化工学报 ›› 2021, Vol. 72 ›› Issue (1): 534-542.DOI: 10.11949/0438-1157.20201041
王炼1,2( ),万超1,3,4,程党国1,2(
),万超1,3,4,程党国1,2( ),陈丰秋1,2,詹晓力1,2
),陈丰秋1,2,詹晓力1,2
收稿日期:2020-08-04
修回日期:2020-10-03
出版日期:2021-01-05
发布日期:2021-01-05
通讯作者:
程党国
作者简介:王炼(1995—),女,硕士研究生,基金资助:
WANG Lian1,2( ),WAN Chao1,3,4,CHENG Dangguo1,2(
),WAN Chao1,3,4,CHENG Dangguo1,2( ),CHEN Fengqiu1,2,ZHAN Xiaoli1,2
),CHEN Fengqiu1,2,ZHAN Xiaoli1,2
Received:2020-08-04
Revised:2020-10-03
Online:2021-01-05
Published:2021-01-05
Contact:
CHENG Dangguo
摘要:
采用溶胶凝胶法制镍锌铈(NiZnCe)复合氧化物催化剂,分别考察了NiZnCe复合氧化物催化剂中Zn和Ce含量对正丁烷氧化脱氢性能的影响。研究结果表明,NiZn0.5Ce0.3催化剂的转化率和烯烃选择性较高。采用H2-TPR、XPS及TPRO的表征方法来揭示催化剂中氧移动循环的不同步骤对催化剂性能的影响。表征结果显示,Ce元素的引入对催化剂的氧移动性产生较大的影响,且催化剂性能与氧容量(参与反应的催化剂中的氧量)呈正相关。此外,Ni与Ce的相互作用导致了Ni阳离子化合价和可接受电子能力的提高,活性Ni更容易吸附活性氧物种,金属与氧气的循环往复反应更活泼。
中图分类号:
王炼, 万超, 程党国, 陈丰秋, 詹晓力. NiZnCe复合氧化物的制备及其催化氧化正丁烷脱氢性能[J]. 化工学报, 2021, 72(1): 534-542.
WANG Lian, WAN Chao, CHENG Dangguo, CHEN Fengqiu, ZHAN Xiaoli. Preparation of NiZnCe composite oxide and its catalytic performance for dehydrogenation of n-butane[J]. CIESC Journal, 2021, 72(1): 534-542.
| 催化剂 | 正丁烷 转化率/% | 选择性/% | ||||||
|---|---|---|---|---|---|---|---|---|
| 二氧化物 | C1~C3 | 反-2-丁烯 | 顺-2-丁烯 | 1-丁烯 | 异丁烯 | 1,3-丁二烯 | ||
| NiO | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| NiZn1 | 10.5 | 74.0 | 4.5 | 5.0 | 4.0 | 10.4 | 0.2 | 1.9 |
| NiZn3 | 10.4 | 62.2 | 18.4 | 4.9 | 3.4 | 8.7 | 0.2 | 2.2 |
| NiZn5 | 7.7 | 51.4 | 24.8 | 6.4 | 4.3 | 11.7 | 0.3 | 1.1 |
| NiZn0.5 | 11.8 | 57.0 | 16.4 | 6.2 | 4.5 | 11.1 | 0.2 | 4.6 |
| NiZn0.5Ce0.2 | 22.0 | 66.1 | 2.7 | 5.4 | 4.7 | 12.7 | 0.08 | 8.3 |
| NiZn0.5Ce0.3 | 23.9 | 64.3 | 3.1 | 4.1 | 3.3 | 8.5 | 0.07 | 16.6 |
| NiZn0.5Ce0.5 | 19.2 | 59.0 | 8.7 | 5.3 | 3.4 | 6.9 | 0.1 | 16.6 |
| NiZn0.5Ce1 | 18.3 | 61.5 | 9.4 | 5.3 | 3.3 | 6.2 | 0.1 | 14.2 |
表1 NiZnx及NiZn0.5Cey氧化物催化剂上正丁烷转化率以及产物选择性
Table 1 n-Butane conversion and products selectivity over NiZnx and NiZn0.5Cey oxide catalyst
| 催化剂 | 正丁烷 转化率/% | 选择性/% | ||||||
|---|---|---|---|---|---|---|---|---|
| 二氧化物 | C1~C3 | 反-2-丁烯 | 顺-2-丁烯 | 1-丁烯 | 异丁烯 | 1,3-丁二烯 | ||
