化工学报 ›› 2020, Vol. 71 ›› Issue (8): 3556-3564.DOI: 10.11949/0438-1157.20200172
李倩倩1( ),唐思扬1(
),唐思扬1( ),岳海荣1,2,刘长军1,马奎1,钟山1,梁斌1,2
),岳海荣1,2,刘长军1,马奎1,钟山1,梁斌1,2
收稿日期:2020-02-24
修回日期:2020-05-22
出版日期:2020-08-05
发布日期:2020-08-05
通讯作者:
唐思扬
作者简介:李倩倩(1994—),女,硕士研究生,基金资助:
Qianqian LI1( ),Siyang TANG1(
),Siyang TANG1( ),Hairong YUE1,2,Changjun LIU1,Kui MA1,Shan ZHONG1,Bin LIANG1,2
),Hairong YUE1,2,Changjun LIU1,Kui MA1,Shan ZHONG1,Bin LIANG1,2
Received:2020-02-24
Revised:2020-05-22
Online:2020-08-05
Published:2020-08-05
Contact:
Siyang TANG
摘要:
采用浸渍还原方法制备Pd-Rh/TiO2催化剂用于常温光催化CO2氧化乙烷脱氢制C2H4。研究不同Pd/Rh比催化剂的光反应性能,利用XRD、EDX-mapping、TEM、HRTEM、XPS技术表征催化剂表面和电子特性,通过UV-Vis、PL技术考察催化剂光响应性能,采用原位红外光谱技术分析Pd-Rh/TiO2光催化CO2氧化乙烷脱氢反应机理。研究表明,Pd-Rh双金属体系可有效提高光反应活性,光照下Pd和Rh金属之间存在内部电子转移的作用,降低了Pd表面的电子云密度,促进光生电子和空穴的分离,同时促进了C2H6和CO2在材料表面的吸附。Ar替换CO2的对比实验证明,反应中的CO2消耗H2,可消除催化剂表面积碳,促进C2H4生成。
中图分类号:
李倩倩, 唐思扬, 岳海荣, 刘长军, 马奎, 钟山, 梁斌. Pd-Rh/TiO2光催化CO2氧化乙烷脱氢研究[J]. 化工学报, 2020, 71(8): 3556-3564.
Qianqian LI, Siyang TANG, Hairong YUE, Changjun LIU, Kui MA, Shan ZHONG, Bin LIANG. Study on the photocatalytic oxidative dehydrogenation of ethane with CO2 over Pd-Rh /TiO2 catalyst[J]. CIESC Journal, 2020, 71(8): 3556-3564.
| 催化剂 | 活性/(μmol·(g cat)-1·h-1) | |||
|---|---|---|---|---|
| H2 | CH4 | C2H4 | CO | |
| Pd | 122.2 | 126.1 | 226.0 | 74.8 |
| 1Pd-1Pt | 461.2 | 214.9 | 222.9 | trace |
| 1Pd-1Ni | 150.2 | 179.6 | 134.3 | trace |
| 1Pd-1Fe | 56.1 | 130.9 | 91.4 | 18.3 |
| 1Pd-1Cu | 417.1 | 149.9 | 294.2 | 25.2 |
| 1Pd-1Ru | 88.7 | 134.2 | 273.9 | 96.3 |
| 1Pd-1Ag | 798.3 | 134.2 | 510.7 | 23.8 |
| 1Pd-1Rh | 467.6 | 200.3 | 428.8 | 190.2 |
表1 Pd-M/TiO2双金属催化剂在CO2存在条件下的氧化乙烷脱氢性能评价
Table 1 Performance of oxidative dehydrogenation of ethane in the presence of CO2 over Pd-M /TiO2 catalysts
| 催化剂 | 活性/(μmol·(g cat)-1·h-1) | |||
|---|---|---|---|---|
| H2 | CH4 | C2H4 | CO | |
| Pd | 122.2 | 126.1 | 226.0 | 74.8 |
| 1Pd-1Pt | 461.2 | 214.9 | 222.9 | trace |
| 1Pd-1Ni | 150.2 | 179.6 | 134.3 | trace |
| 1Pd-1Fe | 56.1 | 130.9 | 91.4 | 18.3 |
| 1Pd-1Cu | 417.1 | 149.9 | 294.2 | 25.2 |
| 1Pd-1Ru | 88.7 | 134.2 | 273.9 | 96.3 |
| 1Pd-1Ag | 798.3 | 134.2 | 510.7 | 23.8 |
| 1Pd-1Rh | 467.6 | 200.3 | 428.8 | 190.2 |
| 催化剂① | Pd负载量②/ %(质量) | Rh负载量②/ %(质量) | 活性(CO2下)/(μmol·(g cat)-1·h-1)③ | 活性(Ar下)/(μmol·(g cat) -1·h-1)④ | |||||
|---|---|---|---|---|---|---|---|---|---|
| H2 | CH4 | C2H4 | CO | H2 | CH4 | C2H4 | |||
| Pd | 0.44 | 0 | 122.2 | 126.1 | 226.0 | 74.8 | 760.7 | 155.6 | 49.8 |
| 3Pd-1Rh | 0.25 | 0.16 | 284.9 | 57.9 | 284.6 | 134.1 | 947.0 | 144.7 | 68.1 |
