy as a habit controlling factor(Ⅰ): Theoretical considerations[J]. Journal of Crystal Growth, 1980, 49(1): 145-156. |
| 17 | Liu N , Zhou C , Shu Y J , et al . Molecular dynamics simulations on crystal morphology of N-guanylurea-dinitramide[J]. Chemical Journal of Chinese Universities, 2017, 38(12): 2231-2237. |
| 18 | van der Spoel D , Lindahl E , Hess B , et al . GROMACS: fast, flexible, and free[J]. Journal of Computational Chemistry, 2005, 26(16): 1701-1718. |
| 19 | Lindorfflarsen K , Piana S , Palmo K , et al . Improved side-chain torsion potentials for the Amber ff99SB protein force field[J]. Proteins Structure Function & Bioinformatics, 2010, 78(8): 1950–1958. |
| 20 | Abascal J L F , Vega C . A general purpose model for the condensed phases of water: TIP4P/2005[J]. Journal of Chemical Physics, 2005, 123(23): 234505. |
| 21 | Darden T , York D , Pedersen L . Particle mesh Ewald: an N⋅log(N) method for ewald sums in large systems[J]. Journal of Chemical Physics, 1998, 98(12): 10089-10092. |
| 22 | Mudi A , Chakravarty C . Effect of the Berendsen thermostat on the dynamical properties of water[J]. Molecular Physics, 2004, 102(7): 681-685. |
| 23 | Bussi G , Zykova-Timan T , Parrinello M . Isothermal-isobaric molecular dynamics using stochastic velocity rescaling[J]. Journal of Chemical Physics, 2009, 130(7): 2384. |
| 24 | 唐鼎元, 贺友平, 林斯太, 等 . 紫外倍频晶体尿素的生长[J]. 人工晶体学报, 1982, (z 1): 82-83. |
| Tang D Y , He Y P , Lin S T , et al . The growth of UV frequency doubling crystal urea[J]. Journal of Synthetic Crystals, 1982, (z 1): 82-83 | |
| 25 | Janimak J J , Cheng S Z D , Giusti P A , et al . Isotacticity effect on crystallization and melting in polypropylene fractions(Ⅱ): Linear crystal growth rate and morphology study[J]. Macromolecules, 1991, 24(9): 2253-2260. |
| 26 | Pimentel G C , Mcclellan A L . Hydrogen bond[J]. Annual Review of Physical Chemistry, 2003, 22(1): 347-385. |
| 27 | Michalet X . Mean square displacement analysis of Single-Particle trajectories with localization error[J]. Physical Review E Statistical Nonlinear & Soft Matter Physics, 2010, 100(3): 041914. |
| 28 | 潘晓莉, 李代禧, 魏冬青 . 离子液体中胰岛素结构稳定性的分子动力学模拟[J]. 化工学报, 2016, 67(12): 5215-5221. |
| Pan X L , Li D X , Wei D Q . Structural stability of insulin in imidazolium ionic liquids by molecular simulation[J]. CIESC Journal, 2016, 67(12): 5215-5221. | |
| 29 | 郝保红, 黄俊华 . 晶体生长机理的研究综述[J]. 北京石油化工学院学报, 2006, 14(2): 58-64. |
| Hao B H , Huang J H . Summary on the growth mechanism of crystal[J]. Journal of Beijing Institute of Petro-chemical Technology, 2006, 14(2): 58-64 | |
| 30 | 姜兆华, 孙德智, 邵光杰 . 应用表面化学与技术[M]. 哈尔滨: 哈尔滨工业大学出版社, 2002. |
| Jiang Z H , Sun D Z , Shao G J . Applied Surface Chemistry and Technology[M]. Harbin: Harbin Institute of Technology Press, 2002. |
| [1] | 晁京伟, 许嘉兴, 李廷贤. 基于无管束蒸发换热强化策略的吸附热池的供热性能研究[J]. 化工学报, 2023, 74(S1): 302-310. |
| [2] | 宋明昊, 赵霏, 刘淑晴, 李国选, 杨声, 雷志刚. 离子液体脱除模拟油中挥发酚的多尺度模拟与研究[J]. 化工学报, 2023, 74(9): 3654-3664. |
| [3] | 杨学金, 杨金涛, 宁平, 王访, 宋晓双, 贾丽娟, 冯嘉予. 剧毒气体PH3的干法净化技术研究进展[J]. 化工学报, 2023, 74(9): 3742-3755. |
| [4] | 胡建波, 刘洪超, 胡齐, 黄美英, 宋先雨, 赵双良. 有机笼跨细胞膜易位行为的分子动力学模拟研究[J]. 化工学报, 2023, 74(9): 3756-3765. |
| [5] | 赵佳佳, 田世祥, 李鹏, 谢洪高. SiO2-H2O纳米流体强化煤尘润湿性的微观机理研究[J]. 化工学报, 2023, 74(9): 3931-3945. |
| [6] | 赵亚欣, 张雪芹, 王荣柱, 孙国, 姚善泾, 林东强. 流穿模式离子交换层析去除单抗聚集体[J]. 化工学报, 2023, 74(9): 3879-3887. |
| [7] | 盛冰纯, 于建国, 林森. 铝基锂吸附剂分离高钠型地下卤水锂资源过程研究[J]. 化工学报, 2023, 74(8): 3375-3385. |
| [8] | 张瑞航, 曹潘, 杨锋, 李昆, 肖朋, 邓春, 刘蓓, 孙长宇, 陈光进. ZIF-8纳米流体天然气乙烷回收工艺的产品纯度关键影响因素分析[J]. 化工学报, 2023, 74(8): 3386-3393. |
| [9] | 高燕, 伍鹏, 尚超, 胡泽君, 陈晓东. 基于双流体喷嘴的磁性琼脂糖微球的制备及其蛋白吸附性能探究[J]. 化工学报, 2023, 74(8): 3457-3471. |
| [10] | 汪林正, 陆俞冰, 张睿智, 罗永浩. 基于分子动力学模拟的VOCs热氧化特性分析[J]. 化工学报, 2023, 74(8): 3242-3255. |
| [11] | 陈吉, 洪泽, 雷昭, 凌强, 赵志刚, 彭陈辉, 崔平. 基于分子动力学的焦炭溶损反应及其机理研究[J]. 化工学报, 2023, 74(7): 2935-2946. |
| [12] | 董明, 徐进良, 刘广林. 超临界水非均质特性分子动力学研究[J]. 化工学报, 2023, 74(7): 2836-2847. |
| [13] | 王杰, 丘晓琳, 赵烨, 刘鑫洋, 韩忠强, 许雍, 蒋文瀚. 聚电解质静电沉积改性PHBV抗氧化膜的制备与性能研究[J]. 化工学报, 2023, 74(7): 3068-3078. |
| [14] | 崔张宁, 胡紫璇, 吴雷, 周军, 叶干, 刘田田, 张秋利, 宋永辉. 可降解纤维素基材料的耐水性能研究进展[J]. 化工学报, 2023, 74(6): 2296-2307. |
| [15] | 王新悦, 王俊杰, 曹思贤, 王翠, 李灵坤, 吴宏宇, 韩静, 吴昊. 玻璃内包材界面修饰对机械应力诱导的单克隆抗体聚集体形成的影响[J]. 化工学报, 2023, 74(6): 2580-2588. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备 11010102001995号
京公网安备 11010102001995号