| NiO | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| NiZn1 | 10.5 | 74.0 | 4.5 | 5.0 | 4.0 | 10.4 | 0.2 | 1.9 |
| NiZn3 | 10.4 | 62.2 | 18.4 | 4.9 | 3.4 | 8.7 | 0.2 | 2.2 |
| NiZn5 | 7.7 | 51.4 | 24.8 | 6.4 | 4.3 | 11.7 | 0.3 | 1.1 |
| NiZn0.5 | 11.8 | 57.0 | 16.4 | 6.2 | 4.5 | 11.1 | 0.2 | 4.6 |
| NiZn0.5Ce0.2 | 22.0 | 66.1 | 2.7 | 5.4 | 4.7 | 12.7 | 0.08 | 8.3 |
| NiZn0.5Ce0.3 | 23.9 | 64.3 | 3.1 | 4.1 | 3.3 | 8.5 | 0.07 | 16.6 |
| NiZn0.5Ce0.5 | 19.2 | 59.0 | 8.7 | 5.3 | 3.4 | 6.9 | 0.1 | 16.6 |
| NiZn0.5Ce1 | 18.3 | 61.5 | 9.4 | 5.3 | 3.3 | 6.2 | 0.1 | 14.2 |
| 催化剂 | 原子比① | SBET/(m2·g-1) | 孔容/(cm3·g-1) | ||
|---|---|---|---|---|---|
| Ni | Zn | Ce | |||
| NiO | 1 | 0 | 0 | 2.7 | 0.008 |
| NiZn1 | 1 | 0.97 | 0 | 2.4 | 0.009 |
| NiZn3 | 1 | 3.19 | 0 | 2.9 | 0.011 |
| NiZn5 | 1 | 5.09 | 0 | 3.1 | 0.010 |
| NiZn0.5 | 1 | 0.54 | 0 | 3.3 | 0.013 |
| NiZn0.5Ce0.2 | 1 | 0.56 | 0.15 | 8.5 | 0.041 |
| NiZn0.5Ce0.3 | 1 | 0.55 | 0.28 | 6.7 | 0.028 |
| NiZn0.5Ce0.5 | 1 | 0.53 | 0.47 | 7.8 | 0.046 |
| NiZn0.5Ce1 | 1 | 0.52 | 0.83 | 9.1 | 0.049 |
表2 NiZnx及NiZn0.5Cey氧化物催化剂的元素组成、比表面积和孔容
Table 2 Atomic ratio, BET surface area, total pore volume of NiZnx and NiZn0.5Cey oxide catalysts
| 催化剂 | 原子比① | SBET/(m2·g-1) | 孔容/(cm3·g-1) | ||
|---|---|---|---|---|---|
| Ni | Zn | Ce | |||
| NiO | 1 | 0 | 0 | 2.7 | 0.008 |
| NiZn1 | 1 | 0.97 | 0 | 2.4 | 0.009 |
| NiZn3 | 1 | 3.19 | 0 | 2.9 | 0.011 |
| NiZn5 | 1 | 5.09 | 0 | 3.1 | 0.010 |
| NiZn0.5 | 1 | 0.54 | 0 | 3.3 | 0.013 |
| NiZn0.5Ce0.2 | 1 | 0.56 | 0.15 | 8.5 | 0.041 |
| NiZn0.5Ce0.3 | 1 | 0.55 | 0.28 | 6.7 | 0.028 |
| NiZn0.5Ce0.5 | 1 | 0.53 | 0.47 | 7.8 | 0.046 |
| NiZn0.5Ce1 | 1 | 0.52 | 0.83 | 9.1 | 0.049 |
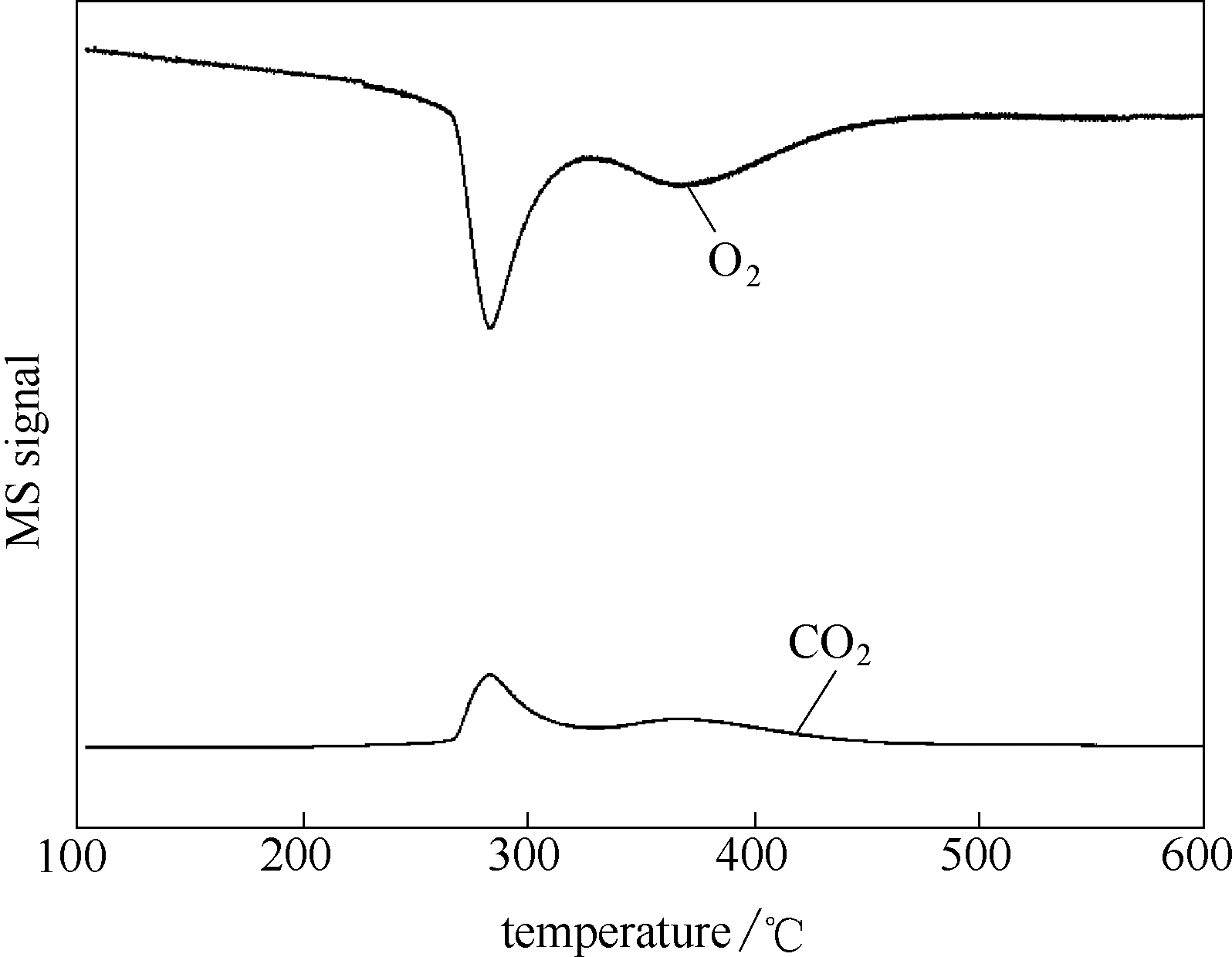
图4 NiZn0.5Ce0.3氧化物催化剂的 TPRO-MS 信号图
Fig.4 MS signals of TPRO of NiZn0.5Ce0.3 oxide catalyst[m/z =32 (?O2), 44(?CO2) were recorded during TPRO analysis]
| 1 | 杨为民. 碳四烃转化与利用技术研究进展及发展前景[J]. 化工进展, 2015, 34(1): 1-9. |
| Yang W M. Progress and perspectives on conversion and utilization of C4 hydrocarbons[J]. Chemical Industry and Engineering Progress, 2015, 34 (1): 1-9. | |