| 1Pd-1Rh | 0.21 | 0.39 | 467.6 | 200.3 | 428.8 | 190.2 | 1102.9 | 134.8 | 72.6 |
| 1Pd-3Rh | 0.07 | 0.34 | 427.4 | 47.4 | 336.4 | 155.5 | 1533.8 | 136.8 | 161.6 |
| Rh | 0.70 | 0 | 190.9 | 48.6 | 170.1 | 269.7 | 1908.9 | 147.8 | 43.9 |
| P25 | —⑤ | — | 0.3 | 16.7 | 10.1 | trace | — | — | — |
表2 不同金属比例Pd-Rh/TiO2分别在CO2与Ar条件下的乙烷脱氢性能评价
Table 2 Ethane dehydrogenation performance under CO2 and Ar conditions with different metal ratios of Pd-Rh/TiO2
| 催化剂① | Pd负载量②/ %(质量) | Rh负载量②/ %(质量) | 活性(CO2下)/(μmol·(g cat)-1·h-1)③ | 活性(Ar下)/(μmol·(g cat) -1·h-1)④ | |||||
|---|---|---|---|---|---|---|---|---|---|
| H2 | CH4 | C2H4 | CO | H2 | CH4 | C2H4 | |||
| Pd | 0.44 | 0 | 122.2 | 126.1 | 226.0 | 74.8 | 760.7 | 155.6 | 49.8 |
| 3Pd-1Rh | 0.25 | 0.16 | 284.9 | 57.9 | 284.6 | 134.1 | 947.0 | 144.7 | 68.1 |
| 1Pd-1Rh | 0.21 | 0.39 | 467.6 | 200.3 | 428.8 | 190.2 | 1102.9 | 134.8 | 72.6 |
| 1Pd-3Rh | 0.07 | 0.34 | 427.4 | 47.4 | 336.4 | 155.5 | 1533.8 | 136.8 | 161.6 |
| Rh | 0.70 | 0 | 190.9 | 48.6 | 170.1 | 269.7 | 1908.9 | 147.8 | 43.9 |
| P25 | —⑤ | — | 0.3 | 16.7 | 10.1 | trace | — | — | — |
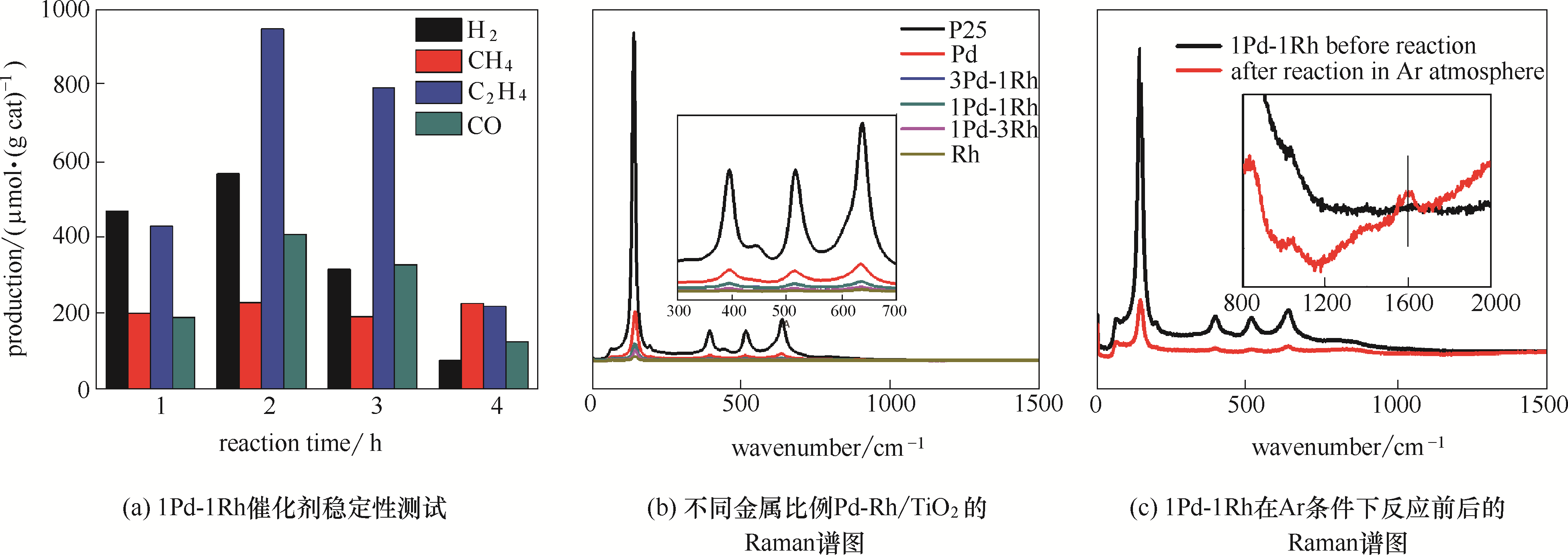
图1 催化剂稳定性测试和积碳检测(稳定性测试条件:温度 298 K;压力0.2 MPa;反应气C2H6∶CO2=1∶1;1Pd-1Rh催化剂用量25 mg循环利用)
Fig.1 Stability test and carbon deposition test(stability test reaction condition:298 K, 0.2 MPa, C2H6∶CO2=1∶1, 25 mg 1Pd-1Rh catalyst for recycling)
| 1 | Baños R, Manzano-Agugliaro F, Montoya F G, et al. Optimization methods applied to renewable and sustainable energy: a review [J]. Renewable and Sustainable Energy Reviews, 2011, 15(4): 1753-1766. |
| 2 | Porosoff M D, Myint M N Z, Kattel S, et al. Identifying different types of catalysts for CO2 reduction by ethane through dry reforming and oxidative dehydrogenation [J]. Angewandte Chemie International Edition, 2015, 54(51): 15501-15505. |
| 3 | Mimura N, Takahara I, Inaba M, et al. High-performance Cr/H-ZSM-5 catalysts for oxidative dehydrogenation of ethane to ethylene with CO2 as an oxidant [J]. Catalysis Communications, 2002, 3(6): 257-262. |
| 4 | Shi L, Wang Y, Yan B, et al. Progress in selective oxidative dehydrogenation of light alkanes to olefins promoted by boron nitride catalysts [J]. Chemical Communications, 2018, 54(78): 10936-10946. |
| 5 | Ayoub M, Abdullah A Z. Critical review on the current scenario and significance of crude glycerol resulting from biodiesel industry towards more sustainable renewable energy industry [J]. Renewable and Sustainable Energy Reviews, 2012, 16(5): 2671-2686. |
