化工学报 ›› 2024, Vol. 75 ›› Issue (4): 1167-1182.DOI: 10.11949/0438-1157.20231127
申州洋( ), 薛康(
), 薛康( ), 刘青, 史成香, 邹吉军, 张香文, 潘伦(
), 刘青, 史成香, 邹吉军, 张香文, 潘伦( )
)
收稿日期:2023-11-01
修回日期:2024-01-04
出版日期:2024-04-25
发布日期:2024-06-06
通讯作者:
薛康,潘伦
作者简介:申州洋(1999—),男,硕士研究生,szy_0315@tju.edu.cn
基金资助:
Zhouyang SHEN( ), Kang XUE(
), Kang XUE( ), Qing LIU, Chengxiang SHI, Jijun ZOU, Xiangwen ZHANG, Lun PAN(
), Qing LIU, Chengxiang SHI, Jijun ZOU, Xiangwen ZHANG, Lun PAN( )
)
Received:2023-11-01
Revised:2024-01-04
Online:2024-04-25
Published:2024-06-06
Contact:
Kang XUE, Lun PAN
摘要:
吸热型纳米流体燃料是一种有潜力解决高超声速飞行器发动机过热问题的新型燃料。为探究吸热型纳米流体燃料的裂解性能,并为后续研究提供参考,首先介绍了纳米流体燃料的分散稳定性机理,并概述了其制备及提高稳定性的方法;其次,综述了吸热型纳米流体燃料的裂解研究进展,从纳米添加剂、有机保护配体及反应机理和路径等多方面分析了影响裂解的关键因素;最后对吸热型纳米流体燃料的未来发展趋势提出展望。
中图分类号:
申州洋, 薛康, 刘青, 史成香, 邹吉军, 张香文, 潘伦. 吸热型纳米流体燃料研究进展[J]. 化工学报, 2024, 75(4): 1167-1182.
Zhouyang SHEN, Kang XUE, Qing LIU, Chengxiang SHI, Jijun ZOU, Xiangwen ZHANG, Lun PAN. Research progress on endothermic nanofluid fuels[J]. CIESC Journal, 2024, 75(4): 1167-1182.
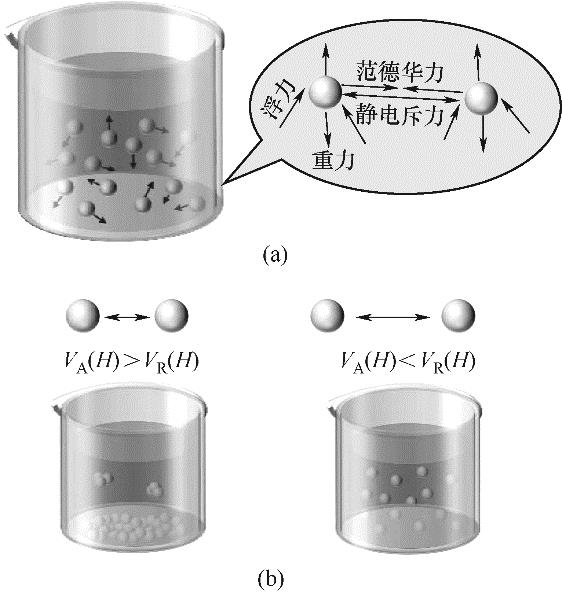
图3 纳米颗粒的布朗运动(a)和DLVO理论与纳米流体稳定性(b)示意图[23-25]
Fig.3 Schematic diagram of Brownian motion of nanoparticles (a) and DLVO theory and nanofluid stability (b)[23-25]

图4 0.5% GO浓度的JP-10静置0 h(a)、3 h(b)、和2周(c)时的照片[41]
Fig.4 Photographs of GO dispersed at a concentration of 0.5% in JP-10 with different time: (a) 0 h; (b) 3 h; (c) 2 weeks[41]
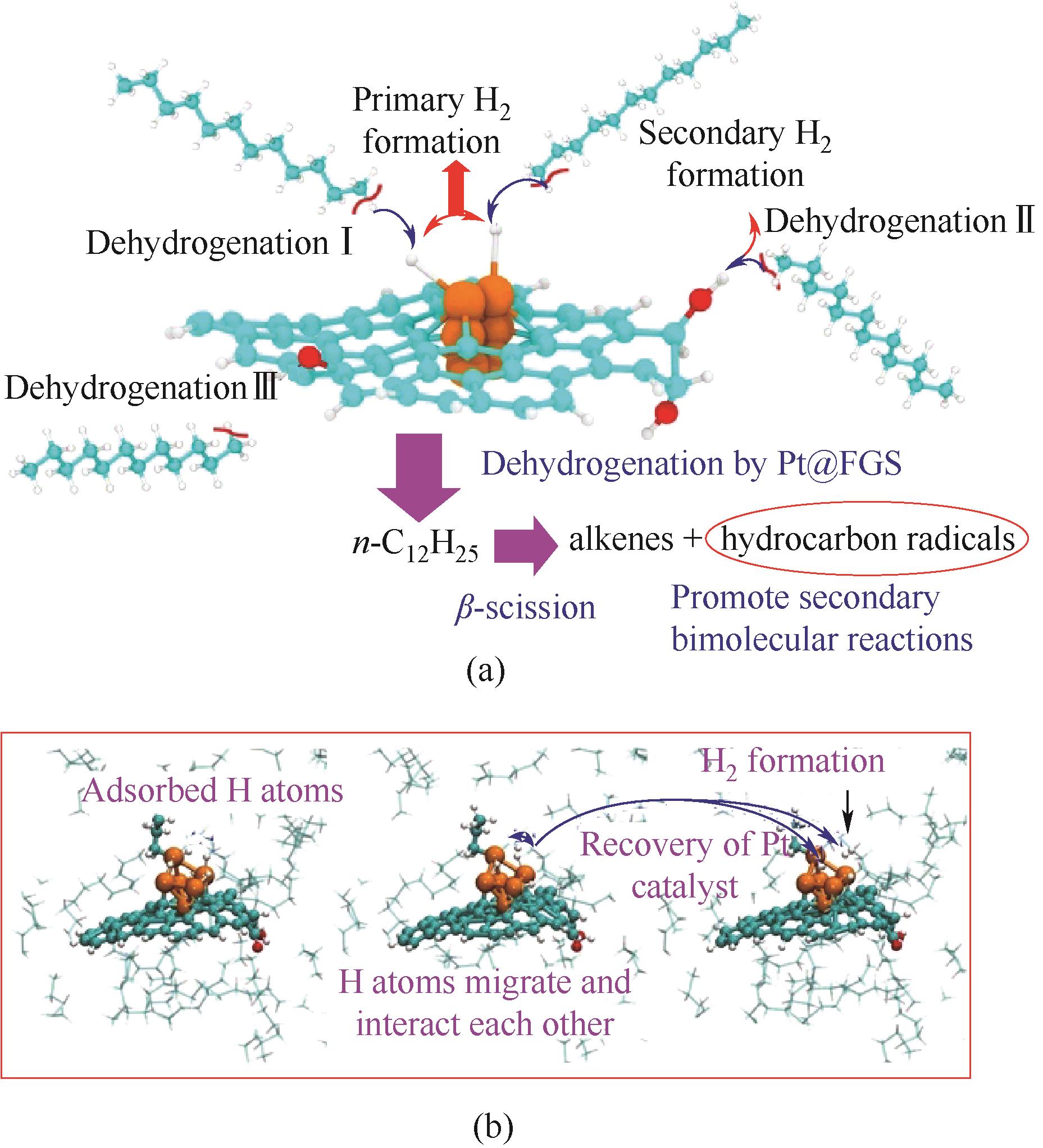
图5 (a)Pt@FGS提高n-C12H26分解机理示意图;(b)Pt@FGS表面H2生成和Pt团簇复原的分子动力学示意图(青色:碳;白色:氢;红色:氧;橙色:铂)[59]
Fig.5 (a) Schematic of the proposed enhancing mechanisms of n-C12H26 fuel decomposition in the presence of Pt@FGS;(b) MD snapshots for an example of H2 formation from the surface of Pt@FGS and the recovery of Pt-cluster on the FGS (cyan: C, white: H, red: O, and orange: Pt)[59]
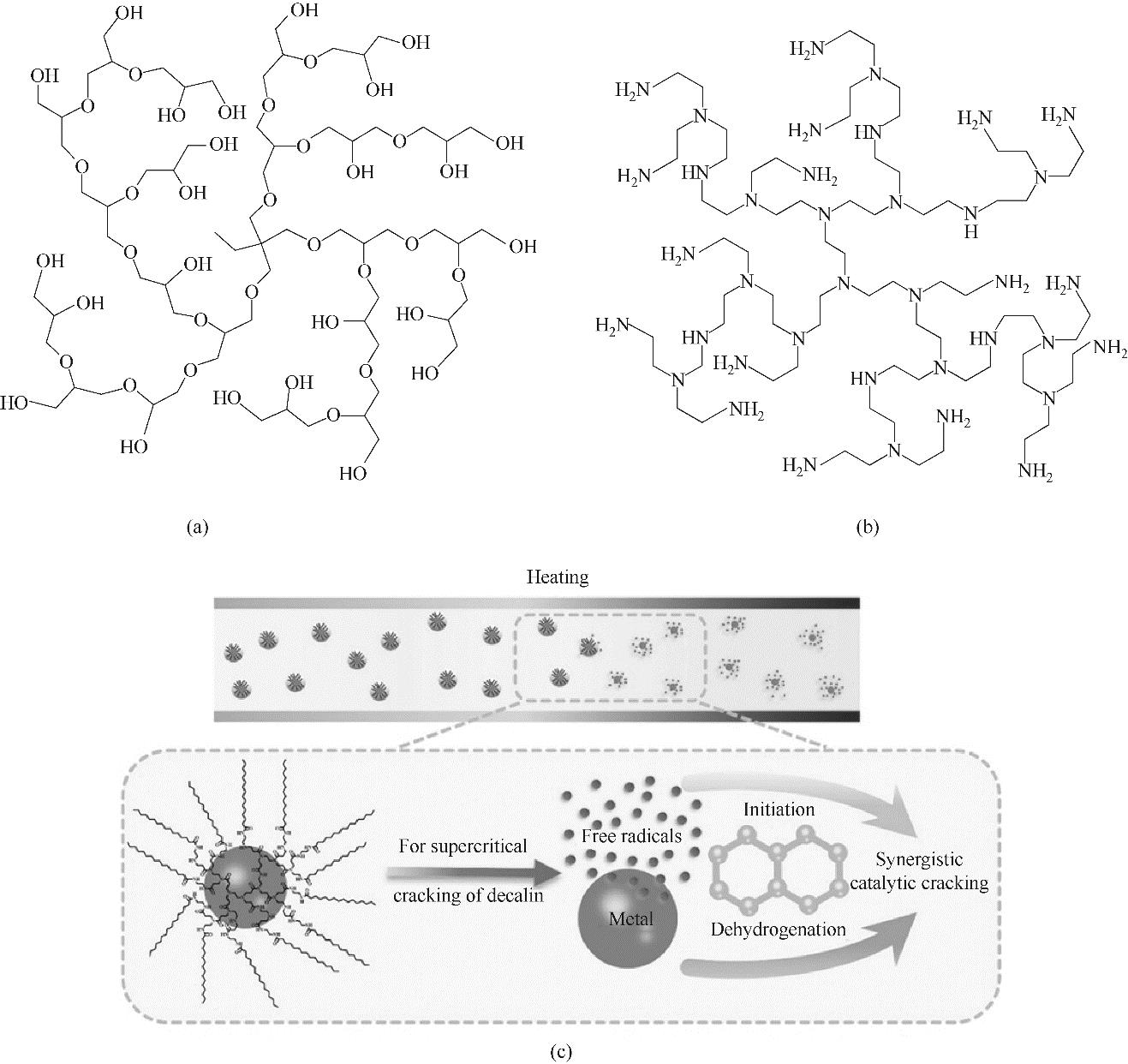
图7 超支化聚甘油(HMS)[61](a),超支化聚乙烯亚胺(HPEI)[50](b)和超支化聚合物包覆金属纳米颗粒协同催化十氢萘裂解的示意图[40](c)
Fig.7 Schematic of hyperbranched polyglycerol (HMS)[61] (a), hyperbranched polyethyleneimine (HPEI)[50] (b), and the synergistic catalytic cracking of decalin by hyperbranched poly(amidoamine)-encapsulated metal nanoparticles (HEMNs)[40] (c)
| 文献 | 纳米颗粒 | 燃料基液 | 温度/℃ | 压力/MPa | 转化率/% | 热沉/(MJ/kg) |
|---|---|---|---|---|---|---|
| [ | 钯 | 十氢萘 | 750 | 3.50 | 91.9 | 3.50 |
| [ | 铂 | JP-10 | 680 | 4.00 | 55.2 | 2.70 |
| [ | 铂 | 十氢萘 | 675 | 3.50 | 50.7 | 2.62 |
| [ | 钯 | 十氢萘 | 725 | 4.00 | 77.0 | 3.61 |
| [ | 铂 | 甲基环己烷 | 650 | 3.50 | 33.0 | 2.24 |
| [ | 铂 | 甲基环己烷 | 650 | 3.50 | 61.5 | 2.39 |
表1 贵金属纳米流体燃料裂解性能
Table 1 Cracking performance of precious metal nanofluid fuels
| 文献 | 纳米颗粒 | 燃料基液 | 温度/℃ | 压力/MPa | 转化率/% | 热沉/(MJ/kg) |
|---|---|---|---|---|---|---|
| [ | 钯 | 十氢萘 | 750 | 3.50 | 91.9 | 3.50 |
| [ | 铂 | JP-10 | 680 | 4.00 | 55.2 | 2.70 |
| [ | 铂 | 十氢萘 | 675 | 3.50 | 50.7 | 2.62 |
| [ | 钯 | 十氢萘 | 725 | 4.00 | 77.0 | 3.61 |
| [ | 铂 | 甲基环己烷 | 650 | 3.50 | 33.0 | 2.24 |
| [ | 铂 | 甲基环己烷 | 650 | 3.50 | 61.5 | 2.39 |
| 文献 | 纳米颗粒 | 燃料基液 | 温度/℃ | 压力/MPa | 转化率/% | 热沉/(MJ/kg) |
|---|---|---|---|---|---|---|
| [ | Beta分子筛 | JP-10 | 700 | 4.00 | 63.2 | 2.80 |
| [ | ZSM-5 | 正癸烷 | 758 | 3.00 | — | 4.64 |
表2 分子筛纳米流体燃料裂解性能
Table 2 Cracking performance of molecular sieve nanofluid fuels
| 文献 | 纳米颗粒 | 燃料基液 | 温度/℃ | 压力/MPa | 转化率/% | 热沉/(MJ/kg) |
|---|---|---|---|---|---|---|
| [ | Beta分子筛 | JP-10 | 700 | 4.00 | 63.2 | 2.80 |