| 2 | 陈伟明. 全球基础石化原料多元化对丁二烯市场的影响[J]. 中国石油和化工经济分析, 2017, (5): 62-64. |
| Chen W M. Impact of global diversification of basic petrochemical raw materials on the butadiene market[J]. Economic Analysis of China Petroleum and Chemical Industry, 2017, (5): 62-64. | |
| 3 | Li X Y, Cheng D G, Chen F Q, et al. Dual bed catalyst system for oxidative dehydrogenation of mixed-butenes: a synergistic mechanism[J].Journal of Zhejiang University-SCIENCE A (Applied Physics & Engineering), 2017, 18(3): 225-233. |
| 4 | 林春光. 正丁烷应用方案研究[J]. 辽宁化工, 2015, (7): 827-829. |
| Lin C G. Research on utilization of butane[J]. Liaoning Chemical Industry, 2015, (7): 827-829. | |
| 5 | 万超. 丁烯氧化脱氢制丁二烯铋钼基催化剂研究[D]. 杭州: 浙江大学, 2015. |
| Wan C. Study on bismuth molybdenum-based catalystsfor oxidative dehydrogenation of 1-butene to 1, 3-butadiene[D]. Hangzhou: Zhejiang University, 2015. | |
| 6 | Li X Y, Cheng D G, Zhao Z J, et al. Temperature-induced deactivation mechanism of ZnFe2O4 for oxidative dehydrogenation of 1-butene[J]. Reaction Chemistry & Engineering, 2017, 2(2): 215-225. |
| 7 | Batiot C, Hodnett B K. The role of reactant and product bond energies in determining limitations to selective catalytic oxidations[J]. Applied Catalysis A General, 1996, 137(1): 179-191. |
| 8 | Slyemi S, Barama A, Barama S, et al. Comparative study of physico-chemical, acid-base and catalytic properties of vanadium based catalysts in the oxidehydrogenation of n-butane: effect of the oxide carrier[J]. Reaction Kinetics, Mechanisms and Catalysis, 2019, 128(2): 831-845. |
| 9 | Zhang J, Liu X, Blume R, et al. Surface-modified carbon nanotubes catalyze oxidative dehydrogenation of n-butane[J]. Science, 2008, 322(5898): 73-77. |
| 10 | Chaar M A, Patel D, Kung M C, et al. Selective oxidative dehydrogenation of butane over V-Mg-O catalysts[J]. Journal of Catalysis, 1987, 105(2): 483-498. |
| 11 | Jackson S D, Rugmini S. Dehydrogenation of n-butane over vanadia catalysts supported on θ-alumina[J]. Journal of Catalysis, 2007, 251: 59-68. |