| 6 | Ren T, Patel M, Blok K. Olefins from conventional and heavy feedstocks: energy use in steam cracking and alternative processes [J]. Energy, 2006, 31(4): 425-451. |
| 7 | Roy S C, Varghese O K, Paulose M, et al. Toward solar fuels: photocatalytic conversion of carbon dioxide to hydrocarbons [J]. ACS Nano, 2010, 4(3): 1259-1278. |
| 8 | Rahmani F, Haghighi M, Amini M. The beneficial utilization of natural zeolite in preparation of Cr/clinoptilolite nanocatalyst used in CO2-oxidative dehydrogenation of ethane to ethylene [J]. Journal of Industrial and Engineering Chemistry, 2015, 31: 142-155. |
| 9 | Cheng Y, Zhang F, Zhang Y, et al. Oxidative dehydrogenation of ethane with CO2 over Cr supported on submicron ZSM-5 zeolite [J]. Chinese Journal of Catalysis, 2015, 36(8): 1242-1248. |
| 10 | Shen Z, Liu J, Xu H, et al. Dehydrogenation of ethane to ethylene over a highly efficient Ga2O3/HZSM-5 catalyst in the presence of CO2 [J]. Applied Catalysis A: General, 2009, 356(2): 148-153. |
| 11 | Gärtner C A, van Veen A C, Lercher J A. Oxidative dehydrogenation of ethane: common principles and mechanistic aspects [J]. ChemCatChem, 2013, 5(11): 3196-3217. |
| 12 | Cheng Y, Miao C, Hua W, et al. Cr/ZSM-5 for ethane dehydrogenation: enhanced catalytic activity through surface silanol [J]. Applied Catalysis A: General, 2017, 532: 111-119. |
| 13 | Skoufa Z, Heracleous E, Lemonidou A A. On ethane ODH mechanism and nature of active sites over NiO-based catalysts via isotopic labeling and methanol sorption studies [J]. Journal of Catalysis, 2015, 322: 118-129. |
| 14 | Zhang R, Wang H, Tang S, et al. Photocatalytic oxidative dehydrogenation of ethane using CO2 as a soft oxidant over Pd/TiO2 catalysts to C2H4 and syngas [J]. ACS Catalysis, 2018, 8(10): 9280-9286. |
| 15 | Leung D Y C, Fu X, Wang C, et al. Hydrogen production over titania-based photocatalysts [J]. ChemSusChem, 2010, 3(6): 681-694. |
| 16 | Wang W N, An W J, Ramalingam B, et al. Size and structure matter: enhanced CO2 photoreduction efficiency by size-resolved ultrafine Pt nanoparticles on TiO2 single crystals [J]. Journal of the American Chemical Society, 2012, 134(27): 11276-11281. |
| 17 | Asahi R, Morikawa T, Ohwaki T, et al. Visible-light photocatalysis in nitrogen-doped titanium oxides [J]. Science, 2001, 293: 269-271. |
| 18 | Chen X, Liu L, Yu P, et al. Increasing solar absorption for photocatalysis with black hydrogenated titanium dioxide nanocrystals [J]. Science, 2011, 331: 746-750. |