| [ | ZSM-5 | 正癸烷 | 758 | 3.00 | — | 4.64 |

图11 ReaxFF-MD模拟得到含氧FGS催化脱氢的反应路径:(a)去质子化/质子化,(b)再生,(c)1700 K下含有FGS的MCH保持6.0 ns的反应路径[90]
Fig.11 Reaction pathways for catalytic dehydrogenation by the oxygen-containing FGS, derived by ReaxFF MD simulations: (a) deprotonation/protonation, (b) regeneration, (c) reaction pathways for MCH containing added FGS held at 1700 K for 6.0 ns[90]
| 文献 | 纳米颗粒 | 燃料基液 | 温度/℃ | 压力/MPa | 转化率/% | 热沉/(MJ/kg) |
|---|---|---|---|---|---|---|
| [ | Pt@FGS | JP-10 | 420 | 4.00 | 48.5 | — |
| [ | FGS | 甲基环己烷 | 547 | 4.70 | 64.2 | — |
表3 石墨烯纳米流体燃料裂解性能
Table 3 Cracking performance of graphene nanofluid fuels
| 文献 | 纳米颗粒 | 燃料基液 | 温度/℃ | 压力/MPa | 转化率/% | 热沉/(MJ/kg) |
|---|---|---|---|---|---|---|
| [ | Pt@FGS | JP-10 | 420 | 4.00 | 48.5 | — |
| [ | FGS | 甲基环己烷 | 547 | 4.70 | 64.2 | — |
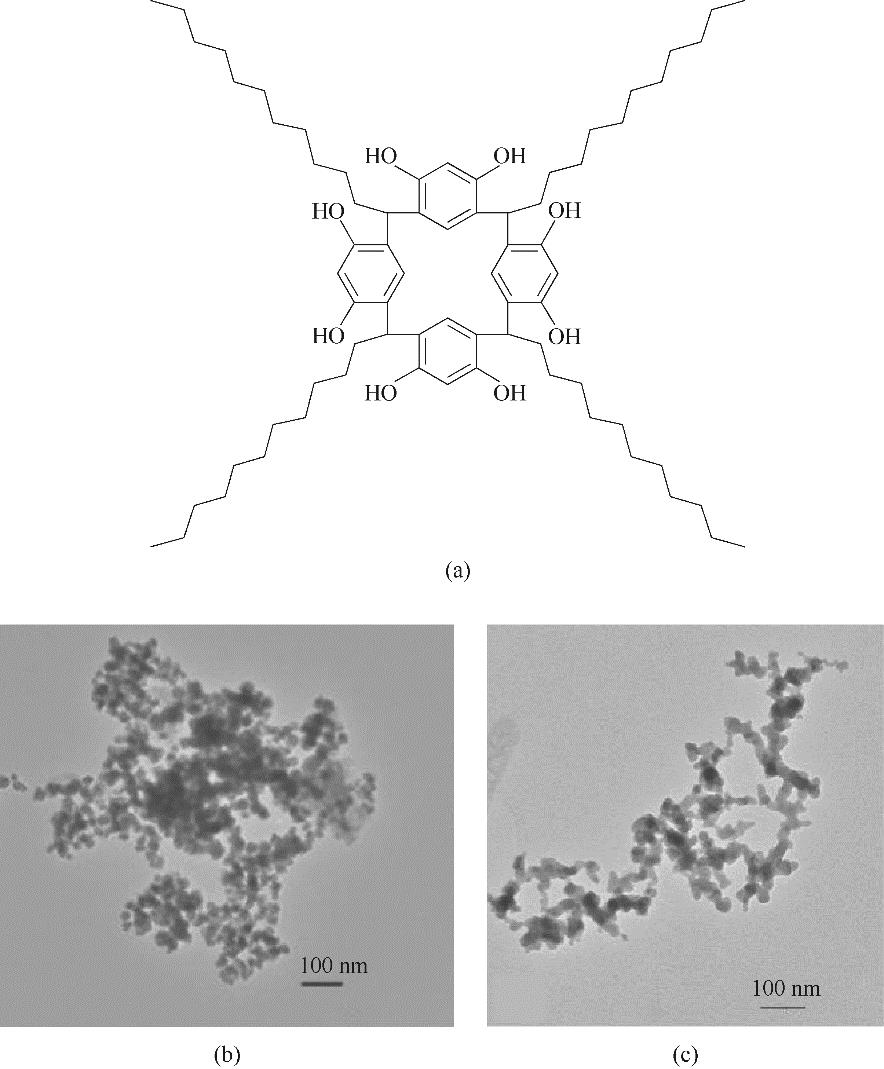
图13 C-十一烷基杯[4]烃间苯二酚分子结构示意图(a); C-十一烷基杯[4]烃间苯二酚包覆镍纳米颗粒(b)和Ni-B非晶合金(c)的TEM图[93-94]
Fig.13 Molecular structure of C-undecyl calix[4]resorcinarene (a); TEM images of C-undecyl calix[4]resorcinarene-encapsulated Ni nanoparticles (b) and Ni-B nano-amorphous alloy (c)[93-94]
| 文献 | 纳米颗粒 | 燃料基液 | 温度/℃ | 压力/MPa | 转化率/% | 热沉/(MJ/kg) |
|---|---|---|---|---|---|---|
| [ | Ni/SiO2-A | 正癸烷 | 750 | 3.50 | 84.0 | 3.93 |
| [ | Ni/SiO2 | 正癸烷 | 750 | 3.50 | — | 4.04 |
| [ | ZSM-5@NiM | 正癸烷 | 780 | 3.00 | — | 4.59 |
表4 过渡金属纳米流体燃料裂解性能
Table 4 Cracking performance of transition metal nanofluid fuels
| 文献 | 纳米颗粒 | 燃料基液 | 温度/℃ | 压力/MPa | 转化率/% | 热沉/(MJ/kg) |
|---|---|---|---|---|---|---|
| [ | Ni/SiO2-A | 正癸烷 | 750 | 3.50 | 84.0 | 3.93 |
| [ | Ni/SiO2 | 正癸烷 | 750 | 3.50 | — | 4.04 |
| [ | ZSM-5@NiM | 正癸烷 | 780 | 3.00 | — | 4.59 |
| 1 | Edwards T. Liquid fuels and propellants for aerospace propulsion: 1903—2003[J]. Journal of Propulsion and Power, 2003, 19(6): 1089-1107. |
| 2 | Edwards T. Cracking and deposition behavior of supercritical hydrocarbon aviation fuels[J]. Combustion Science and Technology, 2006, 178(1/2/3): 307-334. |
| 3 | Dinda S, Vuchuru K, Konda S, et al. Heat management in supersonic/hypersonic vehicles using endothermic fuel: perspective and challenges[J]. ACS Omega, 2021, 6(40): 26741-26755. |
| 4 | 贾贞健, 周伟星, 黄洪雁, 等. 碳氢燃料热裂解与引发裂解换热对比实验[J]. 化工学报, 2014, 65(S1): 138-143. |
| Jia Z J, Zhou W X, Huang H Y, et al. Heat transfer of thermal cracking and initiated cracking of aviation kerosene[J]. CIESC Journal, 2014, 65(S1): 138-143. | |
| 5 | 侯淋, 刘青, 张香文. 面向高超声速飞行的碳氢燃料吸热反应研究进展[J]. 化学工业与工程, 2022, 39(5): 1-10. |
| Hou L, Liu Q, Zhang X W. Progress on endothermic reactions of hydrocarbon fuel for hypersonic flight[J]. Chemical Industry and Engineering, 2022, 39(5): 1-10. | |
| 6 | Xu Q, Lin G W, Li H W. Evaluation model for fast convective heat transfer characteristics of thermal cracked hydrocarbon fuel regenerative cooling channel in hydrocarbon-fueled scramjet[J]. Applied Thermal Engineering, 2021, 199: 117616. |
| 7 | Li H, Liu G Z, Jiang R P, et al. Experimental and kinetic modeling study of exo-TCD pyrolysis under low pressure[J]. Combustion and Flame, 2015, 162(5): 2177-2190. |
| 8 | Huang S Z, Ruan B, Gao X W, et al. Numerical investigation on supercritical turbulent heat transfer of copper/n-decane nanofluid inside a miniature tube[J]. Numerical Heat Transfer, Part A: Applications, 2017, 72(12): 921-935. |