| 12 | Harlin M E, Niemi V M, Krause A O I, et al. Effect of Mg and Zr modification on the activity of VOx/Al2O3 catalysts in the dehydrogenation of butanes[J]. Journal of Catalysis, 2001, 203: 242-252. |
| 13 | Wang C, Chen J G, Xing T, et al. Vanadium oxide supported on titanosilicates for the oxidative dehydrogenation of n-butane[J]. Industrial & Engineering Chemistry Research, 2015, 54(14): 3602-3610. |
| 14 | 朴昌林. 国内外钒工业的概况及对我国发展钒工业的若干建议[J]. 钢铁钒钛, 1982, (3): 95. |
| Piao C L. Overview of vanadium industry at home and abroad and some suggestions for the development of vanadium industry in my country[J]. Iron Steel Vanadium Titanium, 1982, (3): 95. | |
| 15 | Ducarme V, Martin G A. Low temperature oxidative dehydrogenation of ethane over Ni-based catalysts[J]. Catalysis Letters, 1994, 23(1/2): 97-101. |
| 16 | Schuurman Y, Ducarme V, Chen T, et al. Low temperature oxidative dehydrogenation of ethane over catalysts based on group VIII metals[J]. Applied Catalysis A: General, 1997, 163(1/2): 227-235. |
| 17 | Heracleous E, Lee A F, Wilson K, et al. Investigation of Ni-based alumina-supported catalysts for the oxidative dehydrogenation of ethane to ethylene: structural characterization and reactivity studies[J]. Journal of Catalysis, 2005, 231(1): 159-171. |
| 18 | Tanimu G, Abussaud B A, Asaoka S, et al. Kinetic study on n‑butane oxidative dehydrogenation over the (Ni, Fe, Co)-Bi-O/γ-Al2O3 catalyst[J]. Industrial & Engineering Chemistry Research, 2020, 59(7): 2773-2780. |
| 19 | 林志峰, 胡日茗, 周晓龙. 镍基催化剂的研究进展[J]. 化工学报, 2017, 68: 26-36. |
| Lin Z F, Hu R M, Zhou X L. Research progress of Ni-based catalysts[J]. CIESC Journal, 2017, 68: 26-36. | |
| 20 | Heracleous E, Lemonidou A A. Ni-Me-O mixed metal oxides for the effective oxidative dehydrogenation of ethane to ethylene—effect of promoting metal Me[J]. Journal of Catalysis, 2010, 270(1): 67-75. |
| 21 | Berger-Karin C, Radnik J, Kondratenko E V. Mechanistic origins of the promoting effect of tiny amounts of Rh on the performance of NiOx/Al2O3 in partial oxidation of methane[J]. Journal of Catalysis, 2011, 280(1): 116-124. |