| 19 | Maeda K, Domen K. Photocatalytic water splitting: recent progress and future challenges [J]. Journal of Physical Chemistry Letters, 2010, 1(18): 2655-2661. |
| 20 | Foo W J, Zhang C, Ho G W. Non-noble metal Cu-loaded TiO2 for enhanced photocatalytic H2 production [J]. Nanoscale, 2013, 5(2): 759-764. |
| 21 | Wang C, Thompson R, Ohodnicki P R, et al. Size-dependent photocatalytic reduction of CO2 with PbS quantum dot sensitized TiO2 heterostructured photocatalysts [J]. Journal of Materials Chemistry, 2011, 21: 13452-13457. |
| 22 | Li X, Zhuang Z, Li W, et al. Photocatalytic reduction of CO2 over noble metal-loaded and nitrogen-doped mesoporous TiO2 [J]. Applied Catalysis A: General, 2012, (s429/430): 31-38. |
| 23 | Murdoch M, Waterhouse G, Nadeem M, et al. The effect of gold loading and particle size on photocatalytic hydrogen production from ethanol over Au/TiO2 nanoparticles [J]. Nature Chemistry, 2011, 3: 489-492. |
| 24 | Bera S, Lee J E, Rawal S B, et al. Size-dependent plasmonic effects of Au and Au@SiO2 nanoparticles in photocatalytic CO2 conversion reaction of Pt/TiO2 [J]. Applied Catalysis B: Environmental, 2016, 199: 55-63. |
| 25 | Linic S, Christopher P, Ingram D. Plasmonic-metal nanostructures for efficient conversion of solar to chemical energy [J]. Nature Materials, 2011, 10: 911-921. |
| 26 | Ide Y, Matsuoka M, Ogawa M. Efficient visible-light-induced photocatalytic activity on gold-nanoparticle-supported layered titanate [J]. Journal of the American Chemical Society, 2010, 132: 16762-16764. |
| 27 | Yu L, Li D. Photocatalytic methane conversion coupled with hydrogen evolution from water over Pd/TiO2 [J]. Catalysis Science & Technology, 2017, 7(3): 635-640. |
| 28 | Konda S K, Amiri M, Chen A. Photoassisted deposition of palladium nanoparticles on carbon nitride for efficient oxygen reduction [J]. The Journal of Physical Chemistry C, 2016, 120(27): 14467-14473. |
| 29 | Hou W, Cronin S B. A review of surface plasmon resonance-enhanced photocatalysis [J]. Advanced Functional Materials, 2013, 23(13): 1612-1619. |
| 30 | Zhao J, Jin R. Heterogeneous catalysis by gold and gold-based bimetal nanoclusters [J]. Nano Today, 2018, 18: 86-102. |
| 31 | Li G, Wang X, Yan L, et al. PdPt bimetal-functionalized SnO2 nanosheets: controllable synthesis and its dual selectivity for detection of carbon monoxide and methane [J]. ACS Applied Materials & Interfaces, 2019, 11(29): 26116-26126. |
| 32 | Ren X Y, Cao J P, Zhao X Y, et al. Catalytic upgrading of pyrolysis vapors from lignite over mono/bimetal-loaded mesoporous HZSM-5 [J]. Fuel, 2018, 218: 33-40. |