| 9 | Yue L, Lu X X, Chi H, et al. Heat-sink enhancement of decalin and aviation kerosene prepared as nanofluids with palladium nanoparticles[J]. Fuel, 2014, 121: 149-156. |
| 10 | Wang L, Diao Z H, Tian Y J, et al. Catalytic cracking of endothermic hydrocarbon fuels over ordered meso-HZSM-5 zeolites with Al-MCM-41 shells[J]. Energy & Fuels, 2016, 30(9): 6977-6983. |
| 11 | E X T F, Zhi X M, Zhang X W, et al. Ignition and combustion performances of high-energy-density jet fuels catalyzed by Pt and Pd nanoparticles[J]. Energy & Fuels, 2018, 32(2): 2163-2169. |
| 12 | Li S J, Yang Q M, Ye L H, et al. Effect of nanoparticle concentration on physical and heat-transfer properties and evaporation characteristics of graphite/n-decane nanofluid fuels[J]. ACS Omega, 2022, 7(4): 3284-3292. |
| 13 | Aboalhamayie A, Festa L, Ghamari M. Evaporation rate of colloidal droplets of jet fuel and carbon-based nanoparticles: effect of thermal conductivity[J]. Nanomaterials, 2019, 9(9): 1297. |
| 14 | 吴熙, 何桂金, 郭永胜, 等. 超支化聚合物稳定的JP-10基铂纳米流体燃料的制备与催化燃烧性能[J]. 含能材料, 2020, 28(5): 369-375. |
| Wu X, He G J, Guo Y S, et al. Preparation and catalytic combustion of JP-10 based Pt nanofluid fuels stabilized by hyperbranched polymer[J]. Chinese Journal of Energetic Materials, 2020, 28(5): 369-375. | |
| 15 | Van Devener B, Anderson S L. Breakdown and combustion of JP-10 fuel catalyzed by nanoparticulate CeO2 and Fe2O3 [J]. Energy & Fuels, 2006, 20(5): 1886-1894. |
| 16 | Bello M N, Pantoya M L, Kappagantula K, et al. Reaction dynamics of rocket propellant with magnesium oxide nanoparticles[J]. Energy & Fuels, 2015, 29(9): 6111-6117. |
| 17 | Ghadimi A, Saidur R, Metselaar H S C. A review of nanofluid stability properties and characterization in stationary conditions[J]. International Journal of Heat and Mass Transfer, 2011, 54(17/18): 4051-4068. |
| 18 | Meyer J P, Adio S A, Sharifpur M, et al. The viscosity of nanofluids: a review of the theoretical, empirical, and numerical models[J]. Heat Transfer Engineering, 2016, 37(5): 387-421. |
| 19 | Patra A K, Nayak M K, Misra A. Viscosity of nanofluids—a review[J]. International Journal of Thermofluid Science and Technology, 2020, 7(2): 070202. |
| 20 | Lee S, Choi S U S, Li S, et al. Measuring thermal conductivity of fluids containing oxide nanoparticles[J]. Journal of Heat Transfer, 1999, 121(2): 280-289. |
| 21 | Choi S U S, Eastman J A. Enhancing thermal conductivity of fluids with nanoparticles[C]//1995 International Mechanical Engineering Congress and Exhibition. No. ANL/MSD/CP-84938; CONF-951135- 29. San Francisco, United States: Argonne National Laboratory, 1995. |
| 22 | Ojha P K, Karmakar S. Boron for liquid fuel engines—a review on synthesis, dispersion stability in liquid fuel, and combustion aspects[J]. Progress in Aerospace Sciences, 2018, 100: 18-45. |
| 23 | Popa I, Gillies G, Papastavrou G, et al. Attractive and repulsive electrostatic forces between positively charged latex particles in the presence of anionic linear polyelectrolytes[J]. The Journal of Physical Chemistry. B, 2010, 114(9): 3170-3177. |
| 24 | Missana T, Adell A. On the applicability of DLVO theory to the prediction of clay colloids stability[J]. Journal of Colloid and Interface Science, 2000, 230(1): 150-156. |