| 22 | Wu Y, Gao J, He Y, et al. Preparation and characterization of Ni-Zr-O nanoparticles and its catalytic behavior for ethane oxidative dehydrogenation[J]. Applied Surface Science, 2012, 258(11): 4922-4928. |
| 23 | Liu Y M, Wang L C, Chen M, et al. Highly selective Ce-Ni-O catalysts for efficient low temperature oxidative dehydrogenation of propane[J]. Catalysis Letters, 2009, 130 (3/4): 350-354. |
| 24 | Jermy B R, Asaoka S, Al-Khattaf S. Influence of calcination on performance of Bi-Ni-O/gamma-alumina catalyst for n-butane oxidative dehydrogenation to butadiene[J]. Catal. Sci. Technol., 2015, 5(9): 4622-4635. |
| 25 | Madeira L M, Portela M F. Catalytic oxidative dehydrogenation of n-butane[J]. Catalysis Reviews, 2002, 44(2): 247-286. |
| 26 | Jermy B R, Ajayi B P, Abussaud B A, et al. Oxidative dehydrogenation of n-butane to butadiene over Bi-Ni-O/γ-alumina catalyst[J]. Journal of Molecular Catalysis A: Chemical, 2015, 400: 121-131. |
| 27 | Elmutasim O, Tanimu G, Aljundi I H, et al. Bimetallic Bi-Ni oxides over carbide supports for oxidative dehydrogenation of n-butane: experimental and kinetic modelling [J]. The Canadian Journal of Chemical Engineering, 2018, 96(6): 1367-1376. |
| 28 | Tanimu G, Jermy B R, Asaoka S, et al. Composition effect of metal species in (Ni, Fe, Co)-Bi-O/gamma-Al2O3 catalyst on oxidative dehydrogenation of n-butane to butadiene [J]. Journal of Industrial and Engineering Chemistry, 2017, 45: 111-120. |
| 29 | Tanimu G, Asaoka S, Al-Khattaf S. Effect of support in Ni-Bi-O/support catalyst on oxidative dehydrogenation of n-butane to butadiene[J]. Molecular Catalysis, 2017, 438: 245-255. |
| 30 | Arndt S, Uysal B, Berthold A, et al. Supported ZnO catalysts for the conversion of alkanes: about the metamorphosis of a heterogeneous catalyst[J]. Journal of Natural Gas Chemistry, 2012, 21(5): 581-594. |
| 31 | Solsona B, Concepción P, Hernández S, et al. Oxidative dehydrogenation of ethane over NiO-CeO2 mixed oxides catalysts[J]. Catalysis Today, 2012, 180(1): 51-58. |
| 32 | Li C, Luo M F, Shen W J, et al. Reduction property and catalytic activity of Ce1-XNiXO2 mixed oxide catalysts for CH4 oxidation[J]. Applied Catalysis A General, 2003, 246(1): 1-9. |