| 33 | Feng X, Bo X, Guo L. CoM(M=Fe,Cu,Ni)-embedded nitrogen-enriched porous carbon framework for efficient oxygen and hydrogen evolution reactions [J]. Journal of Power Sources, 2018, 389: 249-259. |
| 34 | Zhang X F, Meng H B, Chen H Y, et al. Bimetallic PtCo alloyed nanodendritic assemblies as an advanced efficient and robust electrocatalyst for highly efficient hydrogen evolution and oxygen reduction [J]. Journal of Alloys and Compounds, 2019, 786: 232-239. |
| 35 | Li C, Zhang B, Li Y, et al. Self-assembled Cu-Ni bimetal oxide 3D in-plane epitaxial structures for highly efficient oxygen evolution reaction [J]. Applied Catalysis B: Environmental, 2019, 244: 56-62. |
| 36 | Zhu W, Zhu G, Yao C, et al. Porous amorphous FeCo alloys as pre-catalysts for promoting the oxygen evolution reaction [J]. Journal of Alloys and Compounds, 2020, 828: 154465. |
| 37 | Shao Q, Wang P, Huang X. Opportunities and challenges of interface engineering in bimetallic nanostructure for enhanced electrocatalysis [J]. Advanced Functional Materials, 2019, 29(3): 1806419. |
| 38 | Ali S, Chen L, Li Z, et al. Cux-Nb1.1-x (x = 0.45, 0.35, 0.25, 0.15) bimetal oxides catalysts for the low temperature selective catalytic reduction of NO with NH3 [J]. Applied Catalysis B: Environmental, 2018, 236: 25-35. |
| 39 | Liu M, Zhang Q, Shi Y, et al. AuPd bimetal immobilized on amine-functionalized SBA-15 for hydrogen generation from formic acid: the effect of the ratio of toluene to DMF [J]. The Canadian Journal of Chemical Engineering, 2020, 98(4): 879-891. |
| 40 | Li S, Gong D, Tang H, et al. Preparation of bimetallic Ni@Ru nanoparticles supported on SiO2 and their catalytic performance for CO methanation [J]. Chemical Engineering Journal, 2018, 334: 2167-2178. |
| 41 | Ma Y, Yin L, Cao G, et al. Pt-Pd bimetal popcorn nanocrystals: enhancing the catalytic performance by combination effect of stable multipetals nanostructure and highly accessible active sites [J]. Small, 2018, 14(14): 1703613. |
| 42 | Xiao S, Pan D, Liang R, et al. Bimetal MOF derived mesocrystal ZnCo2O4 on rGO with high performance in visible-light photocatalytic NO oxidization [J]. Applied Catalysis B: Environmental, 2018, 236: 304-313. |
| 43 | Han C, Tang Z R, Liu J, et al. Efficient photoredox conversion of alcohol to aldehyde and H2 by heterointerface engineering of bimetal–semiconductor hybrids [J]. Chemical Science, 2019, 10(12): 3514-3522. |
| 44 | Matsubu J C, Zhang S, DeRita L, et al. Adsorbate-mediated strong metal-support interactions in oxide-supported Rh catalysts [J]. Nat. Chem., 2017, 9(2): 120-127. |