| 25 | Zhu D S, Li X F, Wang N, et al. Dispersion behavior and thermal conductivity characteristics of Al2O3-H2O nanofluids[J]. Current Applied Physics, 2009, 9(1): 131-139. |
| 26 | Hotze E M, Phenrat T, Lowry G V. Nanoparticle aggregation: challenges to understanding transport and reactivity in the environment[J]. Journal of Environmental Quality, 2010, 39(6): 1909-1924. |
| 27 | E X T F, Zhang Y, Zou J J, et al. Oleylamine-protected metal (Pt, Pd) nanoparticles for pseudohomogeneous catalytic cracking of JP-10 jet fuel[J]. Industrial & Engineering Chemistry Research, 2014, 53(31): 12312-12318. |
| 28 | Hwang Y, Lee J K, Lee J K, et al. Production and dispersion stability of nanoparticles in nanofluids[J]. Powder Technology, 2008, 186(2): 145-153. |
| 29 | Albin J, Shijo T. Energy, exergy and corrosion analysis of direct absorption solar collector employed with ultra-high stable carbon quantum dot nanofluid[J]. Renewable Energy, 2022, 181: 725-737. |
| 30 | 夏国栋, 周明正, 周利军, 等. Ag纳米流体浸没射流冲击换热特性[J]. 化工学报, 2011, 62(4): 916-921. |
| Xia G D, Zhou M Z, Zhou L J, et al. Heat transfer of sub-merged jet impingement on pin-fin heat sinks with silver nanofluid[J]. CIESC Journal, 2011, 62(4): 916-921. | |
| 31 | Parsa M S, Yazdani A, Aberoumand H, et al. A critical analysis on the energy and exergy performance of photovoltaic/thermal (PV/T) system: the role of nanofluids stability and synthesizing method[J]. Sustainable Energy Technologies and Assessments, 2022, 51: 101887. |
| 32 | Chang H, Chang Y C. Fabrication of Al2O3 nanofluid by a plasma arc nanoparticles synthesis system[J]. Journal of Materials Processing Technology, 2008, 207(1/2/3): 193-199. |
| 33 | Smirnov V V, Kostritsa S A, Kobtsev V D, et al. Experimental study of combustion of composite fuel comprising n-decane and aluminum nanoparticles[J]. Combustion and Flame, 2015, 162(10): 3554-3561. |
| 34 | Amrute A P, De Bellis J, Felderhoff M, et al. Mechanochemical synthesis of catalytic materials[J]. Chemistry, 2021, 27(23): 6819-6847. |
| 35 | Li S J, Du H Z, Zhuo Z, et al. Dispersion stability, physical properties, and electrostatic breakup of surfactant-loaded aluminum/n-decane nanofluid fuel: nanoparticle size effect[J]. Energy & Fuels, 2020, 34(1): 1082-1092. |
| 36 | Brotton S J, Malek M J, Anderson S L, et al. Effects of acetonitrile-assisted ball-milled aluminum nanoparticles on the ignition of acoustically levitated exo-tetrahydrodicyclopentadiene (JP-10) droplets[J]. Chemical Physics Letters, 2020, 754: 137679. |
| 37 | Brust M, Walker M, Bethell D, et al. Synthesis of thiol-derivatised gold nanoparticles in a two-phase liquid-liquid system[J]. J. Chem. Soc., Chem. Commun., 1994(7): 801-802. |
| 38 | E X T F, Zhang Y, Zou J J, et al. Shape evolution in Brust-Schiffrin synthesis of Au nanoparticles[J]. Materials Letters, 2014, 118: 196-199. |
| 39 | Li D, Fang W J, Wang H Q, et al. Gold/oil nanofluids stabilized by a gemini surfactant and their catalytic property[J]. Industrial & Engineering Chemistry Research, 2013, 52(24): 8109-8113. |
| 40 | Ye D F, Zhao L, Bai S S, et al. New strategy for high-performance integrated catalysts for cracking hydrocarbon fuels[J]. ACS Applied Materials & Interfaces, 2019, 11(43): 40078-40090. |
| 41 | Liu G Z, Jia X K, Tian Y J, et al. Preparations and remarkable catalytic cracking performances of Pt@FGS/JP-10 nanofluids[J]. Fuel, 2019, 252: 228-237. |