| 33 | Andreeva D, Nedyalkova R, Ilieva L, et al. Gold-vanadia catalysts supported on ceria-alumina for complete benzene oxidation[J]. Applied Catalysis B Environmental, 2004, 52(3): 157-165. |
| 34 | Zhu J, Ommen J G V, Bouwmeester H J M, et al. Activation of O2 and CH4 on yttrium-stabilized zirconia for the partial oxidation of methane to synthesis gas[J]. Journal of Catalysis, 2005, 233(2): 434-441. |
| 35 | Jung J C, Kim H, Choi A S, et al. Effect of pH in the preparation of γ-Bi2MoO6 for oxidative dehydrogenation of n-butene to 1, 3-butadiene: correlation between catalytic performance and oxygen mobility of γ-Bi2MoO6[J]. Catalysis Communications, 2007, 8: 625-628. |
| 36 | Jung J C, Lee H, Park D R, et al. Effect of calcination temperature on the catalytic perfomance of γ-Bi2MoO6 in the oxidative dehydrogenation of n-butene to 1, 3-butadiene [J]. Catalysis Letters, 2009, 131: 401-405. |
| 37 | Li J H, Wang C C, Huang C J, et al. Mesoporous nickel oxides as effective catalysts for oxidative dehydrogenation of propane to propene[J]. Applied Catalysis A General, 2010, 382(1): 99-105. |
| 38 | Abdel-Wahab M S, Jilani A, Yahia I S, et al. Enhanced the photocatalytic activity of Ni-doped ZnO thin films: morphological, optical and XPS analysis[J]. Superlattices and Microstructures, 2016, 94: 108-118. |
| 39 | Tomellini M. X-Ray photoelectron spectra of defective nickel oxide[J]. Journal of the Chemical Society Faraday Transactions Physical Chemistry in Condensed Phases, 1988, 84(10): 3501-3510. |
| 40 | Salagre P, Fierro J L G, Medina F, et al. Characterization of nickel species on several γ-alumina supported nickel samples[J]. Journal of Molecular Catalysis A Chemical, 1996, 106(1/2): 125-134. |
| 41 | Vedrine J C, Hollinger G, Duc T M. Investigations of antigorite and nickel supported catalysts by X-ray photoelectron spectroscopy[J]. J.Phys.Chem., 1978, 82(13): 1515-1520. |
| 42 | Biju V, Khadar M A. Electronic structure of nanostructured nickel oxide using Ni 2p XPS analysis[J]. Journal of Nanoparticle Research, 2002, 4(3): 247-253. |