| 45 | Ola O, Maroto-Valer M, Liu D, et al. Performance comparison of CO2 conversion in slurry and monolith photoreactors using Pd and Rh-TiO2 catalyst under ultraviolet irradiation [J]. Applied Catalysis B: Environmental, 2012, 126: 172-179. |
| 46 | Martin N M, Velin P, Skoglundh M, et al. Catalytic hydrogenation of CO2 to methane over supported Pd, Rh and Ni catalysts [J]. Catalysis Science & Technology, 2017, 7(5): 1086-1094. |
| 47 | Halasi G, Tóth A, Bánsági T, et al. Production of H2 in the photocatalytic reactions of ethane on TiO2 -supported noble metals [J]. International Journal of Hydrogen Energy, 2016, 41(31): 13485-13492. |
| 48 | Zhang M, Shao C, Guo Z, et al. Hierarchical nanostructures of copper(II) phthalocyanine on electrospun TiO2 nanofibers: controllable solvothermal-fabrication and enhanced visible photocatalytic properties [J]. ACS Applied Materials & Interfaces, 2011, 3: 369-377. |
| [1] | 张义飞, 刘舫辰, 张双星, 杜文静. 超临界二氧化碳用印刷电路板式换热器性能分析[J]. 化工学报, 2023, 74(S1): 183-190. |
| [2] | 胡超, 董玉明, 张伟, 张红玲, 周鹏, 徐红彬. 浓硫酸活化五氧化二钒制备高浓度全钒液流电池正极电解液[J]. 化工学报, 2023, 74(S1): 338-345. |
| [3] | 宋瑞涛, 王派, 王云鹏, 李敏霞, 党超镔, 陈振国, 童欢, 周佳琦. 二氧化碳直接蒸发冰场排管内流动沸腾换热数值模拟分析[J]. 化工学报, 2023, 74(S1): 96-103. |
| [4] | 吴雷, 刘姣, 李长聪, 周军, 叶干, 刘田田, 朱瑞玉, 张秋利, 宋永辉. 低阶粉煤催化微波热解制备含碳纳米管的高附加值改性兰炭末[J]. 化工学报, 2023, 74(9): 3956-3967. |
| [5] | 米泽豪, 花儿. 基于DFT和COSMO-RS理论研究多元胺型离子液体吸收SO2气体[J]. 化工学报, 2023, 74(9): 3681-3696. |
| [6] | 李艺彤, 郭航, 陈浩, 叶芳. 催化剂非均匀分布的质子交换膜燃料电池操作条件研究[J]. 化工学报, 2023, 74(9): 3831-3840. |
| [7] | 陈美思, 陈威达, 李鑫垚, 李尚予, 吴有庭, 张锋, 张志炳. 硅基离子液体微颗粒强化气体捕集与转化的研究进展[J]. 化工学报, 2023, 74(9): 3628-3639. |
| [8] | 程业品, 胡达清, 徐奕莎, 刘华彦, 卢晗锋, 崔国凯. 离子液体基低共熔溶剂在转化CO2中的应用[J]. 化工学报, 2023, 74(9): 3640-3653. |
| [9] | 陈杰, 林永胜, 肖恺, 杨臣, 邱挺. 胆碱基碱性离子液体催化合成仲丁醇性能研究[J]. 化工学报, 2023, 74(9): 3716-3730. |
| [10] | 范孝雄, 郝丽芳, 范垂钢, 李松庚. LaMnO3/生物炭催化剂低温NH3-SCR催化脱硝性能研究[J]. 化工学报, 2023, 74(9): 3821-3830. |
| [11] | 杨百玉, 寇悦, 姜峻韬, 詹亚力, 王庆宏, 陈春茂. 炼化碱渣湿式氧化预处理过程DOM的化学转化特征[J]. 化工学报, 2023, 74(9): 3912-3920. |
| [12] | 陈哲文, 魏俊杰, 张玉明. 超临界水煤气化耦合SOFC发电系统集成及其能量转化机制[J]. 化工学报, 2023, 74(9): 3888-3902. |
| [13] | 杨学金, 杨金涛, 宁平, 王访, 宋晓双, 贾丽娟, 冯嘉予. 剧毒气体PH3的干法净化技术研究进展[J]. 化工学报, 2023, 74(9): 3742-3755. |
| [14] | 杨绍旗, 赵淑蘅, 陈伦刚, 王晨光, 胡建军, 周清, 马隆龙. Raney镍-质子型离子液体体系催化木质素平台分子加氢脱氧制备烷烃[J]. 化工学报, 2023, 74(9): 3697-3707. |
| [15] | 孟令玎, 崇汝青, 孙菲雪, 孟子晖, 刘文芳. 改性聚乙烯膜和氧化硅固定化碳酸酐酶[J]. 化工学报, 2023, 74(8): 3472-3484. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公网安备 11010102001995号
京公网安备 11010102001995号