| 42 | Balanta A, Godard C, Claver C. Pd nanoparticles for C-C coupling reactions[J]. Chemical Society Reviews, 2011, 40(10): 4973-4985. |
| 43 | Jeyakumar N, Narayanasamy B. Investigation of performance, emission, combustion characteristics of municipal waste plastic oil fueled diesel engine with nano fluids[J]. Energy Sources, Part A: Recovery, Utilization, and Environmental Effects, 2020: 1745958. |
| 44 | Praveena V, Martin M L J, Geo V E. Experimental characterization of CI engine performance, combustion and emission parameters using various metal oxide nanoemulsion of grapeseed oil methyl ester[J]. Journal of Thermal Analysis and Calorimetry, 2020, 139(6): 3441-3456. |
| 45 | E X T F, Pan L, Wang F, et al. Al-nanoparticle-containing nanofluid fuel: synthesis, stability, properties, and propulsion performance[J]. Industrial & Engineering Chemistry Research, 2016, 55(10): 2738-2745. |
| 46 | Piacenza E, Presentato A, Turner R J. Stability of biogenic metal(loid) nanomaterials related to the colloidal stabilization theory of chemical nanostructures[J]. Critical Reviews in Biotechnology, 2018, 38(8): 1137-1156. |
| 47 | Pelaz B, del Pino P, Maffre P, et al. Surface functionalization of nanoparticles with polyethylene glycol: effects on protein adsorption and cellular uptake[J]. ACS Nano, 2015, 9(7): 6996-7008. |
| 48 | Ao W, Gao Y, Zhou S, et al. Enhancing the stability and combustion of a nanofluid fuel with polydopamine-coated aluminum nanoparticles[J]. Chemical Engineering Journal, 2021, 418: 129527. |
| 49 | Zhang R Q, Zhai Z W, Wang L, et al. Heat sink enhancement of decalin by symmetrical imidazolium ionic liquid-capped metal nanoparticles[J]. Energy & Fuels, 2023, 37(9): 6545-6557. |
| 50 | Wu X, Ye D F, Jin S D, et al. Cracking of platinum/hydrocarbon nanofluids with hyperbranched polymer as stabilizer and initiator[J]. Fuel, 2019, 255: 115782. |
| 51 | Qin X M, Yang S H, Chen Y P, et al. Thermal conductivity and stability of hydrocarbon-based nanofluids with palladium nanoparticles dispersed by modified hyperbranched polyglycerol[J]. ACS Omega, 2020, 5(48): 31156-31163. |
| 52 | Liu Q, Jia T H, Pan L, et al. Relationship between molecular structure and pyrolysis performance for high-energy-density fuels[J]. Fuel, 2024, 358: 130342. |
| 53 | Vandewiele N M, Magoon G R, Van Geem K M, et al. Experimental and modeling study on the thermal decomposition of jet propellant-10[J]. Energy & Fuels, 2014, 28(8): 4976-4985. |
| 54 | 岳磊. 超临界条件下碳氢燃料的裂解及其热化学过程调控[D]. 杭州: 浙江大学, 2015. |
| Yue L. Cracking and thermochemical process regulation of hydrocarbon fuels under supercritical conditions[D]. Hangzhou: Zhejiang University, 2015. | |
| 55 | Zhao H L, Meng F X, Guo W, et al. Pd/HZSM-5 coating catalyst for supercritical cracking of endothermic fuel[J]. Journal of Fuel Chemistry and Technology, 2008, 36(4): 462-467. |
| 56 | Zhang J, Cheng M, Jiao Y, et al. Enhanced cracking conversion of supercritical fuel over stabilized Pt nanoparticles supported SiO2-Al2O3 catalyst coating[J]. Applied Surface Science, 2021, 559: 149950. |
| 57 | Rahimi N, Karimzadeh R. Catalytic cracking of hydrocarbons over modified ZSM-5 zeolites to produce light olefins: a review[J]. Applied Catalysis A: General, 2011, 398(1/2): 1-17. |