| 43 | 邢天. 氮化镓催化正丁烷氧化脱氢制烯烃研究[D]. 西安: 陕西师范大学, 2017. |
| Xing T. Study on gallium nitride catalyzed oxidative dehydrogenation of n-butane to olefins[D]. Xi'an: Shaanxi Normal University, 2017. | |
| 44 | Mueller D N, Machala M L, Bluhm H, et al. Redox activity of surface oxygen anions in oxygen-deficient perovskite oxides during electrochemical reactions[J]. Nature Communications, 2015, 6: 6097. |
| 45 | Wan C, Cheng D G, Chen F Q, et al. Oxidative dehydrogenation of 1-butene over vanadium modified bismuth molybdate catalyst: an insight into mechanism[J]. RSC Advances, 2015, 5(53): 42609-42615. |
| [1] | 胡超, 董玉明, 张伟, 张红玲, 周鹏, 徐红彬. 浓硫酸活化五氧化二钒制备高浓度全钒液流电池正极电解液[J]. 化工学报, 2023, 74(S1): 338-345. |
| [2] | 宋瑞涛, 王派, 王云鹏, 李敏霞, 党超镔, 陈振国, 童欢, 周佳琦. 二氧化碳直接蒸发冰场排管内流动沸腾换热数值模拟分析[J]. 化工学报, 2023, 74(S1): 96-103. |
| [3] | 张义飞, 刘舫辰, 张双星, 杜文静. 超临界二氧化碳用印刷电路板式换热器性能分析[J]. 化工学报, 2023, 74(S1): 183-190. |
| [4] | 范孝雄, 郝丽芳, 范垂钢, 李松庚. LaMnO3/生物炭催化剂低温NH3-SCR催化脱硝性能研究[J]. 化工学报, 2023, 74(9): 3821-3830. |
| [5] | 杨百玉, 寇悦, 姜峻韬, 詹亚力, 王庆宏, 陈春茂. 炼化碱渣湿式氧化预处理过程DOM的化学转化特征[J]. 化工学报, 2023, 74(9): 3912-3920. |
| [6] | 陈美思, 陈威达, 李鑫垚, 李尚予, 吴有庭, 张锋, 张志炳. 硅基离子液体微颗粒强化气体捕集与转化的研究进展[J]. 化工学报, 2023, 74(9): 3628-3639. |
| [7] | 程业品, 胡达清, 徐奕莎, 刘华彦, 卢晗锋, 崔国凯. 离子液体基低共熔溶剂在转化CO2中的应用[J]. 化工学报, 2023, 74(9): 3640-3653. |
| [8] | 陈杰, 林永胜, 肖恺, 杨臣, 邱挺. 胆碱基碱性离子液体催化合成仲丁醇性能研究[J]. 化工学报, 2023, 74(9): 3716-3730. |
| [9] | 米泽豪, 花儿. 基于DFT和COSMO-RS理论研究多元胺型离子液体吸收SO2气体[J]. 化工学报, 2023, 74(9): 3681-3696. |
| [10] | 李艺彤, 郭航, 陈浩, 叶芳. 催化剂非均匀分布的质子交换膜燃料电池操作条件研究[J]. 化工学报, 2023, 74(9): 3831-3840. |
| [11] | 陈哲文, 魏俊杰, 张玉明. 超临界水煤气化耦合SOFC发电系统集成及其能量转化机制[J]. 化工学报, 2023, 74(9): 3888-3902. |
| [12] | 杨学金, 杨金涛, 宁平, 王访, 宋晓双, 贾丽娟, 冯嘉予. 剧毒气体PH3的干法净化技术研究进展[J]. 化工学报, 2023, 74(9): 3742-3755. |
| [13] | 李锦潼, 邱顺, 孙文寿. 煤浆法烟气脱硫中草酸和紫外线强化煤砷浸出过程[J]. 化工学报, 2023, 74(8): 3522-3532. |
| [14] | 杨菲菲, 赵世熙, 周维, 倪中海. Sn掺杂的In2O3催化CO2选择性加氢制甲醇[J]. 化工学报, 2023, 74(8): 3366-3374. |
| [15] | 洪瑞, 袁宝强, 杜文静. 垂直上升管内超临界二氧化碳传热恶化机理分析[J]. 化工学报, 2023, 74(8): 3309-3319. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备 11010102001995号
京公网安备 11010102001995号