| 58 | Krumeich F, Ihli J, Shu Y Y, et al. Structural changes in deactivated fluid catalytic cracking catalysts determined by electron microscopy[J]. ACS Catalysis, 2018, 8(5): 4591-4599. |
| 59 | Sim H S, Yetter R A, Hong S, et al. Enhanced fuel decomposition in the presence of colloidal functionalized graphene sheet-supported platinum nanoparticles[J]. ACS Applied Energy Materials, 2020, 3(8): 7637-7648. |
| 60 | Yue L, Wu J Z, Gong Y, et al. Thermodynamic properties and pyrolysis performances of hydrocarbon-fuel-based nanofluids containing palladium nanoparticles[J]. Journal of Analytical and Applied Pyrolysis, 2016, 120: 347-355. |
| 61 | Wu X, Chen X Y, Jin S D, et al. Highly stable macroinitiator/platinum/hydrocarbon nanofluids for efficient thermal management in hypersonic aircraft from synergistic catalysis[J]. Energy Conversion and Management, 2019, 198: 111797. |
| 62 | Zhang Q H, Zhang S G, Deng Y Q. Recent advances in ionic liquid catalysis[J]. Green Chemistry, 2011, 13(10): 2619-2637. |
| 63 | Migowski P, Dupont J. Catalytic applications of metal nanoparticles in imidazolium ionic liquids[J]. Chemistry, 2007, 13(1): 32-39. |
| 64 | Jeong B J, Shin M C, Park J H, et al. Catalytic cracking of methylcyclohexane over H-ZSM-5 zeolite and activated charcoal wall-coated microchannel reactor with wavy structure under supercritical conditions[J]. Journal of Industrial and Engineering Chemistry, 2022, 105: 502-512. |
| 65 | 白宇恩, 张彬瑞, 刘东阳, 等. ZSM-5分子筛酸性能和孔结构的协同作用对C5烯烃催化裂解性能的影响[J]. 化工学报, 2023, 74(1): 438-448. |
| Bai Y E, Zhang B R, Liu D Y, et al. Influence of synergistic effect of acid properties and pore structure of ZSM-5 zeolite on the catalytic cracking performance of pentene[J]. CIESC Journal, 2023, 74(1): 438-448. | |
| 66 | 汪然明. HZSM-5和Y型分子筛催化正十二烷超临界裂解研究[D]. 天津: 天津大学, 2010. |
| Wang R M. Supercritical catalytic cracking of n-dodecane on HZSM-5 and Y zeolites[D]. Tianjin: Tianjin University, 2010. | |
| 67 | Bao S G, Liu G Z, Wang L, et al. Preparation and properties of hydrocarbon dispersible HZSM-5 nanocrystals for quasi-homogeneous catalytic cracking of n-dodecane[J]. Microporous and Mesoporous Materials, 2011, 143(2/3): 458-466. |
| 68 | Iwase Y, Sakamoto Y, Shiga A, et al. Shape-selective catalysis determined by the volume of a zeolite cavity and the reaction mechanism for propylene production by the conversion of butene using a proton-exchanged zeolite[J]. The Journal of Physical Chemistry C, 2012, 116(8): 5182-5196. |
| 69 | Tian Y J, Zhang B F, Liang H R, et al. Synthesis and performance of pillared HZSM-5 nanosheet zeolites for n-decane catalytic cracking to produce light olefins[J]. Applied Catalysis A: General, 2019, 572: 24-33. |
| 70 | Castro-Marcano F, van Duin A C T. Comparison of thermal and catalytic cracking of 1-heptene from ReaxFF reactive molecular dynamics simulations[J]. Combustion and Flame, 2013, 160(4): 766-775. |
| 71 | Corma A, González-Alfaro V, Orchillés A V. Decalin and tetralin as probe molecules for cracking and hydrotreating the light cycle oil[J]. Journal of Catalysis, 2001, 200(1): 34-44. |
| 72 | Kubička D, Kumar N, Mäki-Arvela P, et al. Ring opening of decalin over zeolites(Ⅰ): Activity and selectivity of proton-form zeolites[J]. Journal of Catalysis, 2004, 222(1): 65-79. |
| 73 | Kubička D, Kumar N, Mäki-Arvela P, et al. Ring opening of decalin over zeolitesII. Activity and selectivity of platinum-modified zeolites[J]. Journal of Catalysis, 2004, 227(2): 313-327. |
| 74 | Bao S G, Liu G Z, Zhang X W, et al. New method of catalytic cracking of hydrocarbon fuels using a highly dispersed nano-HZSM-5 catalyst[J]. Industrial & Engineering Chemistry Research, 2010, 49(8): 3972-3975. |
| 75 | 韩之雄. 正癸烷微纳米催化裂解流动传热实验研究[D]. 哈尔滨: 哈尔滨工业大学, 2016. |
| Han Z X. Experimental investigation on flow and heat transfer of n-decane micro/nanocatalytic pyrolysis[D]. Harbin: Harbin Institute of Technology, 2016. | |
| 76 | Bao S G, Liu G Z, Wang L, et al. Quasi-homogeneous catalytic activities of hydrocarbon dispersible HZSM-5 nanocrystals grafted with different alkyl groups[J]. Applied Catalysis A: General, 2011, 405(1/2): 61-68. |
| 77 | 孙文娟. 可控自组装制备多级孔Beta纳米分子筛及其催化性能研究[D]. 天津: 天津大学, 2015. |
| Sun W J. Synthesis and catalytic performance of hierarchical beta nanozeolites through controllable self-assembly[D]. Tianjin: Tianjin University, 2015. | |
| 78 | Sun W J, Liu G Z, Wang L, et al. Quasi-homogeneous catalytic cracking of JP-10 over high hydrocarbon dispersible nanozeolites[J]. Fuel, 2015, 144: 96-102. |
| 79 | Seo Y, Cho K, Jung Y, et al. Characterization of the surface acidity of MFI zeolite nanosheets by 31P NMR of adsorbed phosphine oxides and catalytic cracking of decalin[J]. ACS Catalysis, 2013, 3(4): 713-720. |
| 80 | Tian Y J, Qiu Y, Hou X, et al. Catalytic cracking of JP-10 over HZSM-5 nanosheets[J]. Energy & Fuels, 2017, 31(11): 11987-11994. |
| 81 | Xing Y, Li D, Xie W J, et al. Catalytic cracking of tricyclo[5.2.1.02.6]decane over HZSM-5 molecular sieves[J]. Fuel, 2010, 89(7): 1422-1428. |
| 82 | Kissin Y V. Chemical mechanisms of catalytic cracking over solid acidic catalysts: alkanes and alkenes[J]. Catalysis Reviews, 2001, 43(1/2): 85-146. |
| 83 | 阚仁俊, 达志坚, 张久顺, 等. 十氢萘催化裂解制低碳烯烃研究进展[J]. 石油炼制与化工, 2022, 53(7): 112-121. |
| Kan R J, Da Z J, Zhang J S, et al. Research progress in deep catalytic cracking of decalin to low-carbon olefins[J]. Petroleum Processing and Petrochemicals, 2022, 53(7): 112-121. | |
| 84 | Sang Y, Jiao Q Z, Li H S, et al. HZSM-5/MCM-41 composite molecular sieves for the catalytic cracking of endothermic hydrocarbon fuels: nano-ZSM-5 zeolites as the source[J]. Journal of Nanoparticle Research, 2014, 16(12): 2755. |
| 85 | 桑宇. HZSM-5分子筛结构调控及其催化性能研究[D]. 北京: 北京理工大学, 2015. |
| Sang Y. Structure controlling and catalytic performance of HZSM-5 zeolites[D]. Beijing: Beijing Institute of Technology, 2015. | |
| 86 | Li Y G, Xie W H, Yong S. The acidity and catalytic behavior of Mg-ZSM-5 prepared via a solid-state reaction[J]. Applied Catalysis A: General, 1997, 150(2): 231-242. |
| 87 | Long L, Lan Z Z, Han Z X, et al. Co3O4 nanosheet wrapped commercial HZSM-5 for promoting catalytic cracking of n-decane and anticoking activities[J]. ACS Applied Energy Materials, 2018, 1(8): 4130-4139. |
| 88 | Sadeghinezhad E, Mehrali M, Saidur R, et al. A comprehensive review on graphene nanofluids: recent research, development and applications[J]. Energy Conversion and Management, 2016, 111: 466-487. |
| 89 | Zhu Y W, Murali S, Cai W W, et al. Graphene and graphene oxide: synthesis, properties, and applications[J]. Advanced Materials, 2010, 22(35): 3906-3924. |
| 90 | Sim H S, Yetter R A, Hong S, et al. Functionalized graphene sheet as a dispersible fuel additive for catalytic decomposition of methylcyclohexane[J]. Combustion and Flame, 2020, 217: 212-221. |
| 91 | Wang Z, Lin R S, Guo Y S, et al. Tributylamine as an initiator for cracking of heptane[J]. Energy Conversion and Management, 2008, 49(6): 1584-1594. |
| 92 | Qi Y, Liu Q Q, Chen Z Y, et al. Nickel-Passivating element selection in FCC process and mechanistic study on the passivation of nickel by lanthanum and phosphorus[J]. Chemical Engineering Journal, 2023, 467: 143452. |
| 93 | Guo Y S, Yang Y Z, Fang W J, et al. Resorcinarene-encapsulated Ni-B nano-amorphous alloys for quasi-homogeneous catalytic cracking of JP-10[J]. Applied Catalysis A: General, 2014, 469: 213-220. |
| 94 | Guo Y S, Yang Y Z, Xiao J, et al. A novel well-dispersed nano-Ni catalyst for endothermic reaction of JP-10[J]. Fuel, 2014, 117: 932-938. |
| 95 | Herbinet O, Sirjean B, Bounaceur R, et al. Primary mechanism of the thermal decomposition of tricyclodecane[J]. The Journal of Physical Chemistry. A, 2006, 110(39): 11298-11314. |
| 96 | Wang L L, Chen T, Zhang J, et al. High catalytic activity and stability quasi homogeneous alkali metal promoted Ni/SiO2 aerogel catalysts for catalytic cracking of n-decane[J]. Fuel, 2020, 268: 117384. |
| 97 | Wang L L, Liu L, Gong X L, et al. Combined strategy and Ni NPs/SiO2 aerogel catalyst for cracking hydrocarbon fuels[J]. Journal of Power Sources, 2021, 506: 230172. |
| 98 | Shen Z B, Ke M, Yu P, et al. Catalytic activities of Mo-modified Ni/Al2O3 catalysts for thioetherification of mercaptans and di-olefins in fluid catalytic cracking naphtha[J]. Transition Metal Chemistry, 2012, 37(6): 587-593. |
| 99 | Long L, Zhou W X, Qiu Y F, et al. Coking and gas products behavior of supercritical n-decane over NiO nanoparticle/nanosheets modified HZSM-5[J]. Energy, 2020, 192: 116540. |
| [1] | 冯彬彬, 卢明佳, 黄志宏, 常译文, 崔志明. 碳载体在质子交换膜燃料电池中的应用及优化[J]. 化工学报, 2024, 75(4): 1469-1484. |
| [2] | 贾旭东, 杨博龙, 程前, 李雪丽, 向中华. 分步负载金属法制备铁钴双金属位点高效氧还原电催化剂[J]. 化工学报, 2024, 75(4): 1578-1593. |
| [3] | 张劲, 郭志斌, 罗来明, 卢善富, 相艳. 5 kW重整甲醇高温质子交换膜燃料电池系统设计与性能[J]. 化工学报, 2024, 75(4): 1697-1704. |
| [4] | 孙铭泽, 黄鹤来, 牛志强. 铂基氧还原催化剂:从单晶电极到拓展表面纳米材料[J]. 化工学报, 2024, 75(4): 1256-1269. |
| [5] | 张领先, 刘斌, 邓琳, 任宇航. 基于改进TSO优化Xception的PEMFC故障诊断[J]. 化工学报, 2024, 75(3): 945-955. |
| [6] | 谭耀文, 姜攀星, 杜青, 余婉秋, 温小飞, 詹志刚. 工作电压对PEMFC膜电极衰退影响模拟研究[J]. 化工学报, 2024, 75(3): 974-986. |
| [7] | 陈宏, 江坤, 唐廷江, 黄易元, 池滨, 廖世军. 大功率质子交换膜燃料电池电堆膜电极一致性研究[J]. 化工学报, 2024, 75(2): 637-646. |
| [8] | 余洋, 罗祎青, 魏荣辉, 张文慧, 袁希钢. 考虑节点中断风险的弹性供应链设计方法[J]. 化工学报, 2024, 75(1): 338-353. |
| [9] | 赵文琪, 邓燕君, 朱春英, 付涛涛, 马友光. 纳米粒子稳定的Pickering乳液及其液滴聚并动力学研究进展[J]. 化工学报, 2024, 75(1): 33-46. |
| [10] | 李艺彤, 郭航, 陈浩, 叶芳. 催化剂非均匀分布的质子交换膜燃料电池操作条件研究[J]. 化工学报, 2023, 74(9): 3831-3840. |
| [11] | 陈哲文, 魏俊杰, 张玉明. 超临界水煤气化耦合SOFC发电系统集成及其能量转化机制[J]. 化工学报, 2023, 74(9): 3888-3902. |
| [12] | 仪显亨, 周骛, 蔡小舒, 蔡天意. 光纤后向动态光散射测量纳米颗粒的浓度适用范围研究[J]. 化工学报, 2023, 74(8): 3320-3328. |
| [13] | 杨欣, 彭啸, 薛凯茹, 苏梦威, 吴燕. 分子印迹-TiO2光电催化降解增溶PHE废水性能研究[J]. 化工学报, 2023, 74(8): 3564-3571. |
| [14] | 涂玉明, 邵高燕, 陈健杰, 刘凤, 田世超, 周智勇, 任钟旗. 钙基催化剂的设计合成及应用研究进展[J]. 化工学报, 2023, 74(7): 2717-2734. |
| [15] | 李勇, 高佳琦, 杜超, 赵亚丽, 李伯琼, 申倩倩, 贾虎生, 薛晋波. Ni@C@TiO2核壳双重异质结的构筑及光热催化分解水产氢[J]. 化工学报, 2023, 74(6): 2458-2467. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备 11010102001995号
京公网安备 11010102001995号